Chapter 59 Ocular Ischemic Syndrome
In 1963, Kearns and Hollenhorst1 reported on the ocular symptoms and signs occurring secondary to severe carotid artery obstructive disease. They called the entity “venous stasis retinopathy” and noted that it occurred in approximately 5% of patients with severe carotid artery insufficiency or thrombosis. Some confusion has since arisen with this term because it has also been used to designate mild central retinal venous obstruction.2 A number of additional alternative names have been proposed, including ischemic ocular inflammation,3 ischemic oculopathy,4 and the ocular ischemic syndrome.5,6 Histopathologic examination of eyes with the entity generally does not reveal inflammation,7,8 and therefore the descriptive term the present authors and Dr. Larry Magargal have thought preferable is the ocular ischemic syndrome.5,6
Demographics and incidence
The mean age of patients with the ocular ischemic syndrome is about 65 years, with a range generally from the fifties to the eighties. No racial predilection has been identified, and males are affected more than females by a ratio of about 2 : 1. Either eye can be affected, and in approximately 20% of patients, ocular involvement is bilateral. The incidence of the disease has not been extensively studied, but from the work of Sturrock and Mueller9 an annual estimate of 7.5 cases/million persons can be made. This number may be falsely low, since it is possible that a number of cases are misdiagnosed.
![]() For additional online content visit http://www.expertconsult.com
For additional online content visit http://www.expertconsult.com
Etiology
In general, a 90% or greater stenosis of the ipsilateral carotid arterial system is present in eyes with the ocular ischemic syndrome.5 It has been shown that a 90% carotid stenosis reduces the ipsilateral central retinal artery perfusion pressure by about 50%.10,11 The obstruction can occur within the common carotid or internal carotid artery. In about 50% of cases, the affected vessel is 100% occluded, while in 10% there is bilateral 100% carotid artery obstruction.5
Occasionally, obstruction of the ipsilateral ophthalmic artery can also be responsible.5,12,13 Rarely, an isolated obstruction of the central retinal artery alone can mimic the dilated retinal veins and retinal hemorrhages seen in eyes with the ocular ischemic syndrome.14
Atherosclerosis within the carotid artery is the cause for the great majority of cases of the ocular ischemic syndrome.5 Dissecting aneurysm of the carotid artery has been reported as a cause,15 as has giant cell arteritis.16 Hypothetically, entities such as fibromuscular dysplasia,17 Behçet disease,18 trauma,19 and inflammatory entities that cause carotid artery obstruction could lead to the ocular ischemic syndrome.
Clinical presentation
Visual loss
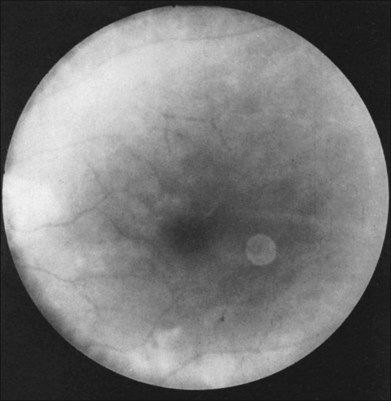
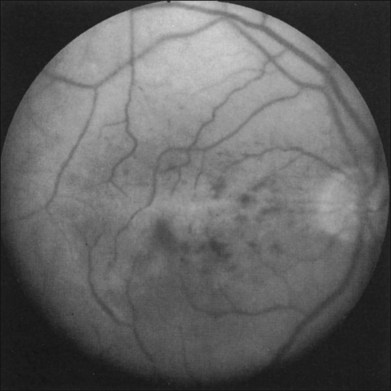
Greater than 90% of patients with the ocular ischemic syndrome relate a history of visual loss in the affected eye(s).5 In two-thirds of cases, it occurs over a period of weeks, but it is abrupt in approximately 12%. In this latter group, with sudden visual loss, there is often a cherry-red spot present on funduscopic examination (Fig. 59.1).
Prolonged light recovery
Prolonged recovery following exposure to a bright light has been described in patients with severe carotid artery obstruction.20 Concurrent attenuation of the visual evoked response has also been observed in these cases after light exposure. The phenomenon has been attributed to ischemia of the macular retina. In cases of bilateral, severe carotid artery obstruction, the visual loss after exposure to bright light occurs in both eyes, mimicking occipital lobe ischemia due to vertebrobasilar disease.21
Scintillating scotomas
Dissection of the internal carotid artery has been reported to cause scintillating scotomas that resemble a migraine aura.22 While these could theoretically be associated with the classic ocular ischemic syndrome, they have not been observed by the authors.
Amaurosis fugax
A history of amaurosis fugax is elicited in about 10% of ocular ischemic syndrome patients.5 Amaurosis fugax, or fleeting loss of vision for seconds to minutes, is thought to most commonly be caused by emboli to the central retinal arterial system, although vasospasm may also play a role.23 Although the majority of people with amaurosis fugax alone do not have the ocular ischemic syndrome, it can be an indicator of concomitant, ipsilateral carotid artery obstructive disease. About one-third of patients with amaurosis fugax have an ipsilateral carotid artery obstruction of 75% or greater.24 Rarely, it has been associated with a stenosis of the ophthalmic artery.24
Pain
Pain is present in the affected eye or orbital region in about 40% of cases,5 and has been referred to as “ocular angina.” Most often, it is described as a dull ache. It can occur secondary to neovascular glaucoma, but in those cases in which the intraocular pressure is normal, the cause may be ischemia to the globe and/or ipsilateral dura.
Visual acuity
The presenting visual acuities of patients with the ocular ischemic syndrome are bimodally distributed, with 43% of affected eyes having vision ranging from 20/20 to 20/50, and 37% having counting fingers or worse vision.25 Absence of light perception is generally not seen early, but can develop in the later stages of the disease, usually secondary to neovascular glaucoma. Among all eyes with the ocular ischemic syndrome after one year of follow-up, including those with and without treatment, approximately 24% remain in the 20/20–20/50 group and 58% have counting fingers vision or worse.
External collaterals
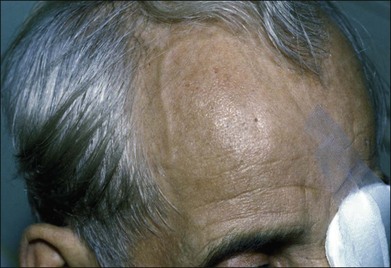
Prominent collateral vessels are occasionally seen on the forehead (Fig. 59.2). These vessels connect the external carotid system on one side of the head to that on the other. These vascular collaterals should not be mistaken for the enlarged tender vessels seen with giant cell arteritis, since temporal artery biopsy may shut down this important source of collateral blood flow to the brain.
Anterior segment changes
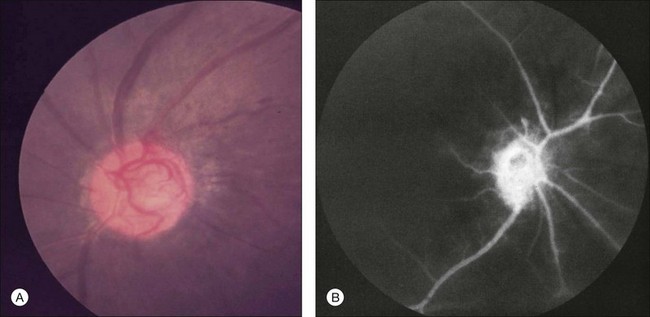
Neovascularization of the iris is encountered in approximately two-thirds of eyes with the ocular ischemic syndrome at the time of presentation5 (Fig. 59.3). Nevertheless, only slightly over half of these eyes have or develop an increase in intraocular pressure, even if the anterior chamber angle is closed by fibrovascular tissue. Impaired ciliary body perfusion, with a subsequent decrease in aqueous production, probably accounts for this phenomenon.
Flare in the anterior chamber is usually present in eyes with rubeosis iridis. An anterior chamber cellular response is seen in almost one-fifth of eyes with the ocular ischemic syndrome,5 but it rarely exceeds grade 2+, on a 0 to 4+ range, as per the Schlaegel classification.26 Keratic precipitates can be present, but are typically small.
Posterior segment findings
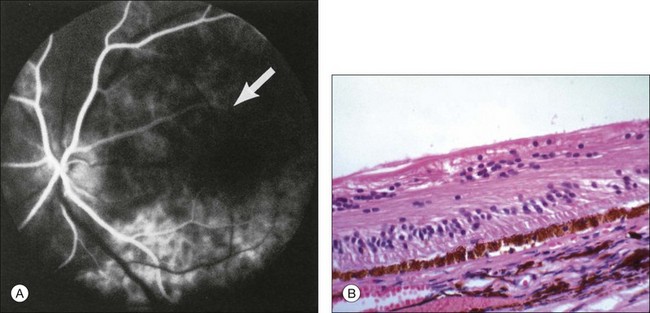
The retinal arteries are usually narrowed and the retinal veins are most often dilated, but not tortuous (Fig. 59.4). The venous dilation may be accompanied by beading, but usually not to the extent seen in eyes with marked preproliferative or proliferative diabetic retinopathy. Dilation of the veins is probably a nonspecific response to the ischemia from the inflow obstruction. Nevertheless, in some eyes both the retinal arteries and veins are narrowed. In contrast, eyes with central retinal vein obstruction usually also have dilated retinal veins, but they are often tortuous. The fact that the ocular ischemic syndrome occurs secondary to impaired inflow, while central retinal vein obstruction is usually associated with compromised outflow resulting from thrombus formation at or near the lamina cribrosa, may account for this difference.27
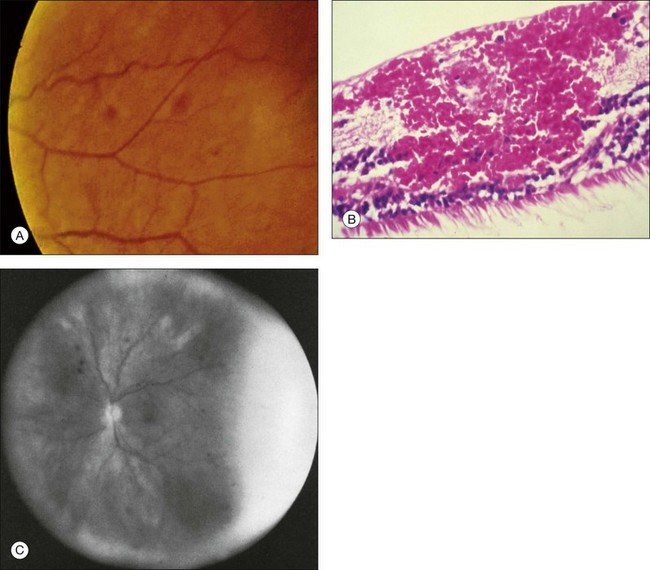
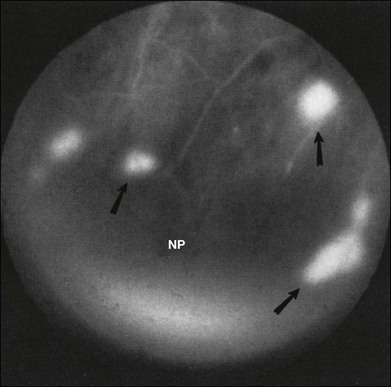
Retinal hemorrhages are seen in about 80% of affected eyes. They are most commonly present in the midperiphery, but can also extend into the posterior pole (Fig. 59.5, Fig. 59.6 online). While dot and blot hemorrhages are the most common variant, superficial retinal hemorrhages in the nerve fiber layer are occasionally seen. The hemorrhages probably arise secondary to leakage from the smaller retinal vessels, which have sustained endothelial damage as a result of the ischemia. Similar to the case with diabetic retinopathy, they may also result from the rupture of microaneurysms. In general, the hemorrhages seen with the ocular ischemic syndrome are less numerous than those accompanying central retinal vein obstruction. They are almost never confluent.
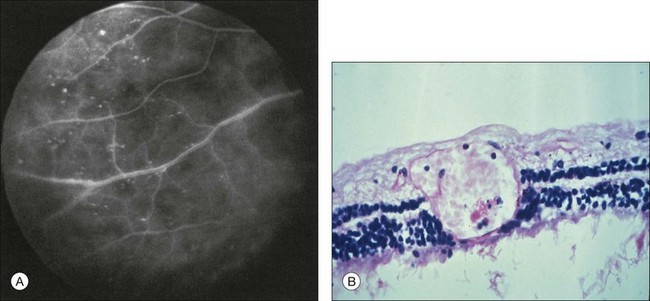
Microaneurysms are frequently observed outside the posterior pole, but can be seen in the macular region also. Hyperfluorescence with fluorescein angiography (Fig. 59.7) differentiates these abnormalities from hypofluorescent retinal hemorrhages. Retinal telangiectasia has also been described.28
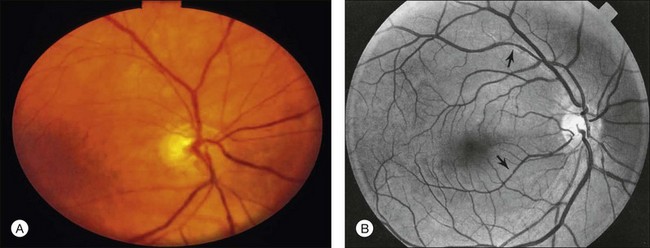
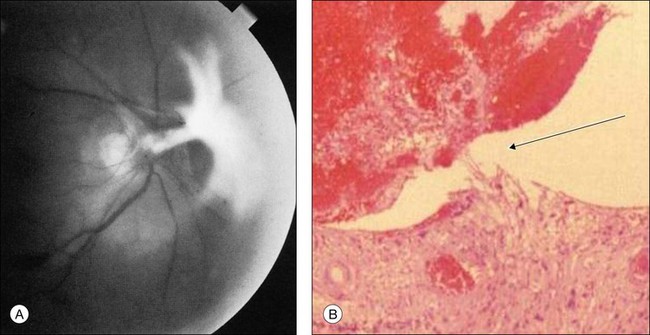
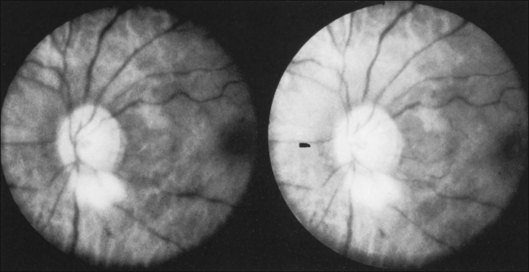
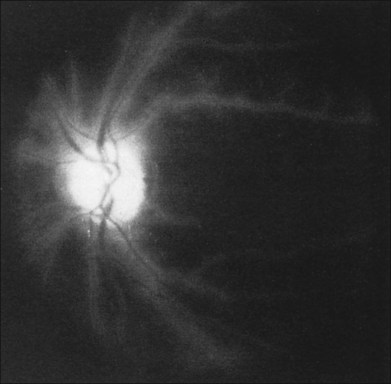
Posterior segment neovascularization can occur at the optic disc or on the retina. Neovascularization of the disc (Fig. 59.8) is encountered in about 35% of eyes, while neovascularization of the retina is seen in about 8%.5 Vitreous hemorrhage arising from traction upon the neovascularization by the vitreous gel has been reported to occur in 4% of eyes with the ocular ischemic syndrome in a retrospective study.5 Rarely, the neovascularization can progress to severe preretinal fibrovascular proliferation (Fig. 59.9 online). Neovascularization of the retina (Fig. 59.10 online) is encountered in 8% of eyes with ocular ischemia. It is usually present concomitant with neovascularization of the disc.
A cherry-red spot is seen in approximately 12% of eyes with the ocular ischemic syndrome (see Fig. 59.1).5 It can occur secondary to inner layer retinal ischemia from embolic obstruction of the central retinal artery, but probably more often develops when the intraocular pressure exceeds the perfusion pressure within the central retinal artery, particularly in eyes with neovascular glaucoma.
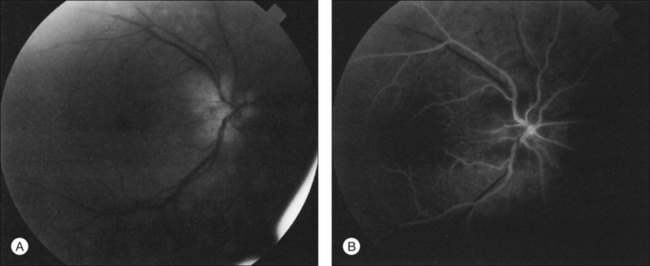
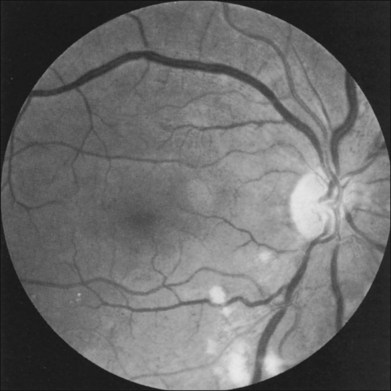
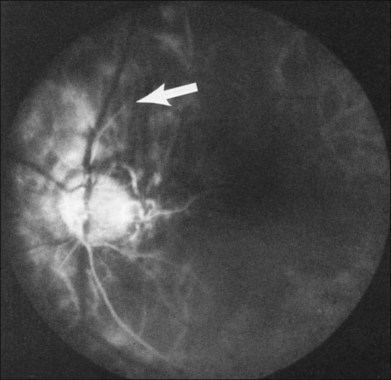
Additional posterior segment signs5 include cotton-wool spots (Fig. 59.11 online) in 6% of eyes, spontaneous retinal arterial pulsations in 4% (Fig. 59.12), and cholesterol emboli within the retinal arteries in 2%. In contrast to spontaneous retinal venous pulsations, which are a normal variant and located at the base of the large veins on the optic disc, the arterial pulsations are usually more pronounced, and may extend a disc diameter or more out from the optic disc into the surrounding retina. Anterior ischemic optic neuropathy (Fig. 59.13) has also been reported in ocular ischemic syndrome eyes.5,29,30 Acquired arteriovenous communications of the retina are rarely seen.31
A list of the anterior and posterior segment signs found with the ocular ischemic syndrome is shown in Table 59.1.1–5,9
Table 59.1 Anterior and posterior segment signs seen in eyes with the ocular ischemic syndrome
| Anterior segment | |
| Rubeosis iridis | 67% |
| Neovascular glaucoma | 35% |
| Uveitis (cells and flare) | 18% |
| Posterior segment | |
| Narrowed retinal arteries | Most |
| Dilated retinal veins | Most |
| Retinal hemorrhages | 80% |
| Neovascularization | 37% |
| Optic disc | 35% |
| Retina | 8% |
| Cherry red spot | 12% |
| Cotton-wool spot(s) | 6% |
| Spontaneous retinal arterial pulsations | 4% |
| Vitreous hemorrhage | 4% |
| Cholesterol emboli | 2% |
| Ischemic optic neuropathy | 2% |
(Adapted from Brown GC, Magargal LE. The ocular ischemic syndrome. Clinical, fluorescein angiographic and carotid angiographic features. Int Ophthalmol 1988;11:239–51.)
Ancillary studies
Fluorescein angiography
The intravenous fluorescein angiographic signs5 associated with the ocular ischemic syndrome are listed in Table 59.2.
Table 59.2 Fluorescein angiographic signs seen in eyes with the ocular ischemic syndrome
| Delayed and/or patchy choroidal filling | 60% |
| Prolonged retinal arteriovenous transit time | 95% |
| Retinal vascular staining | 85% |
| Macular edema | 17% |
| Other signs | |
| Retinal capillary nonperfusion | |
| Optic nerve head hyperfluorescence | |
| Microaneurysmal hyperfluorescence |
Delayed arm-to-choroid and arm-to-retina circulation times are frequently observed in the ocular ischemic syndrome. However, these measurements may be difficult to assess, since they depend upon whether the dye was injected in the antecubital fossa or hand, and also on the rate of injection. The observation of a well-demarcated, leading edge of fluorescein dye within a retinal artery after an intravenous injection is a distinctly unusual finding. It can be seen in eyes with the ocular ischemic syndrome, secondary to hypoperfusion (Fig. 59.14).
Normally, the choroidal filling is completed within 5 seconds after the first appearance of dye. Sixty percent of eyes with the ocular ischemic syndrome demonstrate patchy and/or delayed choroidal filling (Fig. 59.15). In some instances, the filling is delayed for a minute or longer. Although not the most sensitive sign, an abnormality in choroidal filling is the most specific fluorescein angiographic sign in ocular ischemic eyes.
Staining of the retinal vessels in the later phases of the study is seen in about 85% of eyes (Fig. 59.16 online, Fig. 59.17). Both larger and smaller vessels can be involved, the arteries generally more so than the veins. Chronic hypoxic damage to endothelial cells may account for the staining. In contrast, staining of the retinal vessels is uncommon, with central retinal artery obstruction alone. With central retinal vein obstruction, the veins can demonstrate late staining, but the retinal arteries are generally not affected.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree