(1)
Department of Ophthalmology and Visual Sciences, University of Iowa Hospitals and Clinics, Iowa City, IA, USA
Ocular ischemic syndrome (OIS) is relatively uncommon, but is a serious blinding condition. In 1874, Schmidt [1] reported neovascular glaucoma (manifestations of OIS) in a 58-year-old man who had suffered two cerebrovascular accidents – one 2.5 months before and the second 10 months after the development of glaucoma. The same year Loring [2] reported the case of a 62-year-old woman who developed central retinal artery occlusion and neovascular glaucoma (manifestations of OIS) and died of cerebrovascular accident. Thus, apart from being a serious blinding condition, it has a significant associated related risk of cerebrovascular and cardiovascular disease – and hence it has the possibility of being a potentially fatal condition. Iris neovascularization and neovascular glaucoma may be the first manifestation of OIS, and ophthalmologists may be the first to see these patients. That makes it an important ophthalmic disease, in which early detection and management may help to reduce associated complications of blindness and also reduce the risk of devastating systemic complications. Unfortunately, it is often missed or misdiagnosed, primarily because of its diverse and sometimes subtle presentation, and it can masquerade as several other ocular conditions. Unfortunately, most ophthalmologists are not aware of this fact. Moreover, the term ocular ischemia in some cases is applied loosely to any type of ocular ischemic lesion, e.g., anterior ischemic optic neuropathy, central retinal artery occlusion, and others, and that has sometimes created confusion in the literature. OIS is usually unilateral but sometimes may be bilateral.
Causes of Ocular Ischemic Syndrome
Since the early reports detailed above, a large number of cases of OIS with carotid artery disease have been reported in the literature. Except for a few cases, the cause is carotid artery occlusion or severe stenosis, as described by a voluminous literature. Atherosclerosis is the most common cause, as discussed in Chap. 13, 15, and 18. It may also be due to spontaneous internal carotid artery dissection [3–5], after penetrating trauma to the carotid artery [6], after occlusion of both external carotid arteries [7] or common carotid artery occlusion [8].
In addition, OIS is occasionally caused by other conditions, and these include giant cell arteritis [9–13], Takayasu’s arteritis [14–24], systemic lupus erythematosus [25, 26], polyarteritis nodosa [27], scleroderma [28], after radiotherapy of nasopharyngeal carcinoma [29], and radiation-induced ocular ischemia [30]; it can occur after cerebral aneurysm repair surgery [31], after vascular embolization of injectable calcium hydroxylapatite filler for nose augmentation [32], following autologous refined fat injection into the glabellar area to treat wrinkles [33], hypoplasia of the internal carotid artery associated with neurofibromatosis type 1 [34], arterial “steal” from the ophthalmic artery in patients with maxillofacial arteriovenous malformation [35], associated with Eisenmenger’s syndrome [36], mucormycosis [37], moyamoya [38], chronic herpes simplex keratouveitis [39], strabismus fixus convergens [40], and hyperhomocysteinemia [41]. I have also seen it develop following prolonged non-ophthalmic surgeries, e.g., spinal region surgery, orbital trauma, and, in one case, following shoulder surgery in a sitting position with pressure on the eyes.
Pathophysiology of Ocular Ischemic Syndrome
OIS is caused by a reduction of blood flow to the eyeball, which can produce anterior or posterior segment ischemia or both. It can also be associated with orbital ischemic damage. It is well established that occlusion or severe stenosis of the internal carotid artery (and, in some cases, ophthalmic artery occlusion or severe stenosis) is the major, if not the primary, factor in the development of OIS in the vast majority of patients. Not all patients with severe carotid artery disease develop OIS, suggesting that other risk factors must play a role or, presumably, the carotid artery has developed good collateral circulation. My study (see below) showed that the incidence of diabetes mellitus in patients with OIS is much higher than in the comparable general population. This suggests that small blood vessel disease in the eye (produced by diabetes mellitus), in the presence of large vessel disease, most probably increases the risk for OIS; unfortunately, we do not have an acceptable control population to substantiate this theory fully.
The sequence of ophthalmic abnormalities in OIS seems to be as depicted in Fig. 21.1.
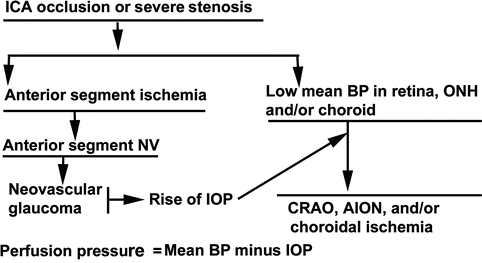

Fig. 21.1
Diagrammatic representation of sequence of events in eyes with ocular ischemic syndrome due to internal carotid artery occlusion or severe stenosis. Abbreviation: AION anterior ischemic optic neuropathy, BP blood pressure, CRAO central retinal artery occlusion, ICA internal carotid artery, IOP intraocular pressure, NV neovascularization, ONH optic nerve head
Ischemia of the Anterior Segment
The anterior segment of the eye is supplied by the anterior ciliary arteries (branches from various recti) and the long posterior ciliary arteries (see Chap. 6). Apart from the factors discussed in Chap. 20, which can produce ischemia of the anterior segment, it is a universal finding in OIS. In the anterior segment, OIS produces the following circulatory changes.
Fluorescein iris angiography shows poor filling of the iris (Figs. 21.2, 21.3, and 21.4). Iris neovascularization is usually the earliest manifestation of OIS (Figs. 21.2, 21.3, 21.4, and 21.5); that, with the development of neovascularization of the angle of the anterior chamber and peripheral anterior synechiae, results in neovascular glaucoma and a rise of intraocular pressure. The latter can play an important role in the development of various other manifestations of OIS discussed below and shown in Fig. 21.1. However, if there is ciliary body ischemia, that can result in ciliary body atrophy [43] and consequently decreased aqueous humor production and hypotony. So it is not uncommon to find no notable rise of intraocular pressure because of the associated chronic ischemia of the ciliary body. Bietti and Neuschuler [44], in 13 patients with internal carotid artery occlusion or severe stenosis, found 3–8 mmHg lower intraocular pressure (IOP) on the involved side than on the other side with a normal internal carotid artery. Some of these patients may be seen for the first time weeks or months after the development of neovascular glaucoma, and the intraocular pressure may be within normal limits or even lower, because of the ciliary body ischemic degeneration after a prolonged initial period of high intraocular pressure, thus misleading one into believing that the eye did not have glaucoma. Other findings of anterior segment ischemia seen in OIS are discussed below.
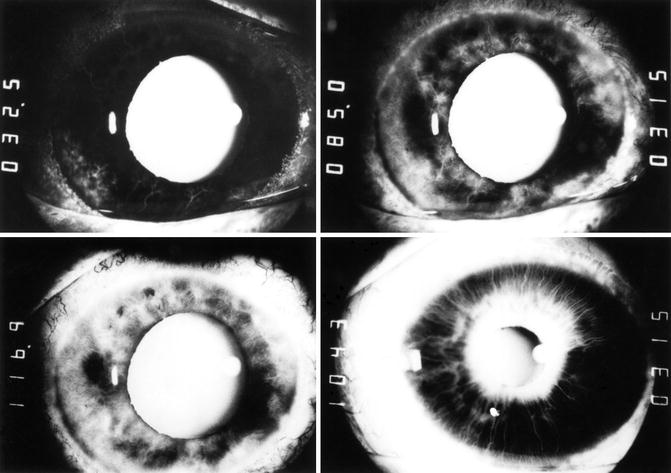
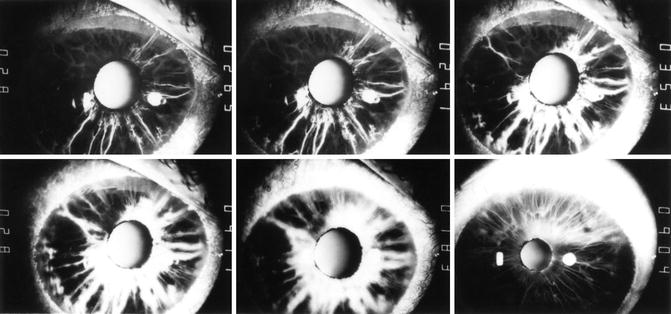
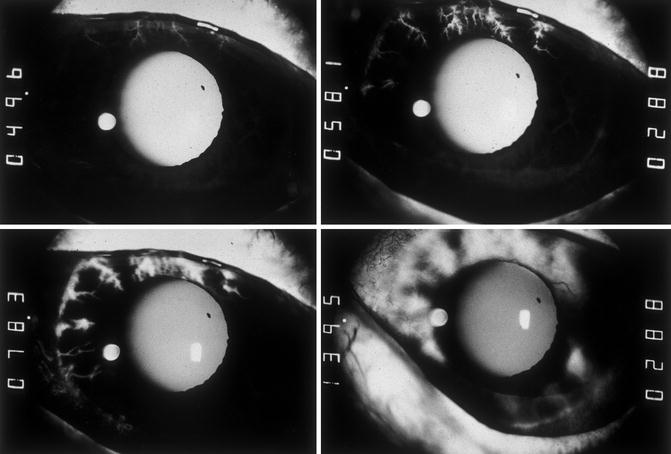
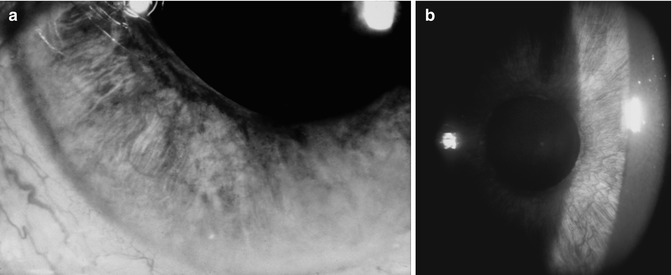

Fig. 21.2
Fluorescein iris angiograms of both eyes of case 2 with OIS. Left eye: top left and right and bottom left. Right eye: bottom right. Time indicated on the left side of each angiogram is in seconds after injection of the dye. In the top left angiogram, most of vessels in the iris, conjunctiva, and episclera showed no filling. Top right showed only patchy filling of the iris, with localized fluorescein leakage and no filling of most of vessels of the nasal conjunctiva and episclera. Bottom left and right show late staining of the iris in areas of iris neovascularization; few patches of the iris (bottom left) still show no filling (Reproduced from Hayreh and Podhajsky [42])

Fig. 21.3
Fluorescein iris angiograms of both eyes of a 72-year-old woman with OIS. Right eye: top row. Left eye: bottom row. Time indicated on the right side of each angiogram is in seconds after injection of the dye. These show late, poor, patchy, and slow progressive filling of iris vessels and late staining of the iris in areas of iris neovascularization

Fig. 21.4
Fluorescein iris angiograms of an 83-year-old man with OIS with most of vessels in the iris, conjunctiva, and episclera showed no filling. Time on the left side of each angiogram is indicated in seconds after injection of the dye

Fig. 21.5
(a, b) These show iris neovascularization with OIS. (b) Shows a slit lamp view
Ischemia of the Posterior Segment
Internal carotid artery occlusion or severe stenosis, in the absence of adequate collateral circulation, must obviously produce an extreme fall in the blood pressure of the ophthalmic artery, central retinal artery and posterior ciliary arteries. Ophthalmic artery stenosis or occlusion alone can produce the same hemodynamic effects as stenosis or occlusion of the internal carotid artery. The ophthalmic artery stenosis or occlusion alone has particular importance because in these cases of OIS Doppler evaluation of the carotid arteries may show no stenosis.
The following formula is used to calculate the ocular blood flow:
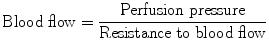
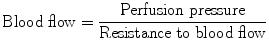
Perfusion pressure = mean arterial blood pressure in the retinal/choroidal vessels minus intraocular pressure.
Thus, in OIS the blood flow in the ocular arteries depends upon three factors: (a) resistance to blood flow, (b) blood pressure, and (c) intraocular pressure.
In OIS, even if the intraocular pressure is “normal” or only moderately elevated, in the presence of a very low mean blood pressure in the central retinal artery and posterior ciliary arteries due to occlusion or marked stenosis of the internal carotid artery or ophthalmic artery, that is more than sufficient to lower the perfusion pressure far enough to impair the retinal, choroidal, and optic nerve head blood flow. Moreover, most likely there are atherosclerotic changes in the ophthalmic and ocular arteries, which would increase vascular resistance. If the intraocular pressure is rising gradually with the onset of neovascular glaucoma, the blood flow in the central retinal artery and posterior ciliary artery may be accordingly compromised gradually, producing gradually progressive visual loss or recurrent episodes of amaurosis fugax rather than sudden blindness. This can be caused even by squeezing of the eyelids on blinking or stooping down, with the consequent rise of intraocular pressure, or by orthostatic hypotension. That may be enough to interfere momentarily with the circulation in the retina, optic nerve head, and choroid; this has been reported in the literature and was also seen in some of my patients with OIS [45]. A sudden and prolonged fall of systemic blood pressure for any reason, e.g., during sleep, can produce a sudden stoppage of the circulation in the central retinal artery and development of central retinal artery occlusion (see Chap. 13). A similar fall in the perfusion pressure in the posterior ciliary arteries would produce choroidal and optic disc ischemia; this was clearly demonstrated on fluorescein angiography. My case report below is a good example of that. Raitta [46] also reported a similar case. Ophthalmoscopically, choroidal infarcts and optic disc ischemia (optic disc cupping or anterior ischemic optic neuropathy) have been seen by me [45] and other workers (see below). Ruckman and Haas [47] and Russell [48] attributed the ocular ischemia in internal carotid artery occlusion to the “steal” phenomenon produced by a retrograde flow of blood in the ophthalmic artery from the external carotid artery to the internal carotid artery, as has also been reported by others [49, 50].
Thus, a patient with complete occlusion or severe stenosis of the internal carotid artery should be considered a potential candidate for anterior segment neovascularization, neovascular glaucoma, central retinal artery occlusion, and anterior ischemic optic neuropathy (Fig. 21.1); the reverse must also be borne in mind. These ocular manifestations may be the earliest clinical signs of a serious carotid artery disease, requiring urgent attention to prevent cerebral ischemic lesions, myocardial ischemia, or other disabling complications.
My Studies on Ocular Ischemic Syndrome
Almost all of the published data are either case reports or small retrospective series. I prospectively studied patients with OIS in my clinic from 1973 to 1993, in an attempt to improve our understanding of its clinical features and association with various systemic disorders [42, 45]. There were 32 patients (39 eyes) in my study [45]. Briefly, following were the findings.
The age range of the patients was 50–86 (mean ± SD 68 ± 8) years, and there were 19 men and 13 women. Symptoms were sudden visual loss in 41 %, gradual loss in 28 %, amaurosis fugax in 15 %, ocular/orbital pain in 13 %, and asymptomatic 21 %. Some of the patients who developed sudden visual loss noticed a red hue with that, or preceding amaurosis fugax or ocular/orbital pain. Visual acuity at initial examination was 20/40 or better in 15 %, 20/50–20/300 in 21 %, and 20/400 or less in 64 %. Final visual acuity on a follow-up (median 1 year) was 20/40 or better in 17 %, 20/50–20/300 in 7 %, and 20/400 or less in 77 %. Visual fields plotted with a Goldmann perimeter showed no defect in 23 %, central scotoma in 27 %, centrocecal defect in 5 %, and temporal island field only in 23 % and other types in 23 %. Intraocular pressure range was 4–60 (median 18) mmHg.
Ocular examination findings at initial visit are given in Table 21.1. In all eyes with iris/angle neovascularization, there was a variable degree of flare, being fairly marked in 62 % of the eyes. Fluorescein fundus angiography could be done in 22 affected eyes. That showed prolonged choroidal filling and retinal arteriovenous circulation times. In five eyes, filling of the posterior ciliary arteries (medial posterior ciliary artery in four and lateral in one) was defective. Three eyes had retinal arteriovenous times greater than 300 s. Four eyes had late staining of both retinal arterioles and veins, three had late staining of veins only, and one had late staining of arterioles only (see below in case 1). None of the eyes showed any retinal capillary nonperfusion.
Table 21.1
Ocular examination signs at initial visit
Segment of the eye | Signs | % of eyes |
|---|---|---|
Anterior segment | Conjunctival injection | 13 |
Cloudy cornea | 18 | |
Iris neovascularization | 87 | |
Angle neovascularization | 59 | |
Peripheral anterior synechiae | 31 | |
Markedly asymmetric cataract | 8 | |
Prior cataract surgery | 15 | |
Posterior segment | No view of posterior pole from cataract or corneal edema | 3 |
Optic disc | Pale | 28 |
Cupped and pale | 10 | |
Cupped (not pale) | 8 | |
Edematous | 8 | |
Neovascularization | 13 | |
Retina | Neovascularization | 3 |
Superficial hemorrhages – mostly in temporal periphery | 23 | |
Boxcarring in vessels | 21 | |
Cherry-red spot | 18 | |
Spontaneous pulsations of CRA | 8 | |
CRA collapse with gentle pressure on globe | 13 | |
Nonischemic central retinal vein occlusion | 3 | |
Multiple embolic plaques | 3 | |
Retinal arterioles attenuated and/or sclerosed | 21 | |
Choroid | Infarcts | 5 |
Chorioretinal degeneration | 3 |
Ipsilateral internal carotid artery disease was as follows: 100 % occlusion in 59 %, and 80–99 % stenosis in 15 %, and 0–49 % in 26 %. Eight patients had carotid endarterectomy on the side of the affected eye during follow-up; there was no significant difference in visual outcome between patients who had carotid endarterectomy and those who did not.
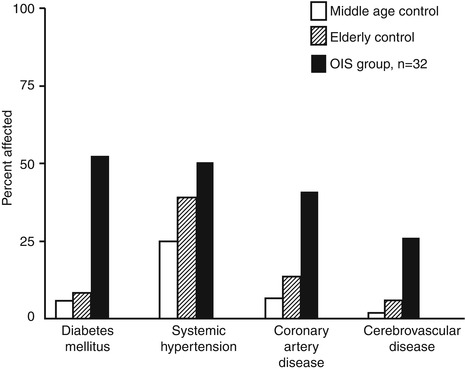
Figure 21.6 shows associated systemic diseases and a graphic representation of the increased incidence of systemic arterial hypertension, coronary artery disease, stroke or transient ischemic attacks, and diabetes mellitus in the patients with OIS compared to the general population [51]. This study showed that the incidence of diabetes mellitus is much higher in patients with OIS than in the comparable general population [51]. Of the 32 patients, 3 were nonsmokers, smoking history was not available for four, and 25 were smokers; the mean number of cigarette pack-years (multiplying the number of packs smoked per day by the number of years smoked) was 50 (range, 15–135 pack-years).
Bilateral OIS was seen in 7 of the 32 (22 %) patients. Among them, the internal carotid artery showed bilateral complete occlusion in one, bilateral severe stenosis in one, complete occlusion on one side and severe stenosis on the other side in two, complete occlusion on one side and mild stenosis on the other side in two, and no detectable stenosis on either side in one. In the remaining 25 patients with unilateral OIS, the internal carotid artery on the side of OIS showed complete occlusion in 17 (68 %), severe stenosis in 2 (8 %), mild stenosis in 3 (12 %), and no stenosis in 3 (12 %). In all but two patients, the OIS occurred on the side ipsilateral to the more severe flow limitation. Thus, in summary, ipsilateral internal carotid artery findings were as follows: 100 % occlusion in 59 %, 80–99 % stenosis in 15 %, and ≤49 % stenosis in 26 %. The fact that 26 % had only mild or no stenosis of the internal carotid artery is important, because of the common belief that all eyes with OIS have severe stenosis or occlusion of the internal carotid artery. In some such cases, the site of stenosis may be in the ophthalmic artery at its site of origin. In my anatomical studies [52] on the human ophthalmic artery, in a few specimen, I found that the orifice of the ophthalmic artery at its origin from the internal carotid artery was markedly stenosed, even when the internal carotid artery was of normal caliber; therefore, ophthalmic artery stenosis or occlusion can produce hemodynamic effects without internal carotid artery stenosis or occlusion.
The following three case reports of patients with OIS, which I reported in 1982 [50], are instructive about clinical findings in OIS.
OIS Case 1
A 61-year-old man was seen in my clinic complaining of intermittent blurring of vision in his right eye for 6 months. At his first visit, his visual acuity was 6/300 in that eye, with relative afferent pupillary defect. Examination of the anterior segment revealed extensive neovascularization of the iris and complete peripheral anterior synechiae of the angle of the anterior chamber and intraocular pressure of 26 mmHg (intraocular pressure in the fellow normal eye was 19 mmHg). Examination of the fundus revealed slightly engorged retinal veins (Fig. 21.7a) and a couple of very tiny retinal hemorrhages in the temporal periphery, with no other abnormality. Fluorescein fundus angiographic findings are shown in Figs. 21.7b–f. They revealed extremely slow filling of the retinal, choroidal, and optic disc circulation, with no retinal microvascular abnormality, except for an occasional microaneurysm. The central retinal artery at the disc did not start to fill till 22 s (Fig. 21.7b) (normally 10–12 s) after the injection of the dye and the retinal arteriole did not fill completely, even 60 s later (Fig. 21.7e) (normally within I second). Nasal choroid (i.e., in the distribution of the medial posterior ciliary artery) did not start to fill until 26 s after the injection of the dye (Fig. 21.7c) – normally it should start filling just before or simultaneously with the start of central retinal artery filling on the disc. The nasal choroid filled completely in about 20 s (normally less than a second), while the temporal choroid (i.e., in the distribution of the lateral posterior ciliary artery) showed no filling till about 43 s after the injection of the dye (normally it should fill synchronously with medial posterior ciliary artery). The optic disc started to fill with the filling of the medial posterior ciliary artery (Fig. 21.7c) which supplied the entire disc. The retinal vein had not filled even 52 s after the injection of the dye (Fig. 21.7e) (normal retinal arteriovenous circulation time is only 2–3 s). Thus, there was a gross delay in the entire circulation of the posterior segment. By the late phase (i.e., about 15 min after the injection of the dye – Fig. 21.7f), the retinal and choroidal vascular bed had filled completely, and the main retinal vessels leaked fluorescein and showed staining. Carotid arteriography revealed a total occlusion of the right internal carotid artery.
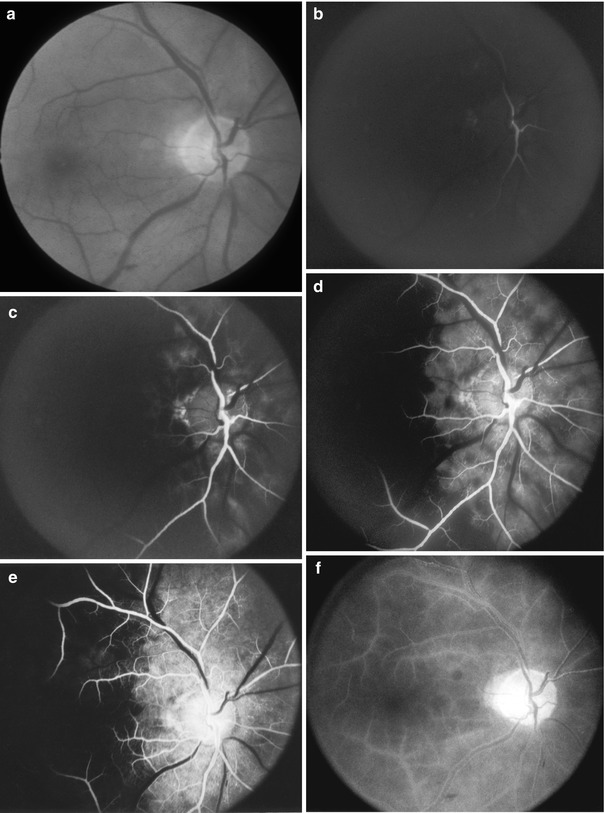

Fig. 21.7
Fundus photograph and fluorescein fundus angiograms of the right eye of case 1 with OIS (Reproduced from Hayreh and Podhajsky [42]). (a) Fundus photograph shows engorged retinal veins with no other abnormality. (b–f) Fluorescein fundus angiograms: time after injection of dye – (b) 21.8 s, (c) 25.7 s, (d) 42.6 s, and (e) 52.1 s. Retinal arteries started to fill on the optic disc (b) in about 20 s (normal, 10–12 s). The choroid, supplied by the medial posterior ciliary artery (PCA), and optic disc started to fill (c) in about 26 s (normally, the choroid starts to fill about 1 s before retinal arteries) and took about 14 s to fill completely, by which time the choroid supplied by the lateral PCA still showed no filling (d) (normally, both PCAs fill almost simultaneously); it showed no appreciable progress in filling (e). Main retinal arteries took more than 5 s to fill (they normally fill in fraction of second), and retinal veins had not filled even by 52 s (normal retinal arteriovenous filling time is 2–3 s). Thus, there was a severe abnormality in the filling sequence of three arteries (central retinal artery and medial and lateral PCAs) indicating different perfusion pressures in each of them. During late phase (f), the entire choroidal and retinal vascular beds filled, with staining of retinal vessels and fluorescein leakage along them
On follow-up, 3 months after the initial visit, the right eye had a visual acuity of bare hand motion, intraocular pressure of 25 mmHg, cupped optic disc, and pulsating central retinal artery on the disc. Three months later, the visual acuity in that eye was no light perception, intraocular pressure 19 mmHg, almost complete obliteration of the retinal vessels and evidence of old peripheral choroidal infarct over the 360° of the fundus (fundus findings identical to those are seen in Figs. 21.8, 21.9, and 21.10).
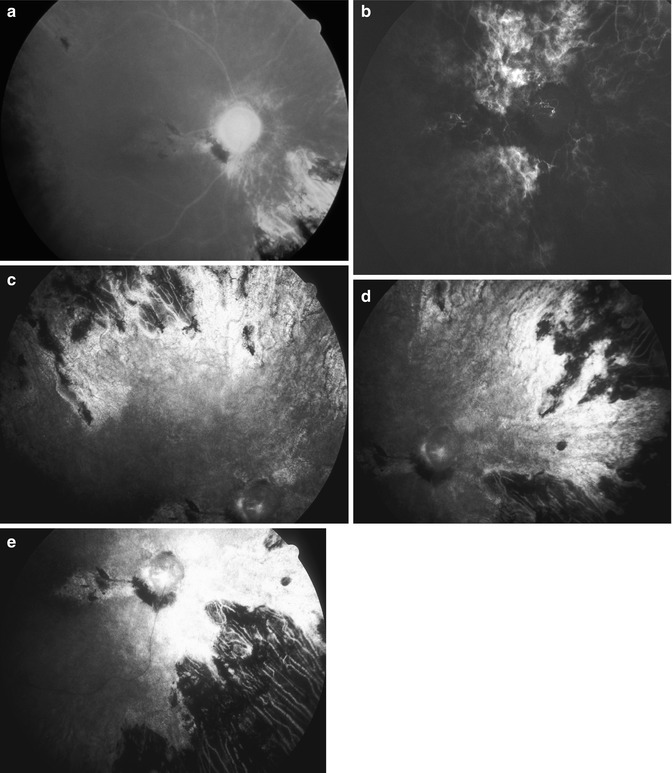
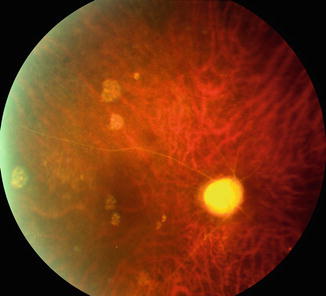
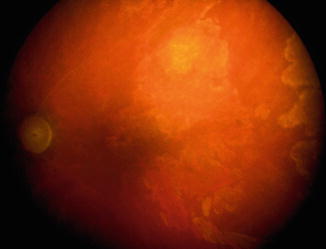

Fig. 21.8
Fundus photograph and fluorescein fundus angiograms of the right eye of a 54-year-old man with OIS and internal carotid artery occlusion, with marked ischemia of anterior and posterior segments of the eye. (a) Fundus photograph shows markedly attenuated and sheathed retinal vessels and chorioretinal lesions due to choroidal ischemia. (b–e) Fluorescein fundus angiograms: (b) during the transit of the dye shows no filling of the central retinal vessels and mild patch filling of the choroid and (c–e) during late phase show areas of chorioretinal lesions in different parts of the periphery and no filling of the retinal vessels

Fig. 21.9
Fundus photograph of right eye of a 76-year-old man with OIS, which developed following shoulder surgery, which lasted for 2 h in sitting position. It shows markedly attenuated and sheathed retinal vessels and chorioretinal lesions due to choroidal ischemia

Fig. 21.10
Fundus photograph of the left eye of a 50-year-old woman with diabetes mellitus, arterial hypertension, carotid artery occlusion, myocardial infarction, and transient ischemic attacks. It shows markedly attenuated and sheathed retinal vessels and extensive chorioretinal lesions due to choroidal ischemia
There can be no disagreement that such fundus changes are due to OIS secondary to carotid artery occlusive disease. These eyes not only have sluggish circulation in the retinal vascular bed but also have as poor or poorer circulation in the choroid, optic disc, and iris. I have discussed this subject in detail elsewhere [45, 50].
OIS Case 2
A 70-year-old man, at his initial visit to my clinic, gave a history that 6 days previously, he woke up in the morning with blurred vision in the left eye and recurrent attacks of pain in and around the eye. Previously, he had had recurrent attacks of amaurosis fugax. On examination, the visual acuity was counting fingers in the left eye and 20/25 in the right eye. The anterior segment in the left eye showed neovascularization of the iris and angle all over (Fig. 21.2), with open angle, and in the right eye, there was a small patch of iris neovascularization without any angle neovascularization. The IOP was 34 mmHg in the left eye and 17 mmHg in the right eye. The left fundus showed evidence of cilioretinal artery occlusion, with the retinal infarct involving the macular region and pulsating and narrow retinal arteries, with a cup-disc ratio of 0.9. The right fundus was within normal limits except for a cup-disc ratio of 0.8.
Systemic Findings
The patient had had a history of dizzy spells for 2–3 years – especially on changing positions of the head, transient ischemic attacks for about 3 weeks, and an abnormal electroencephalography suggesting a focal hemisphere pathologic condition that was greater in the left than the right hemisphere. Aortic arch angiography at that time showed the presence of bilateral occlusion of the internal carotid arteries at their origin, mild stenosis of proximal vertebral arteries bilaterally, and a second stenosis in the midcervical part of the left vertebral artery. He had had three myocardial infarctions between 13 and 9 years earlier. There was a history of angina pectoris and also pain in the legs on walking. He had been discovered to have chemical diabetes and hyperlipidemia 3 years earlier.
Follow-Up
The patient was treated with eye drops for elevated IOP without any change in the IOP. Two days later, while the retinal arteries in the left eye were still pulsating and the eye was painful, with an IOP of 40 mmHg, 500 mg of acetazolamide Sequel orally twice a day was prescribed. The next day, although the IOP was 26 mmHg, the retinal arteries were still pulsating, and the patient was having frequent syncopal episodes. When seen 1 week later, the visual acuity was no light perception in the left eye, and the fundus showed the picture of central retinal artery occlusion. The IOP was 26 mmHg in the left and 12 mmHg in the right eye. He was having blackout attacks. To relieve the constant pain in the left eye, he received a retrobulbar alcohol injection. When seen 18 months after the initial visit, the left eye was phthisical, and, in the right eye, he was having episodes of visual obscuration; the IOP was 21 mmHg in the right eye, with a visual acuity of 20/30, and the fundus was within normal limits. Three years after the initial visit, he underwent a right carotid endarterectomy. When last seen 6 weeks after that, he was relieved of the amaurosis fugax attacks in the right eye. The patient died of cardiac arrest 7½ months after his last visit.
OIS Case 3
A 59-year-old man was first seen in an emergency room with the following history. About 2 weeks previously, he had started to lose vision in his right eye, with right-sided headaches. The visual loss was not sudden but gradual, so that he lost vision completely over 3–4 days. On examination, visual acuity was no light perception in the right and 20/20 in the left eye. The anterior segment of the right eye showed the presence of severe neovascularization of the iris with a fibrovascular sheet in the angle and total peripheral anterior synechiae. The anterior segment of the left eye was normal. The IOP was 45 and 10 mmHg in the right and left eye, respectively. The right fundus showed an almost empty vascular bed with retinal opacity and a pale disc, with no other abnormality. The left fundus was within normal limits.
Systemic Findings
The patient had had high blood pressure for 15 years. He had had hyperglycemia (blood glucose level ≤150 mg/dL, without any therapy) for a few years as well as elevated triglyceride levels. Seventeen months earlier, he had two right-sided strokes at an interval of 1 day. Aortic arch angiography 2 months after that disclosed a totally occluded right internal carotid artery, with the left internal carotid artery within normal limits, and both external carotid arteries were slightly narrowed. He had below-the-knee amputation of both legs – first on the right side 27 months before he was seen in my clinic and then 2 years after that on the left side, because of peripheral vascular disease. He had smoked two packs of cigarettes a day for about 40 years.
Follow-Up
When seen 10 months after the initial visit, the IOP was 45 mmHg in the right eye, and, 6 months after that 22 mmHg, with severe cupping of the right optic disc, triangular sector-shaped chorioretinal degenerative patches in the temporal half, and diffuse chorioretinal degenerative changes in the nasal half caused by choroidal ischemia. The left eye was essentially normal. When seen 21 months after the initial visit, the left eye had developed a mild venous stasis retinopathy with a VA of 20/20. The IOP was 35 and 11 mmHg in the right and left eye, respectively. The right lens had developed cataractous changes. Aortic arch angiography, 2 years after the initial visit, showed total occlusion of the right common and internal carotid arteries, severe occlusion of the left internal carotid artery, and a small plaque in the left common carotid artery. Three days later, he underwent a left carotid endarterectomy. When last seen 2¼ years after the initial visit, the visual status was almost the same as at the last visit, and the patient was feeling much better, in general.
Signs and Symptoms of OIS
The visual symptoms in OIS, shown by these three case reports, include amaurosis fugax (often recurrent), and gradual or sudden visual loss. Some of the patients who develop sudden visual loss notice a red hue with that, or preceding amaurosis fugax or ocular/orbital pain. These are common symptoms of OIS reported in the literature. Amaurosis fugax may be light induced [53–56], i.e., precipitated by exposure to bright light; this is an uncommon manifestation of OIS. When IOP is high, the eye may be painful. Some patients may complain of orbital pain (“orbital angina”), which could be compared to having angina in coronary ischemia; there is moderate to severe pain in the eye, orbit, temple, and upper part of the face on the side of severe carotid artery occlusion – because of ischemia.
Visual acuity and visual fields may vary from being normal to marked loss, even no light perception, depending upon the severity of OIS. Findings in my study are shown in Table 21.1.
Ocular signs of OIS can be categorized into those in the anterior and in the posterior segment.
Anterior Segment Signs
Flare in the anterior chamber is common, and rarely there may be hyphema; these findings may mimic anterior uveitis and have been misdiagnosed as “iridocyclitis.” Iris neovascularization is the most common finding (Figs. 21.2, 21.3, 21.4, and 21.5). Neovascularization may also involve the angle of the anterior chamber, which may result in development of peripheral anterior synechiae and neovascular glaucoma; however, there may be ocular hypotony [43, 44]. There may be iris atrophy. There may be conjunctival and episcleral vascular congestion (Figs. 21.11 and 21.12) and a cloudy cornea from neovascular glaucoma. The pupil may show a sluggish reaction to light, or there may be posterior synechiae (Fig. 21.13). In addition to these changes, OIS can produce lid ischemia [57], development of spontaneous bulbar conjunctival defect, which can progress to subsequent adjacent corneal perforation [58], and corneal edema [57]. Melting and necrosis of sclera have also been reported with OIS [58]. I have seen corneal melting at the limbus (Fig. 21.12). There may be asymmetric cataract formation. In severe OIS cases, there may be signs of anterior segment ischemia, discussed in Chap. 20.
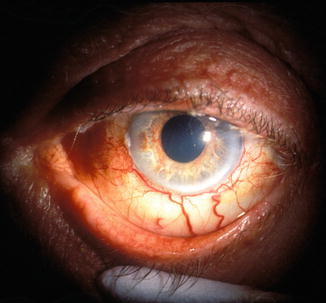
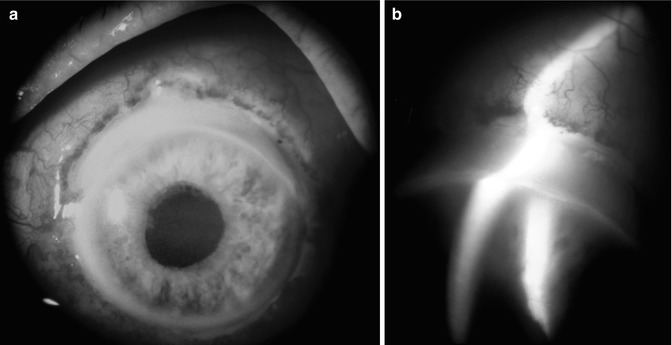
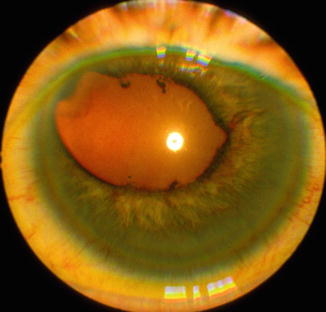

Fig. 21.11
Photograph of the anterior segment of a 55-year-old man with OIS shows iris neovascularization and dilated episcleral vessels

Fig. 21.12
(a, b) Photographs of the anterior segment of an eye shows limbal gutter due to melting of the peripheral cornea in an eye with OIS. (b) Is a slit lamp photograph

Fig. 21.13
Photograph of the anterior segment of the eye with OIS shows eccentric, dilated, and irregular pupil with iris neovascularization and posterior and peripheral anterior synechiae, and neovascular glaucoma
Fluorescein iris angiography shows notably delayed and poor filling of all the vessels in the anterior segment, including the iris, and iris neovascularization. It could be argued that poor and delayed filling of the iris vessels is caused by a raised IOP, but the corresponding delay in filling of the conjunctival and episcleral vessels (Figs. 21.2, 21.3, and 21.4) cannot be explained on that basis. This is due to carotid artery occlusion.
Posterior Segment Signs
These include various ischemic signs of the retina, choroid, and optic nerve head. The patient may present with a spontaneous pulsating central retinal artery at the optic disc, central retinal artery occlusion (Fig. 21.14), cilioretinal artery occlusion, cotton-wool spots, optic disc edema due to anterior ischemic optic neuropathy, patchy choroidal infarcts, chorioretinal degeneration patches from old choroidal infarcts (Figs. 21.8, 21.9, and 21.10), attenuated or sheathed retinal arterioles (Figs. 21.8a, 21.9, 21.10, and 21.14b), engorged retinal veins (Fig. 21.7a), and retinal hemorrhages (often located in midperipheral or temporal periphery). There may be macular edema [59–63] – due to associated diabetic retinopathy or central retinal vein occlusion (see below). The optic disc may be atrophic or cupped (Figs. 21.8a, 21.9, and 21.10). There may be retinal and/or optic disc neovascularization (Figs. 21.15 and 21.16). There may be microaneurysms and perifoveal telangiectasias – due to associated retinopathy.
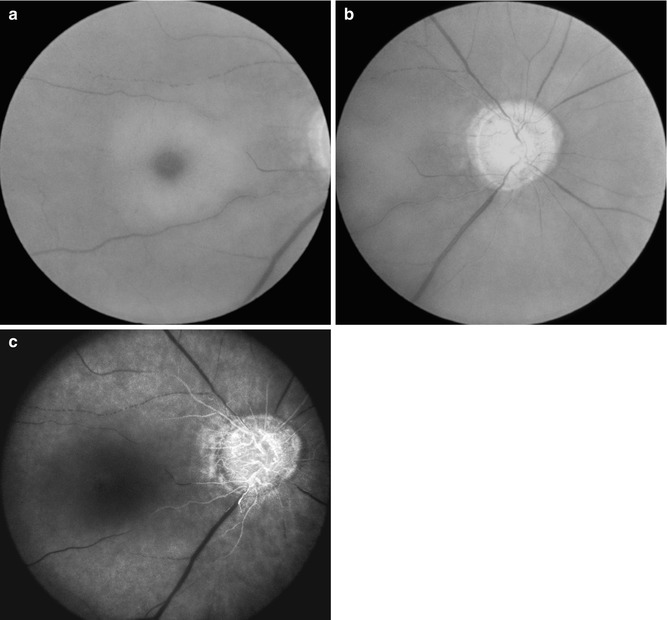
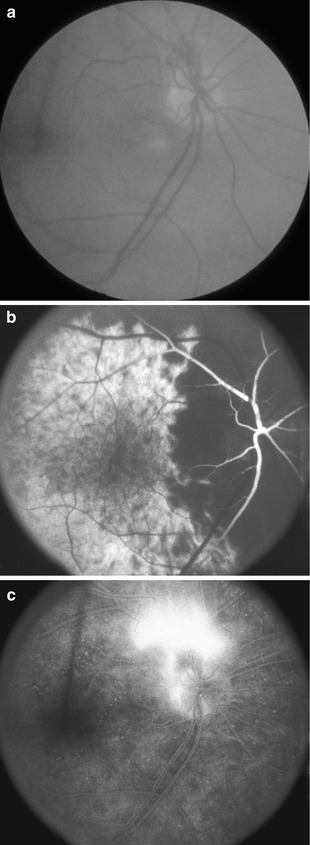
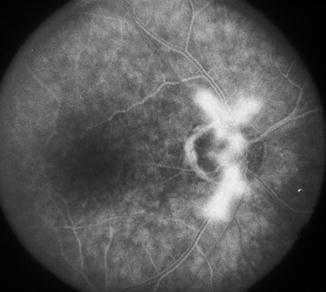

Fig. 21.14
Fundus photographs and fluorescein fundus angiogram of the right eye of a 72-year-old woman with OIS. (a, b) Fundus photographs show central retinal artery occlusion, with cherry-red spot, acute retinal ischemia, attenuated vessels, boxcarring (cattle trucking), and pale optic disc. (c) Fluorescein fundus angiogram shows no filling of the central retinal artery, but shows filling of the optic disc and poor and late filling of the choroid

Fig. 21.15
Fundus photograph and fluorescein fundus angiograms of the right eye of a 55-year-old woman with OIS. (a) Fundus photograph shows optic disc neovascular frond above the optic disc. (b) Fluorescein fundus angiogram shows no filling of the medial posterior ciliary artery with slow, late, and part filling of the central retinal artery. (c) Fluorescein fundus angiogram during the late phase shows fluorescein leak from the neovascular frond

Fig. 21.16
Fluorescein fundus angiogram during the late phase of the right eye with OIS shows fluorescein leak from the optic disc neovascular frond and poor filling of the retinal vessel and the choroid
Fluorescein fundus angiography provides the most useful information about the state of impaired retinal and choroidal circulation (Figs. 21.7, 21.8, 21.14, and 21.15). There may be sluggish filling of the retinal and/or choroidal vascular bed; in severe cases, the retinal vascular bed does not fill at all, and the choroidal vascular bed filling may be markedly reduced or filling of one posterior ciliary artery may be much more delayed than the other posterior ciliary artery. There may be late staining of retinal arterioles and/or veins (Fig. 21.7f). Figures 21.8, 21.9, and 21.10 show advanced changes in the fundus, retina, and choroid in OIS.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree