Ocular Circulation
Alon Harris
Matthew Ralstin
Ehud Rechtman
Thomas A. Ciulla
The eye is a unique organ. Under normal circumstances, most of the human vasculature is inaccessible for direct observation. However, the eye provides an exception. Because of the clarity of the normal ocular media, the retinal vasculature may be observed directly in vivo using simple tools. Since the advent of the ophthalmoscope, there have been attempts to visualize and understand the ocular vasculature.1 The progression of some systemic vasculopathic conditions, such as diabetes mellitus and hypertension can be monitored with serial ophthalmoscopic examinations. Additionally, examination of the external ocular circulation may suggest the presence of other systemic disease states. For example, conjunctival comma-shaped capillaries suggest sickle cell disease and arterialized episcleral vessels are often observed with carotid-cavernous sinus fistulas. In addition to systemic disease states, ophthalmic disorders may give rise to, or result from, ocular circulatory pathologies. Vascular occlusions and vessel malformations are common within the orbit. In disease such as diabetic retinopathy, the vascular component is well established. In others, such as glaucoma, the role of altered hemodynamics is still debated, although mounting evidence suggests it plays a role in the pathogenesis.2,3,4,5,6,7
The first assessments of ocular circulation were confined to subjective physical descriptions of visible vessels. Since that time, the assessment of ocular circulation has progressed to direct and indirect quantitative measurement of a number of ocular hemodynamic parameters. Over the last few decades, advances in technology have led to a variety of methods that are acceptable for in vivo assessment of ocular hemodynamics in humans. The result has been a great increase in the understanding of ocular circulation, both in health and disease.
This chapter outlines the basic anatomy and physiology of the ocular circulation. The unique aspects of each regional intraocular microcirculation are discussed. Additionally, methods for the assessment of ocular hemodynamics are addressed, because the clinical relevance of these techniques is ever increasing. Finally, there is a brief discussion of the role of ocular hemodynamics in the pathogenesis of diabetic retinopathy and primary open-angle glaucoma (POAG).
Gross Anatomy of the Ocular Circulation
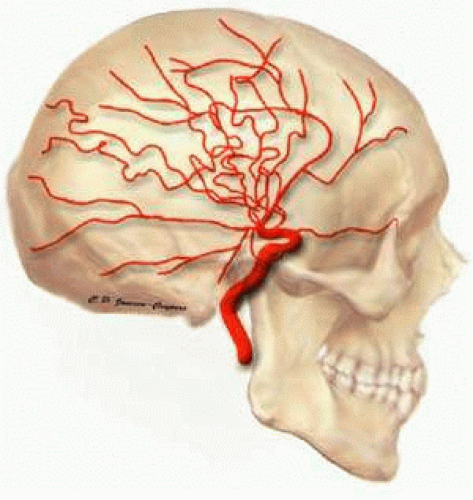
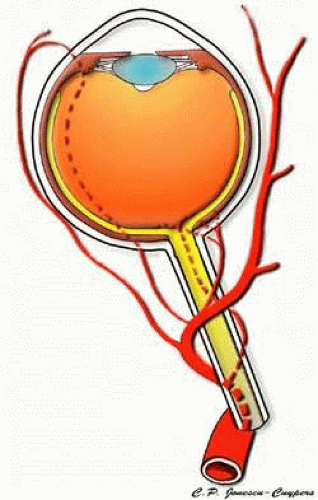
The blood supply to the eye and orbit arises primarily from the ophthalmic artery (OA). The OA is the first branch of the internal carotid artery and the only branch of the internal carotid that originates outside of the cranium (Fig. 1). In <5% of people, the ophthalmic artery receives most of its blood supply from the middle cerebral artery as a result of atypical enlargement of the normal anastomoses. The OA enters the orbit via the optic canal and runs inferolaterally to the optic nerve in most people. As the OA courses nasally and anteriorly, it then runs superior to the optic nerve and gives off most of its major branches.8 Its major branches include branches to extraocular muscles, the central retinal artery, and the posterior ciliary arteries (Fig. 2).9 Several of the branches of the OA, including the supraorbital, lateral and medial palpebral, and lacrimal arteries, communicate with the facial artery to form an anastomosis between the internal and external carotid arteries.
The central retinal artery (CRA) branches off the OA to pierce the ventral surface of the optic nerve sheath approximately 10 to 15 mm posterior to the lamina cribrosa.10,11 The CRA courses adjacent to the central retinal vein (CRV) within the central portion of the optic nerve. In most eyes, the CRA gives off a few (0 to 8) axial, intraneural branches,11,12 but it does not directly supply the lamina cribrosa area. The CRA emerges from the optic nerve within the globe, where it branches into four major vessels. The CRA supplies the inner two thirds of the retina, the superficial nerve fiber layer of the optic nerve head, and contributes to the supply of the retrolaminar optic nerve.
Typically, a medial and lateral posterior ciliary artery (PCA) branch off from the OA. Occasionally, an aberrant PCA is also present. Each PCA divides into one long posterior ciliary artery (LPCA) and seven to ten short posterior ciliary arteries (SPCAs) before piercing the sclera. The two long PCAs pierce the sclera about 3 to 4 mm nasally and temporally from the optic nerve and travel anteriorly within the suprachoroidal space, along the horizontal meridians of the globe. They typically divide in the vicinity of the ora serrata, sending recurrent branches posteriorly to supply the anterior choriocapillaris in concert with the anterior ciliary arteries. The SPCAs supply the posterior choriocapillaris, peripapillary choroid, and the majority of the anterior optic nerve. The choriocapillaris underneath the fovea is supplied by the lateral PCA.
The muscular branches of the OA travel within the rectus muscles. Typically, each rectus muscle is accompanied by two arteries, with the exception of the lateral rectus, which has only one. The muscular arteries supply the rectus muscles as well as running to the anterior aspect of the globe in company with the tendons of the rectus muscles to form the anterior ciliary arteries. The anterior ciliary arteries form a vascular zone beneath the conjunctiva and then pierce the sclera a short distance from the cornea. The anterior ciliary arteries are responsible for supply of the iris and ciliary body and contribute to the anterior choroidal circulation. Simultaneous surgical interruption of too many of the muscular branches may result in ischemic anterior segment damage.
The venous drainage of the eye generally follows the arterial supply. The CRV drains the retina and prelaminar aspect of the optic nerve, delivering the blood back through the orbit to the cavernous sinus. Vascular disease involving the cavernous sinus can, therefore, become manifest inside the eye, such as when a carotid artery-cavernous sinus fistula results in elevated retinal venous pressure and secondarily induces a CRV obstruction.
The choroid is drained through the vortex vein system. Four vortex veins, one in each quadrant, are typically present, although this is somewhat variable. The large and distinctive ampullae of the vortex veins can be visualized by ophthalmoscopy. The vortex veins drain into the superior and inferior orbital veins. The superior orbital vein then drains into the cavernous sinus, and the inferior orbital vein drains into the pterygoid plexus through the inferior orbital fissure. Collateralization between the superior and inferior orbital veins is common.
The iris and ciliary body are drained primarily through the anterior ciliary veins, which participate in the vortex vein system. However, some venous drainage of this area is via the episcleral veins into the external carotid system.
Variability of Vascular Patterns
Marked variation exists in the vascular patterns of the anterior optic nerve, peripallary retina, and the posterior choroid among individuals.9,11,13,14,15,16 Most of the variation arises from differences in arterial supply. Varying numbers of branches have been found in the PCAs, SPCAs, number of branches from the LPCAs, and number of branches from the anterior ciliary arteries (ACAs).
The OA crosses from lateral to medial above the optic nerve in about 80% of cases and below the nerve in about 20%.
Typically, one ciliary artery arises from the first segment of the OA and supplies the choroid of the nasal hemisphere and one ciliary arises from the second segment of the OA and supplies the choroid of the temporal hemisphere. However, instead of only medial and lateral arrangement of the PCAs, a third superior grouping may be present.13 The number of main PCAs varies between one and five, with most eyes having two or three.13,16
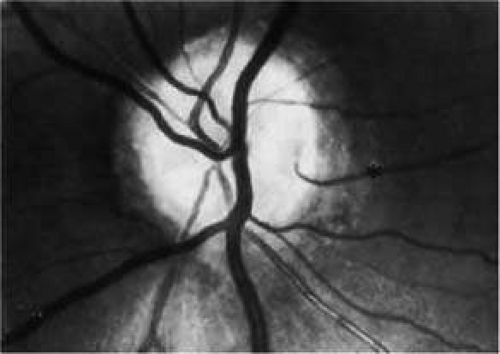
Cilioretinal arteries, which arise directly from ciliary arteries and supply a portion of the retina, occur in 20% to 25% of eyes.17,18 When viewed by ophthalmoscope, they typically appear to arise from the temporal portion of the optic nerve head and to course along the maculopapillary bundle (Fig. 3). Cilioretinal arteries may supply portions of the macula and are responsible for preservation of visual acuity in some individuals with CRA occlusion.19 Upon fluorescein angiography, cilioretinal arteries fill in the choroidal phase before filling of the CRA system.
Microscopic Anatomy of the Ocular Circulation
For the purpose of this discussion, the intraocular circulation is divided into the following areas: retina, optic nerve, choroid, ciliary body and iris. Each regional microcirculation is discussed individually.
Retina
An understanding of the retinal microcirculation is crucial for diagnosis and treatment of acquired retinal disease states, because the vast majority of nonheritable primary retinal diseases are vascular in origin. The nutritional and waste removal needs of the retina are met by two separate sources. The CRA supplies the inner two thirds of the retina. The outer retinal layers, including the photoreceptors and bipolar cells, are normally avascular and are supplied through diffusion from the choriocapillaris. The retinal pigment epithelial layer, which separates the retina from the choroid, actively transports metabolites and waste to and from the outer retina to the choroid. Despite this dual circulation to the retina, there appears to be functionally little overlap. This is evidenced by the structural abnormalities induced by acute obstructions of the retinal circulation (CRA obstruction or branch retinal artery obstruction). These abrupt diminutions in inner retinal blood flow result in physiologic and anatomical dysfunction that is localized to the inner retinal layers.20
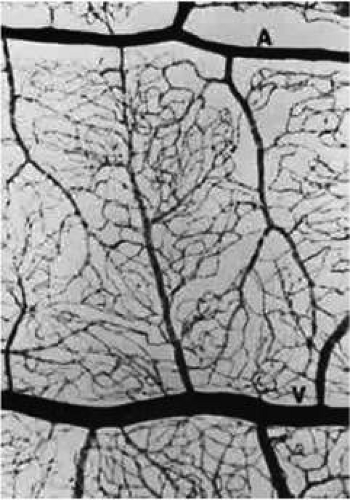
The CRA is an end artery without significant anastomoses.21 In the area of the lamina, its lumen measures about 170 μm in diameter. The CRA typically divides just before its exit from the optic nerve into superior and inferior branches. These, in turn divide again, with each of the four branches supplying roughly a quadrant of the retina (Fig. 4). The anatomy is variable, and the division may occur within the optic cup of the nerve as well.
 FIGURE 4. The CRA typically branches into four major trunks, each of which supplies a quadrant of the retina. Venous drainage of the closely follows the arterial supply. |
The retinal arteries and veins lie within the nerve fiber layer of the superficial retina. The major branch arteries are about 120 μm in diameter and are smaller in diameter and straighter in course than their accompanying draining veins. Branching of arteries is of two types: dichotomous and side-arm. Trifurcations are extremely rare. Smaller vessels arising from dichotomous branches course toward the periphery of the retina, becoming arterioles that supply the retina anterior to the equator. Side-arm branches are small precapillary arterioles branching from a larger vessel to supply the local capillary system; they leave the main arteries and enter the inner plexiform and ganglion cell layers. However, only capillaries are found as deep as the inner nuclear layer. The blood column within retinal arterioles is visible by direct ophthalmoscopy down to the third-order branches. Normally, the walls of the vessels themselves are transparent to direct observation; it is the blood column that makes the vessels visible by ophthalmoscopy.
The retinal capillaries run parallel to the retinal nerve fiber layer (Fig. 5). Throughout most of the posterior retina, the retinal capillaries are arranged in a laminated fashion with two layers.22 The retinal capillaries assume a meshwork configuration to ensure adequate perfusion to all retinal cells. The superficial capillary layer is associated with the ganglion cell layer and the deep capillary layer resides within the inner nuclear layer. In the posterior pole, the retinal capillaries are denser and may consist of three or four layers. In contrast, in the less dense peripheral retina, only one capillary layer is present. The extreme periphery is avascular. The peripapillary retina often has a superficial layer of fine capillaries. The central fovea is an avascular zone, which enables light to reach central photoreceptors without interference from any blood vessels, and underscores the reliance of the photoreceptors on the choroidal circulation.
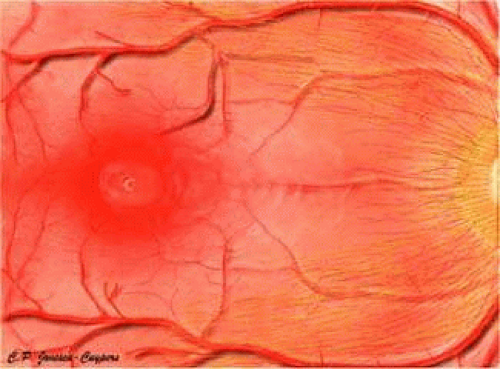 FIGURE 5. Arrangement of retinal capillaries with respect to the retinal nerve fiber layer and macula. |
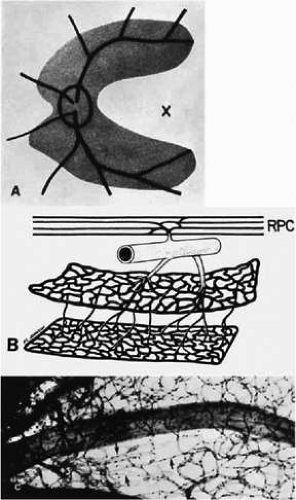
A capillary-free zone is present around each of the larger retinal arteries and veins (Fig. 6). It is more prominent around the arteries, measuring up to 100 μm. The capillary-free zone is a product of the embryologic vascular remodeling process, which is initiated by direct diffusion of oxygen through the walls of large retinal vessels, creating high local oxygen tension.23 During embryogenesis, numerous capillary-sized channels retract from the developing artery, leaving only relatively few right-angled side-arm channels to serve as precapillary arterioles. By reducing side channels to a minimum, the maintenance of adequate perfusion pressure for the remainder of the retina is achieved. In contrast, the radial peripapillary capillaries (RPCs) may develop later than the other capillary beds, after vascular remodeling has already occurred.24
A distinct layer of capillaries, the RPCs, is found adjacent to the optic disc, most prominently in the superior and inferior temporal aspects of the posterior pole (Fig. 7).25 These capillaries only originate from intraretinal arterioles and not from optic disc vasculature. The RPCs tend to follow relatively long, straight paths, having few anastomosis with adjacent or underlying capillary beds. The distribution pattern of RPCs suggests some anatomical correlation with various diseases. Cotton-wool spots are found in a distribution similar to RPCs, and arcuate scotomas observed in glaucoma appear to correspond to their pattern as well.26
Retinal veins and venules generally follow the course of the arteries and arterioles. Occasionally, retinal veins interdigitate with their associated arteries. When the two vessels cross, the artery usually lies anterior (toward the vitreous) to the vein.27 About one third of the time, however, the vein is anterior. Under normal circumstances, the crossing vein’s lumen may be decreased by as much as one third as a result of compression from the accompanying artery.28 There are many more arteriovenous crossings temporally than nasally, because the nasal vessels assume a much straighter course. Arteriovenous crossings are important because they represent the most common site of branch retinal vein obstructions. The retinal veins drain into the CRV, which exit the eye through the optic nerve and parallels the CRA. The CRV drains into the superior ophthalmic vein or, rarely, directly into the cavernous sinus.
Optic Nerve
Most of the blood supply to the intraorbital portion of the optic nerve is through numerous perforating vessels from the pia mater. The pial system is an anastomosing network of capillaries located immediately within the pia mater. The pial vessels obtain their supply either directly from the OA or indirectly from recurrent branches back from the PCAs. The branches of the pial system extend centripetally to nourish the axons of the optic nerve. The CRA may supply several small intraneural branches in the retrolaminar region. Some of the branches may anastomose with the pial system.
The precise microvascular anatomy of the anterior optic nerve and peripapillary region has been a source of considerable controversy. The optic nerve head is situated at the watershed area between three separate circulations: the retinal, the posterior ciliary, and the pial. Each of these systems contributes to the circulation of this area.
The anterior optic nerve is divided into four anatomic regions (Fig. 8). The only region visible by ophthalmoscope is the superficial nerve fiber layer. Immediately posterior to the nerve fiber layer is the prelaminar region, which lies adjacent to the peripapillary choroid. Posterior to the prelaminar region is the laminar region, which is continuous with the sclera and is composed of the lamina cribrosa, a structure consisting of fenestrated connective tissue lamellae, which allow neural fibers to pass through the scleral coat.29,30,31,32,33 Finally, the retrolaminar region lies posterior to the lamina cribrosa marked by the beginning of axonal myelination, and is surrounded by the meninges.
The superficial nerve fiber layer receives blood from recurrent retinal arterioles arising from the branches of the CRA (Fig. 9).11,34 These small vessels, termed epipapillary vessels, originate in the peripapillary nerve fiber layer and run toward the center of the optic nerve head.35 The temporal nerve fiber layer may have an arterial supply from the cilioretinal artery when it is present.36 No direct choroidal or choriocapillaris contribution is observed in this region.
The prelaminar region is mainly supplied by direct branches of the short PCAs and by branches of the circle of Zinn-Haller (Fig. 10).11,37 The circle of Zinn-Haller is a noncontinuous ring of arterioles that is located within the perineural sclera (Fig. 11). This structure is formed by the confluence of branches of the short PCAs. The arterial circle provides multiple perforating branches into the prelaminar region, lamina cribrosa, and retrolaminar pial system, to the peripapillary choroid, and the pial arterial system.10,11,15,16,38,39 Arterial branches emerge from the circle of Zinn-Haller to supply both the prelaminar and laminar regions. The direct arterial supply to the prelaminar regions arising from the choroidal vasculature is minimal. Several small centripetal arteries or arterioles branch from the larger vessels of the peripapillary choroid, but no direct connection between the peripapillary choriocapillaris and the prelaminar region exists.11,12,15,29
The lamina cribrosa region also receives its blood supply from branches of the short PCAs and circle of Zinn-Haller (Fig. 11).15,29,30,37 These precapillary branches perforate the outer aspects of the lamina cribrosa before branching into an intraseptal capillary network. As in the prelaminar regions, the larger vessels of the peripapillary choroid may contribute occasional small arterioles to the lamina cribrosa region.
The retrolaminar region is supplied by two different systems, the CRA and the pial system (Fig. 12).12,15,40 In this region, the pial system feeds off of the circle of Zinn-Haller and may also be fed directly by the short PCAs.10,15,37 The CRA may supply several small intraneural branches in the retrolaminar region, some of which may anastomose with the pial system.12,15,40,41
The venous drainage of the anterior optic nerve is almost exclusively via the CRV.10,15,37,42 In the nerve fiber layer, blood is drained directly into the retinal veins, which then join to form the CRV. In the prelaminar, laminar, and retrolaminar regions, venous drainage also occurs via the CRV or axial tributaries to the CRV.
Choroid
The choroid is the most vascular portion of the eye and by weight is one of the most vascular tissues in the body.41 More than 80% of all ocular blood flow goes to the choroid. The choroidal circulation is responsible for the nourishment of the photoreceptor/retinal pigment epithelium (RPE) complex. Along with the task of providing nourishment to the outer retina and RPE, the choroidal circulation has other roles. It serves as a heat sink, removing the large amount of heat that is created as a result of the metabolic processes that occur when photons strike the photosensitive pigments and RPE.43 Additionally, the choroid probably serves as a mechanical cushion for the internal structures of the eye.
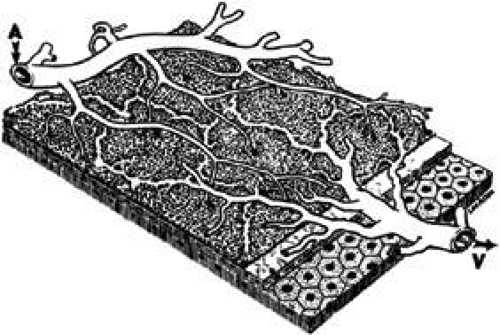
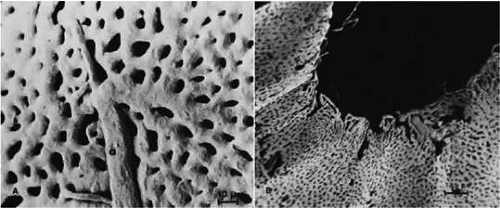
The choroidal circulation is organized in a segmental pattern.14,43,44,45,46 The segmental distribution of blood begins at the level of the posterior ciliary branches and is mirrored in the vortex vein drainage system. Because of the segmental organization, the large and medium-sized choroidal arteries function as end arteries. Each terminal choroidal artery supplies an independent lobule of choroidal capillaries (Figs. 13 and 14). Examination of the lobular structure reveals that feeding arterioles are usually found in the lobule center with draining venules located at the lobular periphery, but reversals of this pattern are frequently seen. There is little or no functional communication between adjacent capillary lobules.47,48,49,50
Within the choroid, the arteries and arterioles lie in stratified layers, with the larger vessels located in the outermost layer. The outer choroid is composed of large nonfenestrated vessels, whereas the vessels in the inner choroid have a much smaller caliber.41 The innermost layer of the choroid is the choriocapillaris, the unique capillary system of the choroid (Fig. 15A). The choriocapillaris is composed of richly anastomotic, fenestrated capillaries that begin at the optic disc margin.51 The capillaries are unusual because of their large diameters, with lumina typically larger than 8 μm. This allows for the simultaneous passage of multiple red blood cells. The choriocapillaris is arranged in a specific lobular pattern to ensure maximum exposure of the overlying outer retina/RPE complex. Within the posterior pole region, there are more venules than arterioles, probably to allow rapid flow. Each lobule acts as an independent system with its own afferent and efferent vessels.52 The capillaries of the choriocapillaris are separate and distinct from the capillary beds of the anterior optic nerve (Fig. 15B).
Venous drainage from the choriocapillaris is mainly through the vortex veins. Minor drainage occurs through the ciliary body via the anterior ciliary veins.
Ciliary Body and Iris
The vascular supply to the anterior segment of the eye provides nutrition for the iris, ciliary muscle and ciliary processes. The production of aqueous humor is dependent upon circulation to the ciliary body. Additionally, circulation to this region also contributes to the nutrition of avascular structures such as the lens and cornea.
The anterior segment depends on two sources for its blood supply: the LPCAs and anterior ciliary arteries. Arteriolar branches from the ACAs pierce the sclera and enter the ciliary muscle just posterior to the limbus.53 Similarly, the long PCAs divide and send multiple branches into the ciliary muscle. Within the ciliary muscle, branches from both the ACAs and PCAs interconnect to form an anastomotic arterial circle, the intramuscular circle.53 Some branches from the intramuscular arterial circle supply the capillary bed of the ciliary muscle, whereas others extend anteriorly toward the root of the iris where they branch to form the major arterial circle of the iris.53 The major arterial circle is a noncontinuous arterial ring that supplies arterioles to the iris and ciliary processes.
The ciliary processes receive blood from both anterior and posterior sources. The anterior arterioles enter the ciliary process and form irregular, dilated capillaries within the margins of the process.54 Posterior arterioles originate from the major arterial circle, enter the center portions of the process and divide into irregular capillaries that are confined to its base.54
The source of blood supply to the iris is the major arterial circle. Arterioles from the major arterial circle travel anteriorly in a centripetal fashion toward the pupil. They often have a corkscrew shape because of the need for the iris stroma to expand and contract during dilation and contraction of the pupil.53 The main branches of these radial vessels form an incomplete circular arterial ring at the collarette named the minor iris circle. Branches from the minor circle extend into the papillary region to form capillary arcades. The density of these vessels is greater than expected for the nutrition of the iris, and they probably account for anterior segment thermal homeostasis and provide high oxygen content for the corneal endothelium.
Because all segments of the anterior segment vasculature, other than ciliary processes, have capillaries with endothelial cells connected by tight junctions, they do not play a role in aqueous humor production. Capillaries of the ciliary processes have fenestrated walls that allow for the passage of macromolecules into the matrix of ciliary processes, which constitutes the first step in the production of aqueous humor.55
Blood from the iris drains into the vortex system. Most ciliary vessels drain posteriorly into the choroidal and vortex systems. The remaining drainage is into the intrascleral venous plexus and the episcleral veins of the limbal region. Small veins and venules are found in the ciliary processes along with the capillaries.
The organization of circulatory system is relevant to anterior segment ischemia, which may occur following extensive extraocular muscle surgery. Because there is ample collateral circulation with the long PCAs in areas supplied by ACAs derived from arteries traveling within horizontal rectus muscles, ligation of these vessels during simple medial and lateral rectus surgery is not associated with anterior segment ischemia in otherwise healthy individuals.56 Arteries traveling within the superior and inferior rectus muscles do not enjoy such rich collateralization with the PCAs. Thus, anterior segment ischemia is sometimes observed in surgeries involving three or more muscles, with involvement of at least one vertical rectus muscle.57
Methods of Measurement
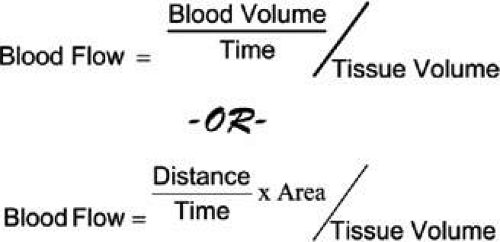
Assessment of ocular blood flow requires quantification of blood volume per unit time to a tissue bed, but a meaningful physiological interpretation of the flow level also requires knowledge of the volume of tissue maintained by that flow (Fig. 16). For example, in glaucoma, comprehensive analysis of blood flow would require numeric descriptions of the volume of blood per unit time per unit tissue volume being delivered to each of the superficial layer, prelaminar layer, and lamina cribrosa region of the optic nerve head. Unfortunately, no single instrument provides volumetric flow per unit tissue mass in real units in any ocular vascular bed, let alone performing such a feat in a comprehensive fashion throughout the retrobulbar, choroidal, and retinal vasculature. Even though a single tool for a comprehensive assessment of ocular blood flow is lacking, through the combination of a number of existing technologies, each providing data on a single vascular bed, an extensive analysis of ocular blood flow is possible.
Color Doppler Imaging
In Color Doppler Imaging (CDI), high frequency sound waves are utilized to assess ocular blood flow. Clinical CDI devices utilize frequencies between 2 to 15 million Hertz. A probe is placed on a subject’s closed eyelid and is used to transmit sound waves into the body and this same probe also records reflected waves. Sound waves are reflected by both stationary structures and moving reflective sources such as blood cells. Moving reflective sources create Doppler shifts in the frequency of reflected sound. By measuring the amount of time between the creation of the sound wave and the return of the echo, the distance to the source of the echo can be calculated (Fig. 17). Grayscale CDI images display structure by mapping the location of reflective sources. Blood flow velocity is displayed on the same image by overlaying a color coded map of Doppler shifts.
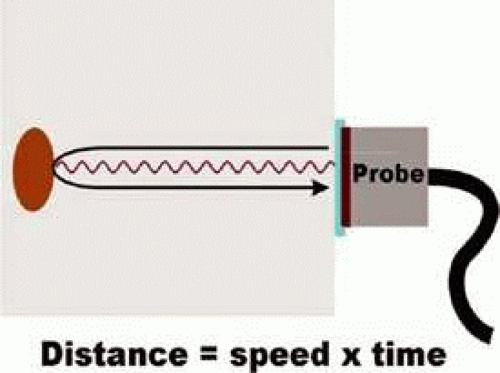 FIGURE 17. The location of reflective tissue with the body is determined by a “time of flight” calculation. |
The CDI measures blood flow velocities within the ophthalmic artery, central retinal artery and vein, nasal short posterior ciliary arteries, and the temporal short posterior ciliary arteries. Unlike other vessels normally measured by CDI (i.e., the internal carotids), the diameters of retrobulbar vessels are too small to be measured by ultrasound.58 It is, therefore, impossible to use CDI to measure flow, because the area term of the flow equation remains unknown. The volume of tissue perfused by the blood in any of these vessels is also unknown. This suggests that even if both velocity and vessel area were known, the same flow rate may have different meanings in different individuals; especially if one suffered a degenerative disease, such as glaucoma, in which tissue is lost.
Flow velocity measurements are performed in real time. The resulting measurement displays blood flow velocity throughout the cardiac cycle. Therefore, peak systolic (PSV) and end diastolic velocities (EDV) are obtained. From these data, Pourcelot’s resistive index (RI) is calculated as:59
In vivo models demonstrate that RI indicates vascular resistance to flow distal to the point of measurement.60 For example, an elevated CRA resistive index suggests increased resistance to flow in the retinal vasculature, which is perfused by the CRA. Numerous in vivo and in vitro models have been used to identify the appearance of various pathologies in the CDI velocity waveform (Fig. 18).60,61,62,63,64,65 With respect to volumetric flow, it has been demonstrated that, in the case of repeated CDI measurements, parallel changes in both PSV and EDV may be interpreted as changes in volumetric blood flow.66
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree