Onchocerciasis
Hugh R. Taylor
Eric Pearlman
DEFINITION
Onchocerciasis, also known as “river blindness,” is an insect-borne disease caused by a nematode worm, Onchocerca volvulus, and transmitted by blackflies of the genus Simulium. It is endemic across much of sub-Saharan Africa, in circumscribed areas of Central and South America, and in the Arabic Peninsula. Recent estimates suggest that worldwide, at least 120 million persons are exposed to the disease and 18 million persons are infected.1 More than 99% of these live in sub-Saharan Africa and at least 1 million persons are either blind or severely disabled by the disease.
Onchocerciasis has a devastating impact in hyperendemic areas, where almost every person will be infected, and half of the population will be blinded by the disease before they die (Fig. 62.1). Once blind, they have a life expectancy of only one third that of the sighted, and most die within 10 years.2
LIFE CYCLE OF O. VOLVULUS
The Vector
Although the species of blackfly vary in different geographic areas, they are all members of the family Simuliidae. In Africa, the chief vector of O. volvulus is Simulium damnosum, a species complex made up of 30 or more closely related cytospecies (Fig. 62.2). Other important vectors include Simulium naevi, which occurs mainly in East Africa, and Simulium albivirgulatum, considered an important vector in the “Cuvette Centrale” of the Democratic Republic of the Congo. In the Americas, the main vectors are the Simulium ochraceum complex in Guatemala and Mexico, Simulium exiguum in Ecuador, Simulium metallicum and S. exiguum in Venezuela, and Simulium guianense in the “Amazonia” focus.1
The blackfly larvae require well-oxygenated water to mature, and eggs are laid in rapids in fast flowing rivers and streams. Because of seasonal variation in volume and rate of flow, a particular stretch of river may be either a permanent breeding site or a temporary breeding site only at certain times of the year. Blackflies usually do not travel more than 1 or 2 km from their breeding sites, although at times they may be carried hundreds of kilometers by monsoonal winds. Female black flies require a blood meal to initiate ovulation, and it is during this meal that they may transmit or receive the onchocercal infection.
The Parasite
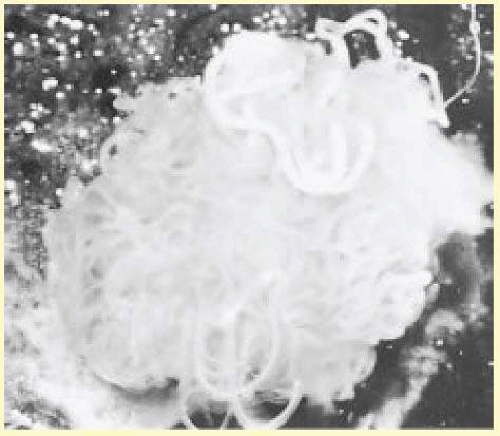
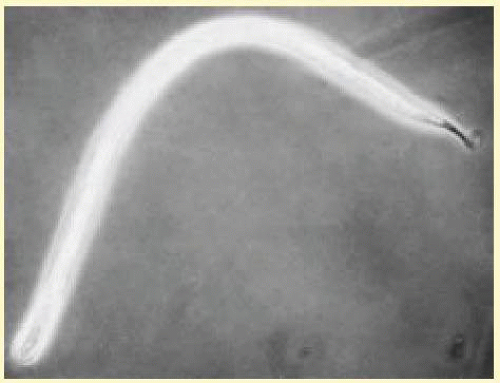
O. volvulus is one of a group of filarial nematodes. Other members include Wuchereria bancrofti and Brugia malayi, which produce lymphatic filariasis and elephantiasis and Loa loa, which causes loiasis. The adult worms live encysted in fibrous nodules, which may be single or conglomerate. Each nodule contains between two to three female worms lying in a twisted, tangled mass (hence the term volvulus). The adult female O. volvulus organism is a white threadlike worm, 30 to 70 cm long and only 0.3 mm across (Fig. 62.3). Adult males have a similar appearance but are thinner and much shorter (about 2.5 to 5 cm long).
 FIG. 62.2 Simulium damnosum, the biting black fly that transmits onchocerciasis in Africa. (Photo courtesy of Charles D. Mackenzie, PhD.) |
Adult female worms have a life span of 8 to 10 years, but may live up to 15 years. During their lifetime, each releases millions of first-stage larvae, also known as microfilariae. A microfilaria is 320 to 360 µm long and 5 to 10 µm in diameter (Fig. 62.4) and lives for 6 to 30 months.1,2 In hyperendemic areas, the total microfilaria load in the body of affected individuals may be as high as 150 million.3
CYCLE OF INFECTION
Microfilariae enter a female blackfly when she bites an infected person. A small percentage of these pass to the fly’s midgut, penetrate the gut wall, and enter its thoracic muscles. After several molts, they become third-stage infective larvae, which then migrate to the fly’s proboscis and are ready to be transferred during the next blood meal. Flies infected during their first blood meal carry developing larvae during their second blood meal, but they are not infectious until their third blood meal. Most flies die after their third reproductive cycle.
After entering the skin of the human host through the bite of an infected blackfly, the infective larvae (usually two to six) migrate through the subcutaneous tissues. They rapidly molt to become fourth-stage larvae and then molt again 4 to 6 weeks later. Over a period of 12 months, each larva develops into a mature adult male or female worm. Adult worms become encapsulated by host fibrous tissue to form characteristic nodules (onchocercomas). The adult worms reproduce sexually, and new microfilariae escape through the nodule wall and migrate throughout the host’s body, particularly the skin. If they are then taken up by a biting blackfly, microfilariae develop into larvae and the cycle continues. There is no replication of the parasite during its development in the vector or during its early development in the human host. One microfilaria can, therefore, only develop into one adult worm. Repeated infections, often accumulated over a period of many years of exposure are required before a heavy load of adult worms and pathogenic microfilariae can build up in the human host.3
NATURAL HISTORY AND EPIDEMIOLOGY
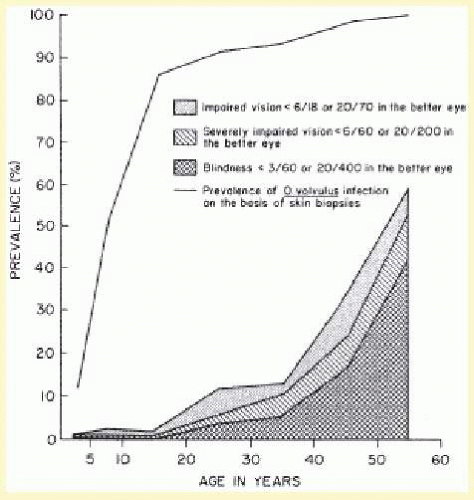
Longitudinal studies carried out in areas of ongoing transmission have shown that onchocerciasis is a progressive disease, and the intensity of infection in a person is the cumulative result of many years of exposure.4,5,6 In these areas, most persons are infected with O. volvulus during the first years of life (Fig. 62.5). The intensity of infection and the number of microfilariae per milligram of skin snip increase with host age. Men are often more heavily infected than women, but this is usually related to different occupational exposures. As a general rule, the closer a person is to the breeding sites and the longer the time spent nearby, the higher the exposure.
About one third of persons older than 15 years have microfilariae in the anterior chamber of the eye. Intraocular microfilariae are more common in those who have high skin-snip counts. In the worst affected areas, 10% of the population and half of those older than 40 years may be blind.1 Severe eye lesions and blindness are more common in heavily infected persons.
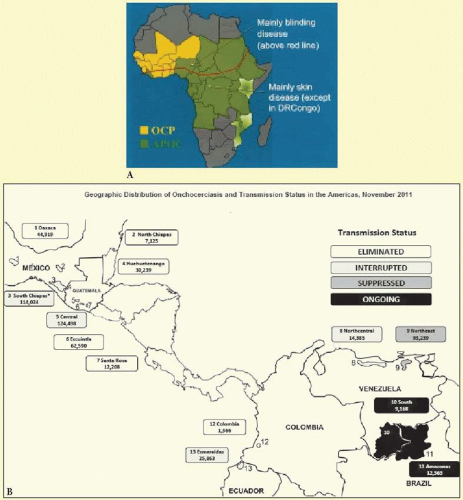
The geographic distribution of onchocerciasis is characterized by local foci of disease. Foci may vary in intensity, but are always located in the vicinity of rivers or streams that form the breeding sites of the blackflies. One measure of the risk of infection is the annual transmission potential (ATP), which is the calculated number of infective larvae transmitted to a person who is continuously exposed to blackflies for 1 year. In the worst areas, the ATP may be 90,000 infective larvae per person per year. ATPs of greater than 1,500 are associated with a high prevalence of blindness, and in West Africa, before current control programs, values over 2,500 were often associated with the subsequent desertion of the involved villages by the populace.1
Regional geographic differences also occur, and these are explained by variation in transmission and by local variation in the parasite-vector complex, although host factors may also be involved3 (Table 62.1 and Fig. 62.6). There are a number of different strains of O. volvulus, each of which is associated with, and transmitted by, a particular species of Simulium, which also vary from region to region. Most of these can now be identified and characterized using deoxyribonucleic acid (DNA) detection techniques.7
PATHOGENESIS OF HOST DISEASE
Death of Microfilariae
Nearly all the lesions of onchocerciasis and their resulting pathologic changes, including those in the eye, are directly or indirectly related to the local death of microfilariae. Access of microfilariae into the eye may occur by a number of different routes.8 It seems likely that they enter the cornea from the skin by way of the conjunctiva. Those in the uveal tract and aqueous may well be blood-borne, although direct invasion along the vessels and nerves also occurs.
Generally, live microfilariae stimulate very little inflammatory response. They appear to pass undetected by the host’s immune system despite the presence of a demonstrable host antibody and cell-mediated immune response.9,10 Inflammation is associated with dead or dying microfilariae and is probably initiated by the release of, or exposure to, microfilarial antigens and Wolbachia endobacteria. The mechanisms that protect live microfilariae are unclear. More heavily infected persons show evidence of immunosuppression, which can be reversed with treatment.
TABLE 62-1 Comparison of General Characteristics of Onchocerciasis in Savanna and Rain Forest Areas of West Africa and Central America | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
In the untreated disease, a relatively small number of microfilariae are dying at any one time, and each initiates a limited inflammatory response. With time, many microfilariae die, and, cumulatively, this produces considerable local inflammation and scarring.
Contribution of Endosymbiotic Wolbachia Bacteria to Disease Manifestations
In 1977, Kozek and Marroquin examined adult O. volvulus worms by transmission electron microscopy and reported that they contained Chlamydia like elementary bodies.11 Twenty years later, Bandi et al. and Taylor et al. used molecular typing to identify them as Wolbachia, which are Rickettsia like bacteria found in insects and other arthropods.12,13 Wolbachia were subsequently shown to inhabit the hypodermis of L4 stage larvae and adult male and female worms, and are transmitted transovarially where they are detected in microfilariae in the uterus.14 The bacteria are therefore transported when the microfilariae migrate through the skin, and Wolbachia are released and the larvae die. Using a mouse model of ocular onchocerciasis, it was demonstrated that corneal disease induced by O.volvulus soluble extracts did not occur unless Wolbachia were present, thereby implicating these bacteria in the pathogenesis of Onchocerca keratitis.15,16 The potential for targeting Wolbachia for antibiotic treatment of patients is discussed below.
The Mazzotti Reaction
Many microfilaricidal drugs used in the past, such as diethylcarbamazine (DEC), killed most microfilariae almost immediately. The synchronous death of millions of microfilariae was believed to release a massive quantity of antigenic or toxic material. This was associated with a marked inflammatory response, which was recognized clinically as the Mazzotti reaction.17 In those with onchocerciasis, DEC almost always caused side effects that, although sometimes mild, could be very severe2 and even precipitate irreversible vision loss. A few deaths have also been reported with its use.
The administration of even a single 50-mg dose of DEC (Mazzotti test) can initiate the previously mentioned response. This was a dramatic and dangerous method of diagnosing onchocerciasis in the past and its use can no longer be recommended.
PATHOLOGY
Skin
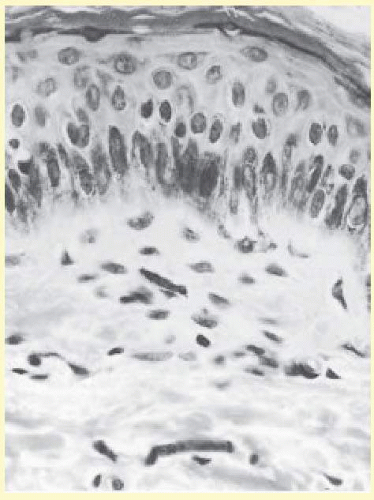
The earliest changes in the skin in onchocerciasis are mild and are often limited to a perivasculitis. In more heavily infected persons, microfilariae can be found, especially at the epidermal-dermal junction (Fig. 62.7). Live microfilariae are usually not surrounded by inflammatory cells.
Later changes include hyperkeratosis, acanthosis, and parakeratosis, with an increase in inflammation and fibrosis. Dermal collagen is disrupted by the deposition of increasing amounts of mucin ground substance and by fibrous scar tissue. With more advanced fibrosis and inflammation, microfilariae become less common in the superficial dermis and are found mainly in the deeper layers. Ultimately, the dermis becomes fibrotic and is covered by a thinned and atrophic epithelium. There are areas of depigmentation and hyperpigmentation (Fig. 62.8). Some patients have a much more severe, reactive onchodermatitis, often confined to one limb. This was first described in Yemen and is called “sowda,” or black limb. Here, the most prominent histologic change is an extensive infiltrate of plasma cells. Microfilariae are seen occasionally in the deep dermis or in skin snips. These patients have a high cellular immune response to onchocercal antigens, which is suppressed if their disease becomes generalized.
Nodules
Adult worms may at times be found free in fascial planes, although they are far more commonly encapsulated in nodules. Some nodules are palpable and visible, lying subcutaneously over bony prominences, especially the iliac crests, greater trochanters, ribs, knees, ankles, coccyx, and skull. Others, probably more numerous, lie deep and impalpable, attached to the fibrous capsules of joints (especially the hip joint) or close to the periosteum of bones or in intramuscular fasciae. Occasionally, nodules are found in even deeper structures such as intracranially or in the wall of the aorta.
When examined microscopically, dense scar tissue surrounds and encases the adult worms, which are coiled up like a ball of string. Developing microfilariae can be found in the uteri of most female worms. Degenerative females often become calcified. Nodules may show an inflammatory infiltrate of varying intensity and occasionally areas of necrosis and liquefaction.
The Eye
As in the skin, many of the changes in the front of the eye at least are related to invasion by, and local death of, microfilariae. The pathophysiology of chorioretinal disease is far less clear and may also be related to immune complex disease,10,18 the presence of anti-retinal antibodies, or other mechanisms.19,20 Much of the chorioretinal disease seems to start at the level of the retinal pigment epithelium.21 Few eyes have been examined histopathologically, and most of these have had end-stage disease that often obscures the earlier changes.22 Live microfilariae cause little intraocular reaction and may be found in the conjunctive, cornea, posterior sclera, anterior and posterior chambers, vitreous, uveal tract, inner retina, and optic nerve and its sheath.
Other Tissues
Microfilariae have been found in many of the deeper organs, including the liver, kidney, spleen, pancreas, lung, peripheral nerves, and arteries. They have also been found in tears, blood, urine, cerebrospinal fluid, sputum, vaginal secretions, and peritoneal fluid.
CLINICAL MANIFESTATIONS
Skin Changes
The actual bite of the blackfly is usually unnoticed, although a painful wheal develops quickly and resolves in 2 or 3 days. Pruritus is the most common early manifestation of onchocerciasis. It develops after a latent or “prepatent” interval of 1 to 2 years following initial infection. During this time, the infective larvae mature into adult worms. Symptoms develop as microfilariae appear in the skin and the prevalence of most types of onchocercal skin disease show statistically significant correlation with onchocercal endemicity. Itching and scratching may be mild and intermittent or severe and unremitting, in which case they often lead to excoriation and secondary infection, a syndrome complex widely known as “filarial scabies” (la gâle filarienne). A standard clinical method of classifying and grading the dermal changes of onchocerciasis has been developed23 and endorsed by World Health Organization (WHO) Expert Committee on Onchocerciasis Control.1
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree