Fig. 8.1
(Patient # 1). Both hands and feet of a patient with chronic VKH showing vitiligo

Fig. 8.2
(Patient # 1). External eye photograph right eye showing poliosis. Left eye too had similar findings (not shown)
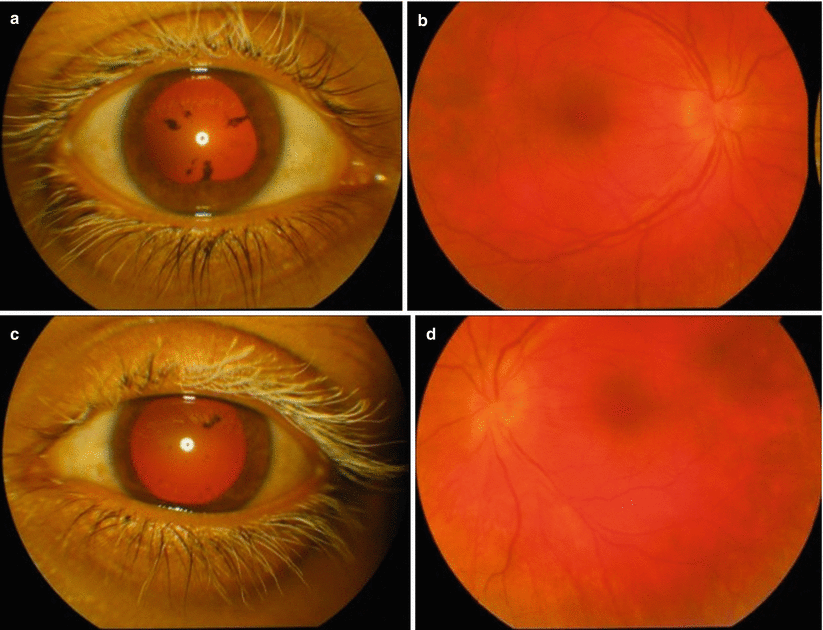
Fig. 8.3
(Patient # 1). (a) External photograph right eye showing poliosis, posterior synechiae, and red glow. (b) Fundus photograph right eye showing sunset glow fundus in chronic VKH. (c) External photograph left eye showing poliosis, pigment on anterior lens surface, and red glow. (d) Fundus photograph left eye showing sunset glow fundus in chronic VKH
Laser Flare Photometry (LFP)
In 1988 Mitsuru Sawa and colleagues developed a new technology to quantify flare and cells in the anterior chamber. This instrument called laser flare/cell photometer comprises of a constant power helium-neon or diode laser light beam, which is directed into the anterior chamber at an angle of 45° to the anteroposterior axis [1]. The backscattered light from the incoming beam is then detected by a photomultiplier-photodetector unit [2]. The commercially available laser flare meters can make two types of measurements: (1) the first type of laser flare photometers measures the level of backscattered light reflected from small molecules such as proteins in anterior chamber in photons per milliseconds (ph/ms) [2, 3] and (2) the second type of laser flare-cell photometer measures the backscattered light from small molecules (proteins) as well as from larger particles such as inflammatory cells in units for particles/cells are number of particles/cells counted in a volume of 0.075 mm [4, 5].
LFP is superior to slit lamp for documenting anterior chamber flare and cells and only method available that quantifies anterior segment inflammation [6–9]. Its advantages over slit lamp are as follows:
Laser flare photometer is useful in diagnosing subclinical inflammation.
More accurate and objective in monitoring response to an intervention.
More sensitive tool to detect the early disease relapses. A flare increase of more than 20 % over the flare level at the previous visit during the regular follow-up period has a positive predictive value for disease relapse [10].
It helps in titrating the dosage of therapy, documenting a therapeutic response in cases when there is a significant decrease of flare before any other clinical parameter change can be detected [11].
Ultrasound Biomicroscopy (UBM)
UBM is noncontact, noninvasive imaging modality that provides high-resolution, cross-sectional biomicroscopic images of the anterior structures of the eye. It uses ultrasound of high frequency (35–100 MHz) than conventional ultrasound and is based on the hypothesis that by increasing the frequency, greater microscopic resolution can be obtained [12, 13]. This results in visualization of the limited tissue depths within its penetration of approximately 4–5 mm allowing imaging of anterior segment structures and ciliary body. Rotating the eye as far as possible can scan the conjunctiva, underlying sclera, as well as peripheral retina [14]. UBM provides the following information:
UBM has been reported to provide essential information that allows to reach a specific diagnosis or to influence the management in patients with uveitis. Tran et al. [15] performed UBM in 77 patients with uveitis where it was thought necessary and they could obtain useful information in 32 of 77 patients (42 %).
UBM is especially found to be useful in cases with opaque media to study ciliary body
UBM is particularly helpful in studying the structure of ciliary body in patients with hypotony and toxocara uveitis where it shows pseudocystic vitreous degeneration [16].
UBM plays a very important role in assessing ciliary body preoperatively in patients with complicated cataract and hypotony. UBM has been reported to show inflammatory cells in the anterior and posterior chamber, exudates, as well as swelling of ciliary body in patients with acute anterior uveitis and these signs resolved on treatment [17].
UBM is also helpful in planning the surgical intervention. In eyes with normal ciliary body processes and cyclitic membrane, seen preoperatively by UBM, pars plana lensectomy with removal of ciliary membrane alone was sufficient to restore intraocular pressure (IOP) in the postoperative period. However, if preoperative ciliary body atrophy was present, additional silicone oil tamponade was required to restore IOP [18].
Fundus Photography
Fundus color photography helps in documenting the retino-choroid pathologies and serves as tool for monitoring these diseases with good inter-observer agreement [19]. It has been reported to be useful for documenting macular edema, epiretinal membranes, retinitis, retinal vasculitis, choroiditis, and parasitic infections like toxocariasis, cysticercosis, onchocerciasis, and masquerade syndromes and for assessing media clarity [20]. Stereo photographs may be helpful in cases with exudative retinal detachment, optic disk edema, macular and choroidal neovascularization, etc. (Fig. 8.4).
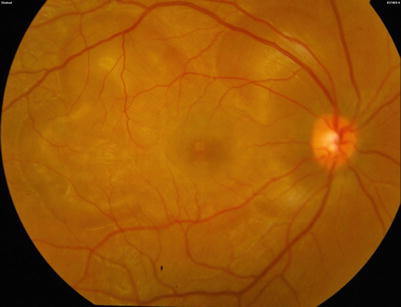

Fig. 8.4
(Patient # 2). Fundus photograph right eye showing multiple areas of serous retinal detachment
Fundus Fluorescein Angiography (FFA)
Fundus fluorescein angiography has established its role in the clinical practice of uveitis diagnosis as well as management and is quite useful in following situations:
Retinitis/Retinochoroiditis
FFA is especially useful for studying the inflammatory lesions of the retina and retinal vessels [21, 22]. Illjima et al. [22] reported FFA findings of acute ocular toxoplasmic retinochoroiditis that include a hyperfluorescent lesion with central hypofluorescence, the arterial occlusion passing through the necrotic lesion showing a dark silhouette, and venous dilation and leakage and optic disk staining with dye leak.
Retinal Vasculitis
Retinal vasculitis is seen in Behçet’s disease, sarcoidosis, toxoplasmosis, tuberculosis, idiopathic retinal vasculitis, aneurysms and neuroretinitis (IRVAN), syphilis, systemic lupus erythematosus, birdshot chorioretinopathy, acute retinal necrosis, frosted branch angiitis, and Eales’disease [23].
Cystoid Macular Edema (CME)
Fluorescein angiogram in CME shows telangiectasia of the parafoveal capillaries, with progressive leakage and accumulation of dye in the cystic spaces producing “petaloid” or “flower petal” pattern of parafoveal hyperfluorescence [21].
Based on fluorescein angiography features, CME has been graded as follows by Miyake [24].
Grade 0: no sign of fluorescein leakage.
Grade I: slight fluorescein leakage into cystic spaces but not enough to enclose the entire fovea centralis.
Grade II: complete circular accumulation of the fluorescein in the cystic space but its diameter is smaller than 2 mm.
Grade III: the circular accumulation of fluorescein is larger than 2.0 mm in diameter.
In 1984, Yannuzzi [25] proposed the following classification:
Grade 0: no perifoveal hyperfluorescence
Grade 1: incomplete perifoveal hyperfluorescence
Grade 2: mild 360° hyperfluorescence
Grade 3: moderate hyperfluorescent area being approximately 1 disk diameter across
Grade 4: severe 360° hyperfluorescence with the hyperfluorescent area being approximately 1.5 disk diameter across
Retinal Vascular Occlusion and Neovascularization
Retinal Macroaneurysms
FFA is quite useful in detecting macroaneurysms in sarcoidosis and IRVAN.
Choroid
FFA may show early choroidal hypofluorescence manifesting as choriocapillaris perfusion delay or non-perfusion in Vogt-Koyanagi-Harada disease and inflammatory choriocapillaropathies, like acute posterior multifocal placoid pigment epitheliopathy (APMPPE), serpiginous choroiditis, and multiple evanescent white dot syndrome (MEWDS). In Vogt-Koyanagi-Harada disease and sympathetic ophthalmia, FFA shows initial pinpoint hyperfluorescent dots or areas of delayed choroidal filling with late pooling of dye in subretinal space that maybe associated with optic disk hyperfluorescence (Figs. 8.5 and 8.6) [30–33] In serpiginous choroiditis, the active borders show early hypofluorescence with progressive diffuse staining in late frames. Additionally, FFA is also useful in diagnosing choroidal neovascular membrane that complicates many of the posterior uveitides.
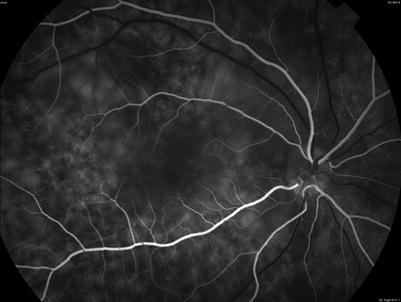
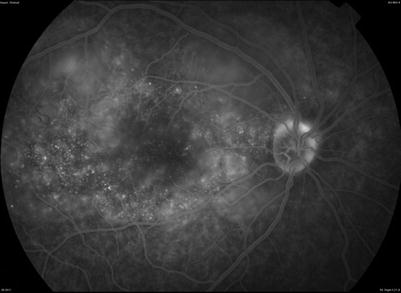

Fig. 8.5
(Patient # 2). Fundus fluorescein angiogram right eye in the early phase shows patches of delayed choroidal filling as well as some punctate hyperfluorescent dots temporal to fovea

Fig. 8.6
(Patient # 2). Fundus fluorescein angiogram right eye in the late phase shows multiple punctate hyperfluorescent dots with areas of dye pooling corresponding to serous retinal detachment and optic disk hyperfluorescence
Indocyanine Green Angiography (ICG)
Indocyanine green angiography is the best imaging modality for imaging choroid including the choriocapillaris and the choroidal stroma [33].
Hypofluorescence on ICG is seen in the following:
(i)
Choriocapillaris non-perfusion is seen as patchy/geographic disposition in the early and intermediate phase and persists in the late frames.
(ii)
Stromal inflammatory infiltration seen as more regular dots with an even distribution in the early phase. If these stromal lesions are full thickness, they shall remain hypofluorescent up to late frames, whereas partial thickness lesions become isofluorescent in late frames. Many a times, these lesions may show fuzzy appearance of large choroidal vessels in the intermediate phase and diffuse choroidal fluorescence in late phase [34, 35].
Hyperfluorescence on ICG is seen in the following:
(i)
Diffuse hyperfluorescence may result from increased leakage from large inflamed stromal choroidal vessels.
(ii)
Hyperfluorescence at the level of disk indicates severe inflammation.
(iii)
Late hyperfluorescent pinpoint lesions are indicative of granulomatous disease (Fig. 8.7).
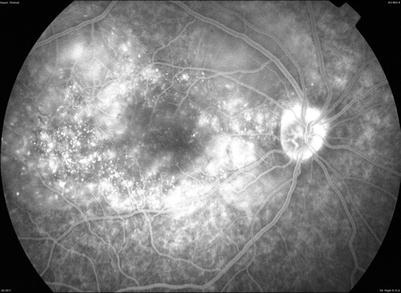

Fig. 8.7
(Patient # 2). Indocyanine green angiography shows late hyperfluorescent pinpoint lesions indicative of granulomatous disease along with disk hyperfluorescence
Based on ICG, choroidal inflammation can be reclassified as inflammation of the choriocapillaris (choriocapillaritis) or inflammation of the choroidal stroma (stromal choroiditis) [35, 36]:
1.
Primary inflammatory choriocapillaropathies
Multiple evanescent white dot syndrome/acute idiopathic blind spot enlargement
Acute posterior multifocal placoid pigment epitheliopathy
Multifocal choroiditis/punctate inner choroidopathy
Serpiginous choroiditis
Rare entities: acute macular neuroretinopathy and acute zonal occult outer retinopathy
2.
Stromal choroiditis
Primary obligatory stromal choroiditis
Vogt-Koyanagi-Harada disease
Sympathetic ophthalmia
Birdshot chorioretinopathy
Stromal choroiditis as a random location of a systemic disease
Sarcoidosis
Tuberculosis
Syphilis
Other infectious choroiditides
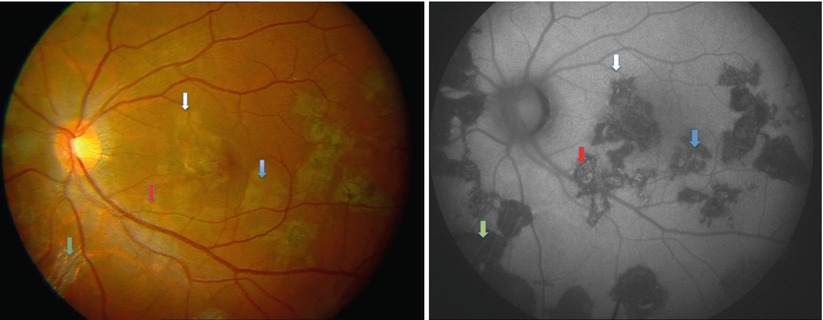
Fundus Autofluorescence (FAF)
Fundus autofluorescence using confocal scanning laser ophthalmoscope (cSLO) is gaining popularity as an imaging technique. FAF detects low-intensity autofluorescence produced by fluorophores such as lipofuscin present in the retinal pigment epithelial cells [37, 38]. The lipofuscin originates from the photoreceptor outer segments and its accumulation in the RPE cell lysosomes and reflects the quality of the RPE cell metabolism that may be indicative of disease activity. FAF imaging has been used to monitor disease activity in serpiginous-like choroiditis with four stages of disease activity [39] (Figs. 8.8 and 8.9):