Parasite
Definitive host
Intermediate host/vector
Accidental hosts
Infecting stage to human
Toxoplasmosis
Domestic cats and members of family Felidae
Birds and rodents
Human
Tissue cysts, sporulated oocysts
Onchocerciasis
Human
Blackfly in the genus Simulium
–
Third-stage larvae
Loa loa
Human
Deer flies of the genus Chrysops
–
Third-stage larvae
Dirofilariasis
Dogs and other carnivores
Mosquitoes
Human
Microfilariae
Gnathostomiasis
Freshwater fish, snakes, or birds, poultry, cats, dogs
Cyclops
Human
Third-stage larvae
Cysticercosis
Human when he harbors the adult worm
Cattle, beef
Human in cases of cysticercosis
Metacestode
Echinococcus granulosus
Dogs
Sheep and cattle
Human
Eggs in dog’s feces
Trematode granulomas
Birds
Snails, Melanoides tuberculata
Human
Metacercaria
Oestrus ovis
Sheep and goats
Sheep botfly
Human
Fly larvae
Echinococcosis
Dogs
Sheep, goats, swine, kangaroos
Human
Eggs
Orbital myiasis
Cattle, sheep, horses, deer
Mosquitoes
Human
Larval stage of a dipterous flies
Thelaziasis
Dogs, cats, rabbits, and wild carnivores
Phortica spp.
Human
Third-stage larvae
Giardiasis
Dogs, cats, calves, human
Food- and waterborne
–
Cysts
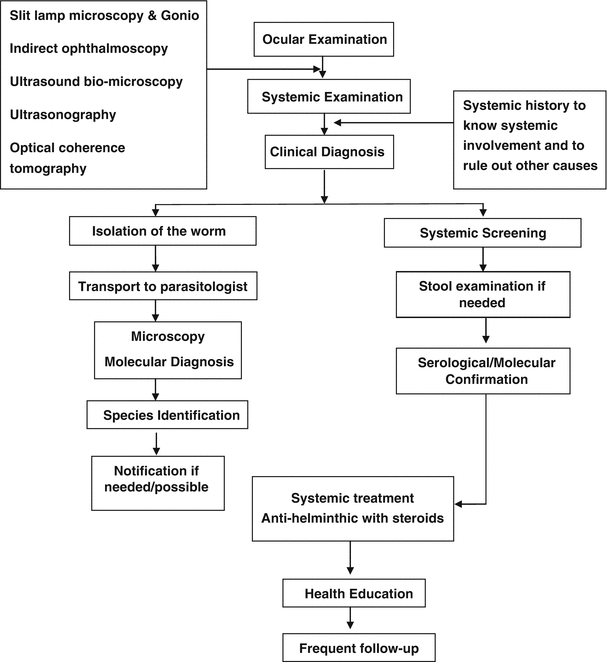
Fig. 13.1
Management algorithm in ocular parasitosis
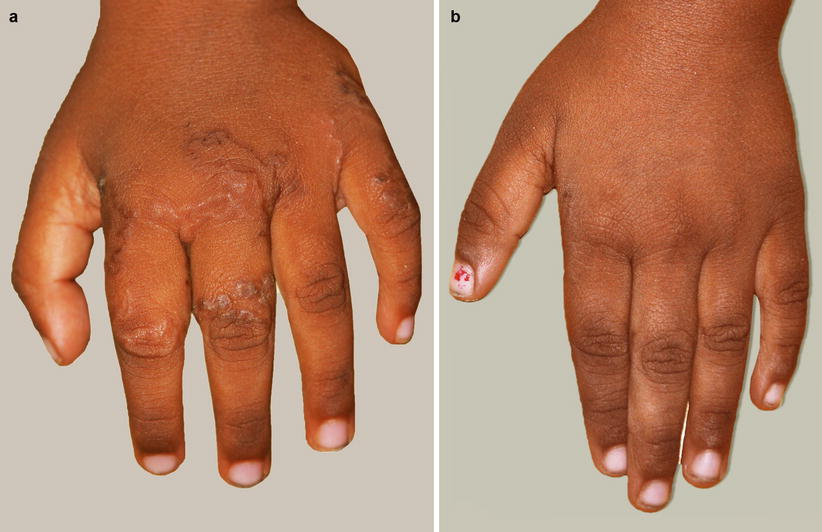
Fig. 13.2
(a) Visceral larva migrans in a young child before treatment. (b) Visceral larva migrans in a young child after treatment with oral albendazole
Table 13.2
Clinical signs and etiological agents in ocular parasitosis
Location of worm | Clinical signs | Etiological agents |
|---|---|---|
Orbit | Orbital cellulitis | Dirofilaria Trypanosoma cruzi Trichinella spiralis Echinococcus, coenurus, Taenia solium (Cysticercus), spargana, Gnathostoma |
Conjunctiva | Epiphora, conjunctivitis, itching | Ectoparasites Thelazia callipaeda, Trichinella spiralis |
Conjuctival granuloma | Rhinosporidiosis: R. seeberi | |
Subconjunctival motile worm | Dirofilaria, Loa loa, Baylisascaris procyonis Angiostrongylus cantonensis | |
Subconjunctival cyst | Cysticercosis | |
Subconjunctival granuloma | Trematode granuloma | |
Cornea | Keratitis with neural pain | Acanthamoeba spp. |
Punctate epithelial keratitis | Microsporidiosis | |
Anterior chamber | Alive motile worm | Onchocerca, Schistosoma, Taenia, spargana, Angiostrongylus, Gnathostoma, Toxocara, Dirofilaria Thelazia, Acanthocheilonema |
Hypopyon Hyphema Anterior chamber granuloma | Ruptured cysticercosis Gnathostomiasis Trematode granuloma | |
Iris | Iris atrophy Iris holes | Gnathostomiasis |
Vitreous | Alive worm Intact mobile cyst Panuveitis | Gnathostomiasis Cysticercosis Ruptured CC |
Subretinal space | Subretinal motile worm (DUSN) or Multifocal chorioretinitis with recurrent crops of retinitis | Ancylostoma caninum, Toxocara, Trichinosis, Baylisascaris procyonis, and filarial worms |
Cyst | Cysticercosis | |
Retina and choroid | Retinochoroiditis | Toxoplasma gondii, Onchocerca volvulus |
Retinochoroidal granuloma Salt pepper fundus Retinal hemorrhage, Retinal vasculitis | Toxocara Giardia lamblia Falciparum malaria, Loa loa W. bancrofti, B. malayi |
There are several challenges in the field of parasitic diseases. It is highly prevalent in developing countries; however, epidemiological investigations are scarce from such endemic regions. The challenges include poor public awareness of risk factors, lack of screening programs, lack of preventive measures, and availability of diagnostic facilities [1–8]. Sensitive molecular tests for parasite detection and differentiation are available at advanced basic research laboratories; still the direct benefit to the patient remains limited [9]. Finally, due to coevolutionary mechanisms, parasitic worms are developing resistance to available drugs, necessitating discovery of next-generation antiparasitic agents [1, 2, 4].
Ocular Ectoparasites
Parasites that live on the surface of the host organs are called ectoparasites and those living inside the organs are called endoparasites [9]. Ocular ectoparasites burrow into the eyelids or glue to the eyelash and remain there for relatively long periods of time. They bite transiently and cause chronic conjunctivitis, blepharitis, or cellulitis. It is important for the ophthalmologist to understand this ectoparasitic infestation which is commonly misdiagnosed as bacterial, viral, or allergic conjunctivitis or seborrheic dermatitis. Careful examination is a must for correct diagnosis and appropriate treatment. Ectoparasites of ophthalmic importance include Phthiriasis palpebrarum, demodicosis, Oestrus ovis, orbital myiasis, and thelaziasis. Other rare genera important to human myiasis include Dermatobia, Gasterophilus, Cordylobia, Chrysomya, Wohlfahrtia, Cochliomyia, and Hypoderma [2].
Phthiriasis palpebrarum
Phthiriasis palpebrarum is a flattened, wingless arthropod, belonging to the class Insecta. This crab louse infests the eyelashes of human beings. This is a rare disease generally associated with poor hygiene and overcrowding. The sucking mouth parts of adult parasite pierce the eyelid margin and suck the blood for relatively long periods of time. Saliva of feeding lice may cause pruritic lid margins and blepharoconjunctivitis leading onto chronic inflammation. Occasionally superficial punctate keratitis may also be seen. Slit-lamp examination can demonstrate swollen eyelids covered with reddish brown crusts of lice and nits. After applying topical anesthetic drops and eyelash clipping, the crusts can be removed [9].
Demodicosis
Demodex mites are ectoparasites often found in the follicles of facial skin and eyelids and on the eyelashes [10]. Demodicosis causes hypersensitivity, leading to granulomatous skin inflammation and chronic blepharitis. Diagnosis of demodicosis is made on slit-lamp microscopic detection of mites from the eyelash. Demodicosis contributes to chronic inflammatory conditions of the skin such as rosacea, folliculitis, and blepharitis and rarely may lead on to basal cell carcinoma. Oral metronidazole and ivermectin have been described to treat facial and ocular demodicosis successfully [11].
Oestrus ovis
Oestrus ovis is a cosmopolitan parasite of sheep and goats. It is the larval form of a hairy, yellowish, sheep botfly which is about the size of a common housefly [12]. The gravid adult female fly ejects a stream of milky fluid and larvae on to the nostrils of the sheep. The larva breeds in the nasal cavity and the sinuses of sheep. When they mature, they fall out from the nasal passage to pupate in the ground and mature as a fly. Men become accidental hosts, when the fly larvae are deposited in the human eyes (Fig. 13.3). Patients present with mucopurulent conjunctivitis and itching [13]. Often they are mistaken for bacterial conjunctivitis [14]. The diagnosis can be made by direct visualization of the larvae using the slit lamp. Manual removal after application of topical anesthesia is the recommended treatment.
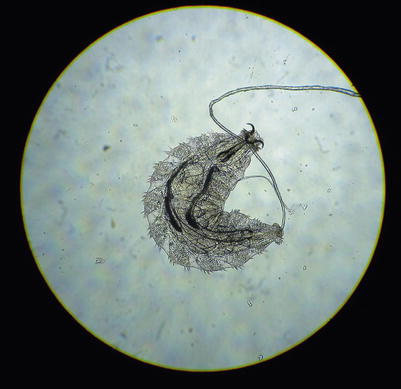

Fig. 13.3
The larval form of Oestrus ovis taken from conjunctival cul-de-sac of a patient
Orbital Myiasis
Myia is a Greek word meaning “fly.” Orbital myiasis is an infestation of the orbit by fly maggots. Fly eggs are usually transported by mosquitoes and hatch to emit larvae that can burrow into the intact skin [15]. Ophthalmomyiasis is classified into external, internal, or orbital, according to the site of the larval infestation. External ophthalmomyiasis refers to a limited infestation of the conjunctiva. Internal ophthalmomyiasis occurs when the larvae penetrate the conjunctiva and sclera and migrate into the subretinal space. Orbital myiasis occurs when large numbers of dipterous larvae invade and destroy the orbital contents. The resultant local inflammation with fistula formation can become superinfected with bacteria. The larvae can infest neglected wounds. Immobile patients with skin wounds in exposed areas are at high risk of developing myiasis. Medical and nursing staff should be alert to the possibility of myiasis in debilitated elderly patients, especially those with ulcerated skin lesions. Magnetic resonance imaging of the orbit is useful to delineate the extent of the infestation and identify residual maggots. In extensive orbital myiasis, exenteration is needed to prevent intracranial extension of the tissue destruction. Maggots can be removed manually. Concomitant bacterial infection needs to be treated with appropriate antibiotics.
Human Thelaziasis
The oriental eyeworm, Thelazia callipaeda, commonly named as eyeworm, infects the eyes and adnexa of mammals, including humans [16]. This parasite usually lives under the nictitating membrane of the eye, where the adult females release first-stage larvae into the lacrimal secretions. These larvae are subsequently ingested by the intermediate arthropod host within which they develop to the infective, third-stage larvae. The latter larvae are then deposited into the eyes of the definitive host. The disease, thelaziasis, is characterized by a range of subclinical to clinical signs, such as epiphora, conjunctivitis, keratitis, corneal opacity, and ulcers. Human thelaziasis is common in poor socioeconomic settings [17, 18].
Other parasites that may affect the orbital and periocular tissues include Coenurus cerebralis, a tapeworm; Dracunculus medinensis, Trichinella, Taenia solium, Ascaris, Onchocerca, Dirofilaria tenuis, group of nematodes; Schistosoma, a flatworm; and Acanthamoeba, Entamoeba, and Leishmania from protozoan group.
Ocular Endoparasites
Ocular endoparasites belong to either simple unicellular protozoans or complex multicellular metazoans. Protozoan parasites of ocular importance are Toxoplasma gondii, Acanthamoeba, Giardia lamblia, and rhinosporidiosis, while metazoa includes helminths such as nematodes (roundworms), cestodes (tapeworms), and trematodes (flatworms). Common nematode species of ocular importance are T. canis, T. catis, and O. volvulus. However, other rare nematodes cause diffuse unilateral subacute neuroretinitis. Cestode species that causes ocular cysticercosis is T. solium. Trematodes can cause granulomatous eye disease in children in some endemic areas.
Protozoa
Toxoplasmosis
Toxoplasmosis is caused by obligate intracellular protozoan parasite, Toxoplasma gondii. It causes ocular toxoplasmosis which is the leading cause of retinochoroiditis throughout the world. This entity is covered as a separate chapter (Chap. 1, Page 1).
Acanthamoeba keratitis
Acanthamoeba spp. is a free-living protozoon that has been isolated from soil, bottled water, eyewash solutions, and air. Acanthamoeba keratitis is common in contact lens users and after corneal trauma [2, 19]. It is characterized by severe pain due to neuritis. It presents as a unilateral central or paracentral corneal infiltrate, often with a ring-shaped peripheral infiltrate. Other characteristic symptoms include eyelid ptosis, conjunctival hyperemia, and lack of discharge. Acanthamoeba keratitis can progress to severe scleritis. A clinical diagnosis can be supported by confocal microscopy although a definitive diagnosis requires culture, histology, or molecular identification [19].
Giardiasis
Giardia lamblia is a binucleate protozoan and is considered to be one of the common causes of gastroenteritis in developing countries and crowded urban areas. Asymptomatic, nonprogressive retinal lesions were reported in younger children with giardiasis. These lesions characteristically showed salt and pepper pattern represented by punctate areas of hyperpigmentation and hypopigmented spots at the posterior pole along the major blood vessels [2, 20]. The mechanism underlying the ocular lesions is not known. Patients present with normal electroretinographic findings in spite of extensive retinal involvement. Functional changes have not been reported in these patients.
Rhinosporidiosis
Rhinosporidiosis is caused by Rhinosporidium seeberi. Recent investigations of RNA genes reveal that it is more closely related to fish parasites than to fungi [21], and it is, therefore, included in protozoan diseases of the eye. It causes a mucocutaneous disease that involves the palpebral conjunctiva and produces polypoid or papillary growths that arise from mucous epithelium [22]. Scleral necrosis can result in blindness (Fig. 13.4). Rhinosporidium seeberi has never been successfully cultured in vitro. At present, the treatment for rhinosporidiosis is surgical excision. Some authors propose medical therapy with dapsone [23], but the results are not convincing. Antimicrobial therapy is ineffective, and the disease may recur after months or years.
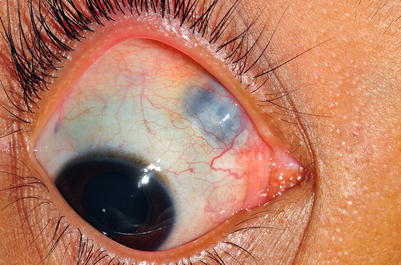

Fig. 13.4
Rhinosporidium causing sclera necrosis and uveal prolapse
Other rare protozoa that can cause ocular infection include Trypanosoma cruzi causing palpebral and periorbital edema. Leishmania and Microsporidia infection results in conjunctivitis, uveitis, and retinal hemorrhage. Microsporidia causes conjunctival hyperemia, punctate epithelial keratitis, hyphema, necrotizing keratitis, and corneal ulcer more often in immunocompromised individuals, while malarial infection causes retinal hemorrhage, papilledema, and cotton wool spots [2].
Metazoa
Nematodes
Toxocara canis/Toxocara cati: Toxocariasis
Toxocara canis and Toxocara cati are parasitic nematodes that reside in the small intestine of dogs, cats, and wild carnivores [24]. Human children are accidental hosts who become infected by ingesting infective eggs in contaminated soil. Following ingestion, the eggs hatch and larvae penetrate the intestinal wall and are carried by the circulation to the eyes, liver, heart, lungs, brain, or muscle. The larvae do not develop further in these sites, but the host inflammatory response against the migrating larvae can cause tissue damage [25]. They cause both visceral and ocular larva migrans in human. Heavy infection may result in fever, anorexia, malaise, irritability, hepatomegaly, respiratory symptoms, pruritic urticaria-like cutaneous lesions, and eosinophilia. Visceral larva migrans results in hepatitis and pneumonitis. Toxocariasis is a worldwide public health problem; however, infection tends to occur more frequently in tropical regions than in temperate regions [26].
Ocular Toxocariasis
Common symptoms are unilateral visual impairment and subsequent strabismus. Inflammation is due to larval localization and granulomatous response around the larva resulting in a whitish elevated posterior segment or peripheral granuloma. A fibrovascular band may be seen running towards the posterior pole or optic nerve. Early lesions may be associated with intense vitreous haze. Chronic lesions may have pigment epithelial hyperplasia [24]. Occasionally, it may also present as uveitis, papillitis, endophthalmitis, optic nerve granuloma, and vitreous lesions [25]. The most serious consequence is invasion of the retina leading to tractional retinal detachment and blindness. Clinically, the lesion may mimic retinoblastoma.
Pathogenesis
The larvae released from the eggs shed immunogenic glycoproteins known as Toxocara excretory–secretory antigens (TES). These antigens seem to allow larvae to evade various organs and survive under the adverse conditions of the host’s immune response [27]. Clinical symptoms vary as a consequence of larvae migration, ranging from asymptomatic form to those with severe organ involvement.
Diagnostic Strategies
Ultrasonography may show a high reflecting peripheral mass and vitreous band [28]. In patients with undiagnosed vitritis with suspicion of toxocariasis, cytology can confirm the clinical diagnosis [29]. Even though the disease is prevalent throughout the world, lack of good diagnostic kits makes the laboratory confirmation difficult. Intraocular fluid analysis for antibody may prove useful. Two recombinant proteins TES-30 and TES-120 are under evaluation [30, 31].
Treatment
Anterior segment inflammation is treated with corticosteroids and cycloplegics; panuveitis is treated with systemic (0.5–1 mg/kg body weight) and periocular corticosteroids. Treatment with oral albendazole in ocular disease is controversial [32]. Pars plana vitrectomy is done for endophthalmitis and for vitreoretinal tractions. An albendazole–chitosan microparticle prepared by ionotropic gelation has been studied in mouse model, and authors claim this agent to be more effective than conventional albendazole for visceral larva migrans [33].
Onchocerca volvulus: Onchocerciasis
Onchocerca volvulus is a filarial nematode transmitted by the Simulium blackfly resulting in onchocerciasis, a blinding disease. The blackfly and the disease are prevalent along the river in the endemic countries; hence, it is called “river blindness” [34]. It is endemic in Africa, Eastern Mediterranean, and Central and South America. Risk for travelers is generally low, unless travel involves extensive exposure to vectors in areas at risk. Humans are the main reservoir of onchocerciasis. The life cycle of Onchocerca volvulus begins when the female blackfly ingests microfilariae from infected human blood and the microfilaria develops into first-, second-, and third-stage larvae in the blackfly. When it bites another human, third-stage larvae get into the human and develop into adult worms. The adult worms settle in subcutaneous fibrous nodules, which are commonly known as onchocercomas. These firm painless nodules are predominantly seen on the head, face, and torso. Usually, the nodules are composed of 2–3 female worms and daughter microfilariae encapsulated in a fibrous coat. Microfilariae move across subcutaneous tissue and spread throughout the body, resulting in itching and dermal depigmentation. However, death of microfilariae causes an intense inflammatory reaction. The lymph nodes that drain the infected areas show granulomatous inflammation, fibrosis, and atrophy on histological examination.
Ocular Features
Ocular onchocerciasis is due to the presence, death, and/or migration of microfilariae in and through ocular structures as well as the host’s response to the migration. Clinical signs include hyperemia, conjunctival nodules, limbal edema, punctate keratitis, sclerosing keratitis, granulomatous or nongranulomatous uveitis, bilateral symmetric chronic chorioretinitis, geographic retinal atrophy, consecutive optic atrophy, and glaucoma. Blindness occurs due to corneal vascularization, glaucoma, complicated cataract, chronic chorioretinitis, and optic atrophy [34]. Slit-lamp examination may confirm the presence of microfilariae in the anterior chamber. A sclerocorneal punch biopsy may also be useful.
Pathogenesis
It is the migration of microfilariae through skin and connective tissue which is responsible for the majority of clinical findings in onchocerciasis. As in leprosy and leishmaniasis, onchocerciasis appears to be a spectrum of disease ranging from mild infection and highly symptomatic host reaction at one end to a severe infection but less severe host reaction at the other end [35]. Immunosuppression in onchocerciasis rises with parasite burden, and it involves regulatory T cells and transforming growth factor-beta (TGF-beta) [36]. Wolbachia is a Gram-negative bacterium, one of the world’s most common parasitic microbes. It plays an endosymbiotic role and infects many species of filarial nematodes including Onchocerca volvulus. Earlier studies using a murine model for river blindness demonstrated that the predominant corneal inflammatory response in onchocerciasis was due to these bacteria. Wolbachia has emerged as an important target for chemotherapy that results in long-term sterility of the worms in onchocerciasis [37].
Diagnosis
Diagnostic procedures include skin snips and demonstration of microfilaria. However, sensitivity of this test is low. When there is a palpable nodule, nodular excision biopsy can demonstrate the microfilaria. Mazzotti test requires the oral administration of diethylcarbamazine (DEC) and the test is considered positive if the patient develops pruritus and intense inflammation following the death of microfilariae [38]. However, this test is rarely used now because it can cause severe, even fatal allergic reaction. Mazzotti test has been replaced by the DEC patch skin test to detect allergic reaction to topical application of DEC. A gauze pad soaked in a 20 % solution of DEC is placed on the hip of the patient. The site of application is later examined for skin inflammation following localized microfilarial death. The patch test is less sensitive than nodulectomies [39].
Immunodiagnostic tests include enzyme-linked immunosorbent assay (ELISA) [40]. Rapid-format antibody card test is also available which requires a drop of blood from a finger prick. A positive test will result in a color change on the card surface. It is less expensive and it is as sensitive as ELISA. Card test does not require a lab setting and provides instantaneous results in the field [41]. Samples from skin snips or skin scratches can be used for polymerase chain reaction for detection of DNA [42].
Treatment
Ivermectin administered as a single dose of 150 mg/kg/day removes the microfilariae but does not eliminate the adult worm or cure the infection [43, 44]. A 6-week course of doxycycline targets Wolbachia endosymbiotic bacteria and also sterilizes the adult female worms. Doxycycline is also macrofilaricidal for O. volvulus, and it is the first well-tolerated macrofilaricidal agent in coinfection with Loa loa [45]. Moxidectin, a microfilaricidal agent, sterilizes or kills the adult worms. WHO has started a phase III comparative trial with ivermectin and moxidectin [46].
Loa loa: Loaiasis
Loaiasis is an infection by the filarial nematode Loa loa. The vectors are the flies from two species of the genus Chrysops, C. silacea, and C. dimidiate. Like onchocerciasis, the adult worms exist in the subcutaneous tissues in human beings and release sheathed microfilariae into the bloodstream in a diurnal rhythm. Microfilariae migrate rapidly in the body. The fly ingests microfilariae during a blood meal. After ingestion, the microfilariae develop into first-, second-, and third-stage larvae. The third-stage infective larvae can infect another human when the fly takes a blood meal [2].
Loa loa has a predilection for ocular tissues and is commonly known as the “eye worm.” Periocular subcutaneous macrofilariasis can present with intermittent attacks of painful lid swelling [47]. Often the worm can be seen in subconjunctival or subcutaneous plane as well. Blood films taken at noon and midnight are important in the detection and quantification of microfilaremia. Definitive diagnosis of filarial species can be made only after examination of the worm under the microscope. Improved molecular diagnostics for L. loa infection are needed not only in endemic areas but also in clinical laboratories of resource-rich countries to confirm infection in immigrants and travelers [48].
After topical anesthesia, a subconjunctival Loa loa worm can be removed with forceps through a conjunctival incision. Persons who undergo surgical removal of a worm may still have larvae in their blood, and they should be treated with diethylcarbamazine (DEC), 6 mg per kg body weight per day in three divided doses, administered orally for 3 weeks. This therapy kills both the larvae and the adult worms. DEC can cross the blood–brain barrier, and sudden death of many microfilariae can cause capillary blockage by dead and dying microfilariae in the brain, meninges, and retina, causing encephalopathy and death. In patients with more than 100 microfilariae per 50 cubic mm of afternoon blood, pretreatment with steroids should be considered before initiation of DEC therapy. Loa loa infection in endemic area complicates the mass treatment of onchocerciasis with ivermectin as its use in Loa loa patients might cause encephalitis. Albendazole therapy appears to be effective in killing adult worms and reduces the larval load. It is said to be useful in some loaiasis patients who have remained uncured after multiple courses of DEC treatment.
It is very important to understand that the patient’s history need not include recent travel to an endemic country. After inoculation by an infected fly, adult worms take approximately 3–4 years to mature in the human host. Consequently, Loa loa may present many years after the patient’s visit to an endemic region [49, 50].
Larva Migrans Ophthalmia Syndrome
Some of the helminthic organisms of other vertebrate animals may be transmitted accidently to humans. In such a scenario, the life cycle of these parasites does not get completed in man, and there is no further reproduction of the worms. Human beings play a role of accidental host; hence extracting the single parasite is the definitive treatment. Some of the examples for accidental human host include dirofilariasis, diffuse unilateral subacute neuroretinitis (DUSN), and gnathostomiasis.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


