Purpose
To evaluate ocular biometric parameters in different subtypes of angle-closure disease in the Iranian population and compare them with normal eyes.
Design
Prospective, cross-sectional.
Methods
In this clinic-based study, 189 eyes of 154 patients consisting of 40 acute angle-closure glaucoma (AACG) eyes, 40 fellow eyes of AACG, 42 chronic angle-closure glaucoma (CACG) eyes, 40 primary angle-closure suspect (PACS) eyes, and 27 normal eyes underwent complete examination including gonioscopy, A-scan biometry, and anterior segment optical coherence tomography. Only 1 eye of CACG, PACS, and control subjects were selected. Main outcome measures included angle opening distance and trabeculo-iris space area at 500 μm from the scleral spur (AOD500, TISA-500), anterior chamber angle, lens vault, lens thickness, anterior chamber depth (ACD), and lens position.
Results
Anterior chamber angle, AOD500, TISA500, ACD, and lens position were less and lens thickness and lens vault were greater in angle-closure than open-angle eyes. ACD was less in AACG than CACG and PACS ( P < .001). It was also less in fellow eyes than PACS eyes ( P = .04). Lens vault was highest in AACG eyes, followed by fellow eyes, PACS, and CACG. It was significantly more in AACG eyes than CACG and PACS eyes ( P < .001 and P = .007, respectively). No difference was observed between AACG and fellow eyes.
Conclusions
The anterior segment was crowded in closed-angle compared to open-angle eyes. Higher lens vault may play a role in the development of an acute attack of angle closure.
Angle-closure disease is an anatomic disorder in which iris- trabecular contact impedes aqueous drainage through the trabecular meshwork. Although pupillary block and angle crowding have been proposed as the 2 main mechanisms in the pathogenesis of angle-closure disease, other factors related to the lens and ciliary body have been shown to play an important role. When aqueous humor cannot flow easily through the iris-lens channel from the posterior to anterior chamber, pupillary block is present. Therefore, the iris, lens, their positions, and their relationship to each other have important roles in angle-closure pathogenesis.
Study of biometric parameters associated with this condition helps us understand the underlying mechanisms and guides us toward more effective diagnosis and treatment. Several ocular risk factors have been identified for angle-closure disease, such as short axial length, shallow anterior chamber, and a thick and anteriorly positioned lens. This condition is more prevalent in those of Chinese ethnicity and female sex; however, these racial and sex differences cannot be explained by axial length or anterior chamber depth variations alone.
With the advent of anterior segment optical coherence tomography (AS-OCT), researchers can capture the entire anterior segment in a single image and assess angle, iris, and lens parameters more precisely. Nongpiur and associates have recently evaluated anterior chamber width, defined as the horizontal scleral spur-to-scleral spur distance in AS-OCT images, in a large sample of subjects. They found that angle-closure patients had smaller anterior chamber width than open-angle subjects.
Lens vault is an additional novel parameter that can be measured with AS-OCT and has been associated with angle closure. It is defined as the perpendicular distance between the anterior lens pole and the horizontal line joining the temporal and nasal scleral spurs. In a recent study, Nongpiur and associates measured lens vault in angle-closure and open-angle eyes, and found that lens vault and lens thickness are greater in closed-angle eyes and lens vault is significantly associated with angle-closure disease regardless of lens thickness or lens position.
Angle-closure disease is classified into different subtypes including primary angle-closure suspect (PACS), acute angle-closure glaucoma (AACG), and chronic angle-closure glaucoma (CACG). Although some ocular biometric parameters such as anterior chamber depth, lens thickness, lens position, axial length, and angle parameters have been shown to differ in various subtypes of angle-closure disease, the difference between these subtypes has not been fully elucidated. Understanding this difference may be helpful in explaining the pathogenesis of angle closure.
In this study, eyes of different subtypes of angle-closure patients including AACG, their fellow eyes, CACG, and PACS, as well as normal eyes, were evaluated. We used AS-OCT and A-scan biometry to measure ocular biometric parameters, including the lens characteristics and the new parameter lens vault.
Methods
The protocol of the study was approved by the institutional review board of Farabi Eye Hospital, Tehran, Iran. This was a prospective study in which all patients gave informed consent to participate in this research protocol.
In this cross-sectional study, 239 eyes of 154 patients were included. Study participants were consecutively recruited from the glaucoma clinic of the Farabi Eye Hospital, Tehran, Iran—a tertiary care center—as part of the Farabi Angle Closure Study. Twenty-eight eyes with poor AS-OCT image quality were excluded. Only the right eye of CACG, PACS, and control patients were selected for analysis. If the left eye was the only affected one, the left eye was chosen. A total of 189 eyes were classified into 1 of the following 5 groups: (1) acute angle-closure glaucoma (AACG; 40 eyes); (2) fellow eye of AACG (40 eyes); (3) chronic angle-closure glaucoma (CACG, 42 eyes); (4) primary angle-closure suspect (PACS, 40 eyes); and (5) control (27 eyes).
AACG was defined by the presence of the following: (1) at least 2 of the symptoms of an acute episode of intraocular pressure (IOP) rise, which are ocular pain or headache, nausea and/or vomiting, decreased vision, and rainbow-colored halos around lights; (2) IOP at presentation of at least 30 mm Hg with Goldmann applanation tonometry; (3) examination findings such as conjunctival injection, corneal epithelial edema, fixed mid-dilated pupil, and shallow anterior chamber; and (4) shallow anterior chamber and narrow angle in the other eye, defined as our second group (fellow eye). AACG attacks were broken with intravenous mannitol or oral glycerin, oral acetazolamide, and topical timolol. The AACG attack was defined as broken when IOP was less than 21 mm Hg with or without medication, and symptoms and signs of acute IOP rise had subsided. Eyes whose attack could not be broken with these medications were excluded from the study and treated with further therapy.
CACG eyes had chronically elevated IOP above 21 mm Hg (prior to treatment) along with glaucomatous optic neuropathy (such as diffuse or localized rim thinning, disc hemorrhage, a notch in the rim, or a vertical cup-to-disc ratio higher than the other eye by ≥0.2) or visual field defects typical of glaucoma (defined as pattern standard deviation with P < .05, glaucoma hemifield test result [ P < .01] outside normal limits, and 4 abnormal points with P < .05 on pattern deviation plot), shallow anterior chamber, and iridotrabecular contact in at least 3 quadrants on gonioscopy, along with variable amount of peripheral anterior synechiae (PAS). There were no history or signs of previous acute glaucoma attack in this group.
PACS eyes were classified based on the posterior trabecular meshwork not being visible in at least 3 quadrants without PAS or any evidence of glaucomatous optic nerve or visual field damage. These patients did not have any history or signs of previous AACG attack, and IOP was ≤21 mm Hg in this group without medication.
The normal controls were recruited from the comprehensive ophthalmology service. The control subjects were included if they did not have any other pathology such as open-angle glaucoma, retinal disease, corneal opacity, or high myopia. They had open angles, healthy optic nerves, normal visual fields, and IOP ≤21 mm Hg.
Individuals with history of trauma, uveitis, surgery, or any kind of laser therapy (eg, laser peripheral iridotomy [LPI]) were excluded. Also, eyes with iris or angle neovascularization, pseudoexfoliation (PEX), any kind of secondary angle closure, or any iris or corneal abnormalities were excluded. Miotic or mydriatic medications had not been used for any of the patients.
Slit-lamp examination of the anterior segment, Goldmann applanation tonometry, and gonioscopy in dark conditions (with and without indentation) was conducted for all the patients. A glaucoma specialist (S.M.) performed gonioscopy using a Zeiss-style 4-mirror goniolens (Model G-4; Volk Optical, Mentor, Ohio, USA) with a narrow 1-mm beam of light. A vertical beam was used to evaluate the superior and inferior angles, whereas a horizontal beam was used for the nasal and temporal angles. Gonioscopic grading of the angle was done according to the following system: 0 = none of the angle structures is visible; 1 = only the Schwalbe line and nonpigmented anterior trabecular meshwork are visible; 2 = posterior trabecular meshwork can also be seen; 3 = scleral spur can be detected; and 4 = all angle components including Schwalbe line, trabecular meshwork, scleral spur, and ciliary band are visible.
A-scan biometry (Echoscan, model U3300; Nidek, Tokyo, Japan) was used to measure axial length (AL), lens thickness, and anterior chamber depth (ACD). Five readings were taken for each eye. After omitting the highest and lowest values, the mean of the other 3 readings was used for analysis. The standard deviations for AL, lens thickness, and ACD were 0.04 mm, 0.03 mm, and 0.02 mm, respectively. These data were used to calculate lens position (lens position = ACD + 1/2 lens thickness), relative lens position (relative lens position = lens position / AL), and lens axial factor (lens axial factor = lens thickness / AL). Anterior segment optical coherence tomography was performed as well for anterior segment parameters.
All subjects underwent static automated white-on-white threshold perimetry (program 24-2, Swedish Interactive Threshold Algorithm standard, model 750, Humphrey Field Analyzer; Humphrey Instruments, Dublin, California, USA). Visual fields were read by a glaucoma specialist who was masked to the clinical data. A reliable field had to meet 3 criteria: false-positive rate 15% or less, false-negative rate 25% or less, and fixation loss less than 25%. If the visual field was deemed unreliable by the grader, then it was repeated.
Anterior Segment Optical Coherence Tomography
LPI was not performed for any of the patients until after the anterior segment optical coherence tomography (AS-OCT) was done. AS-OCT (Visante OCT; Carl Zeiss Meditec, Dublin, California, USA) was performed for all the patients in dark ambient lighting. Scans were centered on the pupil, and were obtained along the horizontal and vertical axes using the enhanced anterior segment single protocol. Two images were captured for each axis, and the one with higher quality was chosen for analysis. The brightness and contrast of each image were adjusted so that the scleral spurs could be detected as clearly as possible. Two experienced ophthalmologists (S.M., Z.V.) determined scleral spur location in each image. The principal investigator (S.M.) validated all the images for quality and scleral spur location.
Because the superior and inferior angle images usually do not have the optimal quality and are not reproducible, only the temporal and nasal angle parameters were used for analysis.
The following parameters were measured using the tools provided be the machine (software version 2.0.1.88). This application has been used previously in other studies to measure anterior segment and angle parameters. This semiautomatic software has algorithms defining borders and curvatures of the anterior chamber structures. However, the observer identifies the scleral spurs and anterior lens border in order for the automated software program to measure the parameters ( Figure 1 ).
- (1)
Angle opening distance at 500 μm from scleral spur (the distance between the posterior corneal surface and the anterior iris surface on a line perpendicular to the trabecular meshwork, 500 μm from the scleral spur).
- (2)
Trabeculo-iris space area at 500 μm from scleral spur (the surface area of a trapezoid with the following boundaries: anteriorly, the angle opening distance at 500 μm from scleral spur; posteriorly, a line drawn from the scleral spur perpendicular to the plane of the inner scleral wall to the iris; superiorly, the inner corneoscleral wall; and inferiorly, the iris surface).
- (3)
Anterior chamber angle (the trabecular–iris angle measured with the apex in the iris recess and the arms of the angle passing through a point on the trabecular meshwork at 500 μm from the scleral spur and the point on the iris perpendicularly opposite).
- (4)
Lens vault, which is the perpendicular distance from the anterior lens surface to the horizontal line connecting the 2 scleral spurs, measured by the chamber tool of the Visante OCT. When the anterior pole of the lens was located anterior to the scleral spur line, lens vault was positive; and when it was posterior to the line, lens vault was reported as negative.
- (5)
Iris curvature, measured by drawing a line between the most peripheral and most central points of the iris pigment epithelium and then measuring the largest perpendicular distance from this line to the iris pigment epithelium.
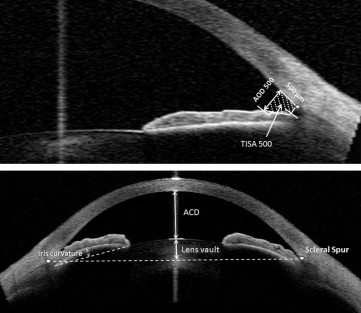
Angle opening distance and trabeculo-iris space area at 500 μm from the sclera spur, anterior chamber angle, and lens vault were generated by the software and recorded. Iris curvature was measured manually with the caliper built into the software. The mean of the nasal and temporal angle opening distance at 500 μm from scleral spur, trabeculo-iris space area at 500 μm from scleral spur, anterior chamber angle, and iris curvature are labeled as AOD 500, TISA 500, ACA, and iris curvature, respectively.
Laser Peripheral Iridotomy
To eliminate the effect of LPI on anterior segment and angle parameters, we performed gonioscopy, AS-OCT, and A-scan before LPI. However, all the angle-closure patients underwent LPI as standard medical care. Patients with prior LPI were excluded. For all angle-closure cases, the pupil was constricted with pilocarpine 2%, and LPI was performed using the ophthalmic neodymium–yttrium-aluminum-garnet (Nd:YAG) laser (Laserex Tango Nd:YAG; Ellex Medical, Adelaide, Australia) and an Abraham iridotomy contact lens.
Grading Nuclear Density
After pupillary dilation, crystalline lens nucleus density was graded according to the Lens Opacities Classification System III (LOCS III). The examiners were not masked for crystalline lens opacification grading. This grading was performed for both angle-closure and control patients. All eyes underwent dilated fundus examination including stereoscopic examination of the optic nerve head.
Statistical Analysis
Statistical analysis was performed using SPSS software version 17 (SPSS Inc, Chicago, Illinois, USA). Parametric variables were analyzed using analysis of variance (ANOVA) and post hoc Tukey tests. Adjusted lens vault for age, sex, lens thickness, and lens nucleus density was calculated and compared using analysis of covariance (ANCOVA). Analysis of nonparametric variables was done by the Kruskal-Wallis test. χ 2 testing was used for analysis of qualitative variables. Pearson and Spearman correlation coefficients were calculated between lens vault and parametric and nonparametric variables, respectively. P value of less than .05 was considered statistically significant. Assuming standard deviation (SD) = 250 μm, we needed 25 patients in each group in order to achieve an 80% power to detect a difference of 200 μm in lens vault in different subtypes of angle closure with a P value of .05.
Our sample size has an 83% power to detect a 0.3-mm difference in lens thickness (SD = 0.4 mm) between angle-closure and control eyes, and 0.2-mm difference in lens thickness (SD = 0.4 mm) between the different subtypes of angle-closure disease, using a type I error level of 0.05. Our study also had an 86% power to differentiate a 0.21-mm difference in ACD (SD = 0.24 mm) between all groups.
Results
The demographic and clinical examination data of the 5 groups are summarized in Table 1 . There was no difference in age and sex among these groups. As would be expected, IOP was significantly greater in CACG eyes than in the other groups, even with medication usage ( P < .001). These eyes also had a larger cup-to-disc ratio ( P < .001) ( Table 1 ).
| AACG | Fellow Eye | CACG | PACS | Control | P Value | |
|---|---|---|---|---|---|---|
| No. eyes | 40 | 40 | 42 | 40 | 27 | – |
| Age (y) | 61.6 ± 9.3 | 59.4 ± 9.2 | 61.1 ± 8.7 | 60.2 ± 8.1 | 62.4 ± 10.6 | .69 |
| Sex (F/M) | 31/9 | 31/9 | 22/20 | 29/11 | 19/8 | .08 |
| IOP (mm Hg) | 13.6 ± 6.8 | 12.7 ± 3.2 | 20.8 ± 7.3 | 15.8 ± 2.6 | 15.4 ± 3.3 | <.001 |
| Medication number | 1.9 ± 0.4 | 1.0 ± 0.7 | 1.7 ± 1.3 | 0.0 ± 0.0 | 0.0 ± 0.0 | <.001 |
| C/D ratio | 0.30 ± 0.13 | 0.30 ± 0.11 | 0.68 ± 0.21 | 0.29 ± 0.09 | 0.29 ± 0.07 | <.001 |
| Gonioscopy average | 0.27 ± 0.34 | 0.44 ± 0.55 | 0.53 ± 0.58 | 0.64 ± 0.57 | 3.66 ± 0.50 | <.001 |
| PAS (degrees) | 45.8 ± 79.6 | 13.1 ± 32.6 | 174.1 ± 150.1 | 0.0 ± 0.0 | 0.0 ± 0.0 | <.001 |
| Lens nucleus opacity (per LOCS III) | 2.8 ± 0.8 | 2.5 ± 0.7 | 2.8 ± 0.9 | 2.6 ± 0.9 | 3.2 ± 1.1 | .01 |
Intraclass correlation coefficient of variables generated by the AS-OCT software ranged from 93.5% to 99.1% for intravisit and 94.8% to 99.1% for intervisit measurements.
Angle Status
CACG eyes had 174.1 ± 150.1 degrees of PAS, which was significantly more than the other 4 groups ( P < .001). Gonioscopically, the anterior chamber angle was narrowest in the AACG eyes, followed by fellow eyes, CACG, PACS, and control; the only significant difference among the angle-closure diagnoses was between AACG and PACS eyes ( P = .01). The same pattern was also observed for ACA, AOD 500, and TISA 500. ACA and TISA 500 were significantly less in the AACG, fellow eye, and CACG groups than the PACS group ( Table 2 ). AOD 500 was less in AACG and fellow eyes than in PACS eyes ( P = .002 and P = .021, respectively) ( Table 2 ). Compared with the control eyes, all angle-closure groups had less AOD 500, TISA 500, and ACA ( P < .001) ( Table 2 ).
| AACG | Fellow Eye | CACG | PACS | Control | P Value | |
|---|---|---|---|---|---|---|
| N_AOD 500 (mm) | 0.027 ± 0.039 | 0.044 ± 0.042 | 0.065 ± 0.065 | 0.111 ± 0.073 | 0.398 ± 0.200 | <.001 |
| N_TISA 500 (mm 2 ) | 0.015 ± 0.022 | 0.022 ± 0.020 | 0.025 ± 0.025 | 0.048 ± 0.029 | 0.148 ± 0.075 | <.001 |
| N_ACA (degree) | 1.82 ± 3.32 | 3.19 ± 3.64 | 4.80 ± 5.43 | 9.28 ± 6.33 | 29.52 ± 11.38 | <.001 |
| T_AOD 500 (mm) | 0.028 ± 0.038 | 0.043 ± 0.041 | 0.067 ± 0.061 | 0.105 ± 0.075 | 0.410 ± 0.262 | <.001 |
| T_TISA 500 (mm 2 ) | 0.014 ± 0.020 | 0.023 ± 0.022 | 0.029 ± 0.025 | 0.054 ± 0.033 | 0.159 ± 0.107 | <.001 |
| T_ACA (degree) | 2.01 ± 3.14 | 3.19 ± 3.74 | 5.08 ± 5.11 | 8.64 ± 6.24 | 29.54 ± 12.11 | <.001 |
| AOD 500 (mm) | 0.028 ± 0.032 | 0.044 ± 0.030 | 0.066 ± 0.054 | 0.108 ± 0.059 | 0.404 ± 0.221 | <.001 |
| TISA 500 (mm 2 ) | 0.014 ± 0.018 | 0.022 ± 0.016 | 0.027 ± 0.021 | 0.051 ± 0.025 | 0.154 ± 0.087 | <.001 |
| ACA (degree) | 1.92 ± 2.67 | 3.19 ± 2.73 | 4.94 ± 4.74 | 8.96 ± 5.08 | 29.53 ± 11.15 | <.001 |
| Lens thickness (mm) | 5.06 ± 0.46 | 4.96 ± 0.32 | 4.85 ± 0.39 | 4.92 ± 0.30 | 4.16 ± 0.49 | <.001 |
| Anterior chamber depth (mm) | 2.26 ± 0.22 | 2.36 ± 0.21 | 2.50 ± 0.24 | 2.53 ± 0.28 | 3.15 ± 0.33 | <.001 |
| Axial length (mm) | 21.84 ± 1.17 | 21.69 ± 1.13 | 22.24 ± 0.80 | 21.97 ± 0.73 | 22.46 ± 4.35 | .26 |
| Lens vault (μm) | 1067.95 ± 252.54 | 977.00 ± 192.32 | 847.14 ± 189.74 | 890.25 ± 221.30 | 266.67 ± 308.38 | <.001 |
| Lens position (mm) | 4.80 ± 0.23 | 4.85 ± 0.23 | 4.93 ± 0.23 | 4.99 ± 0.27 | 5.23 ± 0.34 | <.001 |
| Relative lens position | 0.22 ± 0.02 | 0.22 ± 0.02 | 0.22 ± 0.01 | 0.23 ± 0.01 | 0.32 ± 0.47 | .13 |
| Lens axial factor | 0.23 ± 0.02 | 0.23 ± 0.02 | 0.22 ± 0.02 | 0.22 ± 0.01 | 0.25 ± 0.34 | .87 |
Anterior Chamber Depth
The eyes with a history of AACG attack had similar ACD as their fellow eyes ( P = .46), but had shallower anterior chambers than CACG and PACS eyes ( P < .001 for both) ( Table 3 ). ACD was less in fellow eyes than in PACS eyes ( P = .04). As expected, the anterior chamber was deeper in control eyes than all angle-closure types ( P < .001). The result was the same after adjustment for age, sex, lens thickness, AL, and lens vault ( P = .02 and P = .002 for AACG vs CACG and PACS, respectively; P = .01 for fellow eyes vs PACS; and P < .001 for control vs all angle-closure groups) ( Table 4 ).
| AACG | Fellow Eye | CACG | PACS | |
|---|---|---|---|---|
| Lens vault ( P value) | ||||
| Fellow | .40 | – | – | – |
| CACG | <.001 | .11 | – | – |
| PACS | .007 | .45 | .94 | – |
| Control | <.001 | <.001 | <.001 | <.001 |
| Lens thickness ( P value) | ||||
| Fellow | .79 | – | – | – |
| CACG | .16 | .79 | – | – |
| PACS | .51 | .99 | .95 | – |
| Control | <.001 | <.001 | <.001 | <.001 |
| Lens nucleus opacity (LOCS III) ( P value) | ||||
| Fellow | .46 | – | – | – |
| CACG | .95 | .89 | – | – |
| PACS | .73 | .99 | .99 | – |
| Control | .36 | .01 | .10 | .03 |
| Anterior chamber depth ( P value) | ||||
| Fellow | .41 | – | – | – |
| CACG | <.001 | .11 | – | – |
| PACS | <.001 | .04 | .95 | – |
| Control | <.001 | <.001 | <.001 | <.001 |
| Lens position ( P value) | ||||
| Fellow | .90 | – | – | – |
| CACG | .13 | .59 | – | – |
| PACS | .01 | .15 | .90 | – |
| Control | <.001 | <.001 | <.001 | .004 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


