Purpose
To determine patterns of diffusion of diagnostic tests and therapeutic interventions in the United States through 2010 for patients with newly diagnosed exudative macular degeneration (AMD).
Design
Retrospective longitudinal cohort analysis.
Methods
setting and patient population : A total of 23 941 Medicare beneficiaries with exudative AMD newly diagnosed during 1992-2009. observation procedures : Current Procedural Technology (CPT-4) billing codes were used to identify use of diagnostic tests (optical coherence tomography, fluorescein angiography, and fundus photography) and therapeutic interventions (argon laser photocoagulation, photodynamic therapy, intravitreal corticosteroids, and anti–vascular endothelial growth factor [VEGF] agents) used by these beneficiaries during the first year following diagnosis. main outcome measures : Rates of use of study diagnostic and therapeutic procedures.
Results
Diffusion was rapid for each successive new diagnostic and treatment modality, with use of newer procedures quickly replacing existing ones. The number of beneficiaries treated with anti-VEGF agents for exudative AMD was considerably greater than for prior innovations, rising from use in 4.0% of beneficiaries in 2004-05 to 62.7% in 2009-10. In each year from first diagnosis years 2006-2009 and in different practice settings, use of bevacizumab exceeded that of ranibizumab (60%-78% vs 33%-47%, respectively). Rates of diffusion of the various therapies were relatively similar in communities throughout the United States irrespective of presence of a major teaching hospital in the vicinity.
Conclusions
Newer, more effective therapeutic interventions for exudative AMD diffused rapidly throughout the United States, quickly replacing older, less effective interventions. Although improving patient outcomes, rapid diffusion raises important public policy issues for Medicare and other payers to consider.
Age-related macular degeneration (AMD) is a common cause of legal blindness among older Americans. Exudative AMD is far less prevalent than nonexudative AMD but carries a worse visual prognosis. Although the therapeutic options for managing patients with exudative AMD were limited until recently, new therapies for this sight-threatening condition have emerged, improving the prognosis for patients with this condition.
Beginning in the 1980s, argon laser photocoagulation therapy was the most common, and essentially only, treatment option for exudative AMD. This treatment, although effective for extrafoveal lesions, was associated with iatrogenic vision loss in certain patients. With the approval of photodynamic therapy (PDT) by the US Food and Drug Administration (FDA) in 2000, ophthalmologists had a safer option for treating subfoveal choroidal neovascularization with reduced risk of vision loss from macular scarring; yet the cost-effectiveness of PDT was soon found to be marginal at best. In the mid-2000s, intravitreal corticosteroids, which can inhibit progression of choroidal neovascularization, emerged as another treatment alternative; however, the treatment’s side effects included development or worsening of cataract and glaucoma, and its effectiveness was questionable. Most recently, use of vascular endothelial growth factor (VEGF) inhibitors (anti-VEGFs)—typically involving intravitreal injection in an office setting under local anesthesia—became a viable treatment alternative for exudative AMD. The first of these agents, pegaptanib (Macugen), which reduces visual acuity loss from exudative AMD but produces limited improvement in best-corrected visual acuity (BCVA), received FDA approval in December 2004, followed by ranibizumab (Lucentis) in June 2006. Around the same time, bevacizumab (Avastin), an anti-VEGF with an effectiveness similar to ranibizumab but at much lower cost, became a popular, although off-label, treatment option. Approximately one-third of patients receiving ranibizumab or bevacizumab recover significant BCVA, and ranibizumab therapy is generally more effective than PDT alone. Serious adverse events associated with anti-VEGF agents, although rare, include endophthalmitis, retinal detachment, and intraocular hemorrhage. Furthermore, frequent reinjections are commonly required.
These newer technologies, although not cures for the disease, do improve the clinical course for many patients, yet at a substantial cost to Medicare. This relatively high expense of the newer therapies during a time of overall budgetary constraints raises 2 issues: (1) Should the effectiveness of new therapeutic products be tested in the general population, instead of trials involving a limited population, before coverage is approved by insurance in general and Medicare in particular? (2) To what extent should coverage be limited to those therapies with demonstrated superiority in cost-effectiveness comparisons? To date, the policy choice has been to cover many promising technologies before outcomes and complications are well documented. Coverage of technologies for treating exudative AMD is a case in point.
While it is widely appreciated that there have been technological advances in diagnostic testing and therapeutic interventions for exudative AMD in recent years, what is not yet well documented is how diffusion of new technology has affected use of older technologies. The primary aim of this analysis is to quantify changes in utilization of these different technologies over time among Medicare beneficiaries with newly diagnosed exudative AMD. This study also seeks to better understand whether rates of adoption of technology vary by location relative to academic health centers.
Methods
The Duke University Institutional Review Board approved this study prospectively. The IRB approved this study as a secondary analysis of data collected by someone else.
We used a 5% sample of the Medicare claims with dates of service from 1991 to 2010 to identify a national random sample of Medicare beneficiaries whose first diagnosis of exudative AMD (International Classification of Diseases, Ninth Revision [ICD-9-CM] code 362.52) appeared in these claims during 1992-2009 (N = 106,481). Beneficiaries with a diagnosis of diabetes mellitus (ICD-9-CM 250.xx), vein occlusions (ICD-9-CM 362.3x), cystoid macular edema (ICD-9-CM 362.53), histoplasmosis retinitis unspecified (ICD-9-CM 115.92), chorioretinitis due to toxoplasmosis (ICD-9-CM 130.2), progressive high myopia (ICD-9-CM 360.21), retinal neovascularization not otherwise specified (ICD-9-CM 362.16), retinal edema (ICD-9-CM 362.83), angioid streaks of choroid (ICD-9-CM 363.43), rubeosis iridis (ICD-9-CM 364.42), and glaucoma associated with vascular disorders (ICD-9-CM 365.63) were excluded (N = 62,561) to reduce the possibility of misattributing the use of diagnostic and therapeutic interventions for other retinal conditions besides exudative AMD. To ensure that the first diagnosis of exudative AMD in the claims was the beneficiaries’ first actual diagnosis, beneficiaries with diagnoses in 1991 were excluded (N = 2057). We also excluded beneficiaries who enrolled in a Medicare Advantage HMO (N = 4150), moved outside the United States (N = 131), died (N = 3410), or ceased to be listed in the Medicare enrollment file for another reason (N = 4121) within 1 year of the first exudative AMD diagnosis. Finally, we excluded beneficiaries who did not have a second diagnosis of exudative AMD in the 6 months following the first diagnosis (N = 6110), yielding a total sample of 23 941 enrollees with newly diagnosed exudative AMD. The Duke University Institutional Review Board determined that studies based on these data are exempt from requiring approval since the data are de-identified to the investigators.
We examined diffusion patterns of 4 therapeutic interventions for exudative AMD: argon laser photocoagulation (Current Procedure Terminology [CPT]-4 codes 67210 or 67220); PDT (CPT-4 code 67221 or in the year 2000, code 67299); corticosteroids (CPT-4 codes J1870, J1880, J3300, or J3301 administered by intravitreal injection CPT-4 code 67028 on the same date); and intravitreal anti-VEGF agent injections (CPT-4 codes J2503, J3490, J3590, J9035, Q2024, C9233, or C9399 administered by intravitreal injection CPT-4 code 67028). We required each claim for one of these therapies to also list a diagnosis code of exudative AMD. We measured diffusion in terms of (1) percent of sample persons receiving treatment from the date of first diagnosis of exudative AMD through the following calendar year; and (2) percent of sample persons receiving particular therapeutic procedures in calendar year 2010 by year of first claim with an exudative AMD diagnosis: 2009, 2008, 2007, and 2006.
To assess relationships between use of diagnostic technologies, number of beneficiaries diagnosed with exudative AMD, and use of therapeutic technologies, we examined trends in the use of 3 diagnostic technologies within 7 days of the first exudative AMD diagnosis (including the date of first diagnosis): optical coherence tomography (OCT; CPT-4 code 92135); intravenous fluorescein angiography (IVFA; CPT-4 codes 92230 or 92235), and fundus photography (CPT-4 code 92250).We excluded beneficiaries with any glaucoma diagnosis (ICD-9-CM 365.xx) in our analysis of trends in the use of diagnostic tests since some of these tests could be used to also evaluate glaucoma.
Many technologies are first adopted by major teaching hospitals and later diffuse to the community providers. We sought to ascertain whether this pattern also applies to therapies for exudative AMD. To determine relationships between trends in the fraction of beneficiaries who received each of the 4 therapies after first being diagnosed with exudative AMD and proximity to a major teaching hospital, we stratified beneficiaries into 3 groups: (1) those who lived in a county with a major teaching hospital; (2) those who lived in the Hospital Referral Region containing a major teaching hospital but in a county other than where the teaching hospital was located ; and (3) those living in Hospital Referral Regions containing no major teaching hospitals. Locations where care was received were based on zip codes of the place of residence of each Medicare beneficiary. Hospital Referral Regions are designed to be health care markets for tertiary medical care. Tertiary care facilities typically offer a full range of specialty and subspecialty services and have residency programs in these fields. This geographic unit is widely used in health services research and health policy. There are 306 Hospital Referral Regions in the United States.
We defined “major teaching hospital” as follows. First, we used addresses of members of the American Association of Medical Colleges (AAMC) as of 2010 to designate counties with and without AAMC members. Second, we deleted AAMC members that did not have an accredited ophthalmology residency training program. Of the 103 Hospital Referral Regions with AAMC members, 27 did not have an accredited ophthalmology residency program, leaving 76 Hospital Referral Regions with a major teaching hospital as we defined it. The remaining 230 Hospital Referral Regions did not have a major teaching hospital.
Comparisons of the proportion of enrollees who received the different technologies from one year to the next were performed using t tests, as were t tests performed to determine whether differences exist in the use of the different diagnostic tests and therapies among enrollees who received care in practices located in each of the 3 settings. Since the use of t tests was only for assessing whether or not there is a statistical difference between a proportion receiving a diagnostic test or intervention in one year from the proportion for the next year and not to assess for causal relations among variables, there was no need to adjust for multiple comparisons.
Results
Trends in Number of New Diagnoses of Exudative Macular Degeneration
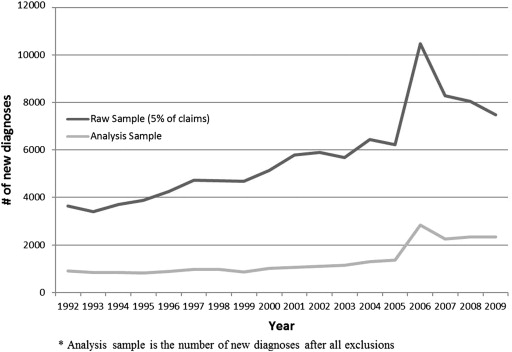
The number of beneficiaries with newly diagnosed exudative AMD in the Medicare 5% sample in a given year rose from 3641 in the baseline year, 1992, to 7490 in the year 2009, a 106% increase ( Figure ). The peak number of newly diagnosed beneficiaries with exudative AMD was in the year 2006 (N = 10 473 from 5% sample of claims), about a year after the introduction of anti-VEGF therapy. After exclusions, the analysis sample varied from 908 in 1992 to 2338 in 2009. On average, newly diagnosed beneficiaries in our analysis sample were 79.5 years old (standard deviation: 7.8); 30.1% were male; 96.5% were white, 1.1% African American, 0.6% Asian, 1.3% other race, and 0.5% Hispanic ethnicity. The demographic characteristics of persons in the study sample remained relatively constant over time (data not shown). All of the study therapeutic procedures were more likely to be administered soon after diagnosis than subsequently. Nearly forty-seven percent (46.9%) of beneficiaries who were newly diagnosed with exudative AMD in 2009 received anti-VEGF therapies in 2010, whereas only 36.8% of those newly diagnosed in 2008 and only 32.8% in 2007 received any anti-VEGF treatment in 2010.
Diffusion of Ancillary Tests to Diagnose Exudative Macular Degeneration
In 1992 (before the existence of OCT) IVFA and fundus photography were performed on the day of or just prior to the date of diagnosis for 53.0% and 43.9% of new cases of exudative AMD, respectively ( Table 1 ). By 2002, the proportions of newly diagnosed exudative AMD patients receiving IVFA and fundus photography were 62.5% and 55.1%, respectively, and only 0.7% of these patients underwent OCT testing. From 2002 to 2008 there was a rapid diffusion of OCT utilization and by 2009, 51.6% of those with incident exudative AMD underwent OCT testing. During the period of rapid diffusion of OCT, proportions of individuals with newly diagnosed exudative AMD who underwent IVFA declined somewhat. For this analysis, use of the 3 diagnostic technologies was not mutually exclusive; thus, some patients may have undergone more than 1 of these tests at the time of diagnosis.
| Year of First Diagnosis | Intravenous Fluorescein Angiography | Fundus Photography | Optical Coherence Tomography |
|---|---|---|---|
| 1992 | 53.0 | 43.9 | 0 |
| 1993 | 54.8 | 47.8 | 0 |
| 1994 | 56.1 | 51.0 | 0 |
| 1995 | 58.0 | 47.7 | 0 |
| 1996 | 55.7 | 51.1 | 0 |
| 1997 | 56.0 | 51.9 | 0 |
| 1998 | 57.7 | 54.6 | 0 |
| 1999 | 64.7 b | 58.1 | 0 |
| 2000 | 63.4 | 55.0 | 0 |
| 2001 | 63.1 | 54.4 | 0 |
| 2002 | 62.5 | 55.1 | 0.7 b |
| 2003 | 67.9 b | 62.6 c | 2.7 c |
| 2004 | 63.2 b | 58.5 | 7.2 c |
| 2005 | 65.5 | 54.4 | 15.1 c |
| 2006 | 52.8 c | 45.2 c | 24.9 c |
| 2007 | 55.7 | 40.9 c | 39.7 c |
| 2008 | 52.0 b | 37.7 | 44.2 c |
| 2009 | 48.5 b | 32.1 c | 51.6 c |
a Tests conducted within a week of date of first diagnosis.
b Compared with previous year ( t test); P < .05.
Diffusion of Therapies for Exudative Macular Degeneration
Argon laser photocoagulation was already a mature technology at baseline. The proportion of beneficiaries with newly diagnosed exudative AMD receiving argon laser photocoagulation peaked in 1994 and 1999 at 22.2% ( Table 2 ). Proportions of patients undergoing argon laser photocoagulation remained stable from 1992 to 2000 but soon thereafter rapidly declined to a level of only 2.4% by the year 2009.
| Year of First Diagnosis | Argon Laser Photocoagulation Therapy | Photodynamic Therapy | Steroids | Anti-VEGF |
|---|---|---|---|---|
| 1992 | 17.6 | 0 | 0 | 0 |
| 1993 | 20.0 | 0 | 0 | 0 |
| 1994 | 22.2 | 0 | 0 | 0 |
| 1995 | 20.7 | 0 | 0 | 0 |
| 1996 | 18.1 | 0 | 0 | 0 |
| 1997 | 17.9 | 0 | 0 | 0 |
| 1998 | 18.1 | 0 | 0 | 0 |
| 1999 | 22.2 a | 1.4 b | 0 | 0 |
| 2000 | 21.0 | 16.2 b | 0 | 0 |
| 2001 | 11.2 b | 25.9 b | 0.1 | 0 |
| 2002 | 11.7 | 28.1 | 0.8 a | 0 |
| 2003 | 11.0 | 29.6 | 3.0 b | 0 |
| 2004 | 9.5 | 37.5 b | 7.7 b | 4.0 b |
| 2005 | 7.7 | 29.5 b | 8.0 | 26.0 b |
| 2006 | 3.9 b | 10.6 b | 3.1 b | 47.1 b |
| 2007 | 2.8 a | 5.7 b | 1.2 b | 58.4 b |
| 2008 | 1.8 a | 4.8 | 0.7 | 60.3 |
| 2009 | 2.4 | 2.7 b | 1.0 | 62.7 |
a Compared with previous year ( t test); P < .05.
First introduced into the mainstream in 2000, PDT diffused rapidly, with the proportion of patients with newly diagnosed exudative AMD undergoing PDT increasing 26-fold, from 1.4% in 1999 (patients who received their diagnosis in 1999) to 37.5% in 2004. From 2005 to 2009, there was a rapid decline in PDT use, down to 2.7%. Use of intravitreal corticosteroids began in 2001. The proportion of patients with newly diagnosed exudative AMD who received intravitreal corticosteroids was low (0.1%), increased to 8.0% in 2005, and then declined to only 1.0% in 2009.
Anti-VEGF therapies were introduced in 2004. That year, 4.0% of newly diagnosed patients with exudative AMD received these agents. By 2005, the proportion undergoing anti-VEGF injections increased 6-fold from the prior year, to 26.0%. The rapid diffusion of this therapy continued from 2005 to 2009, more than doubling to 62.7%. In 2009, use of anti-VEGFs was 10 times greater than all 3 of the other interventions combined for exudative AMD.
Proximity to a Major Teaching Hospital and Diffusion
Both increases and decreases in use of different therapeutic procedures were quite similar irrespective of whether or not the beneficiary with exudative AMD lived in a county with a major teaching hospital, in a Hospital Referral Region with a major teaching hospital but not in the county in which the practice was located, or in a Hospital Referral Region without a major teaching hospital ( Table 3 ). There were higher utilization levels of anti-VEGFs each year for patients receiving care in practices located away from major teaching hospitals relative to those residing in communities close to major teaching facilities in 2005, 2006, 2008, and 2009.
| Year of First Diagnosis | Argon Laser Photocoagulation Therapy | Photodynamic Therapy | Steroids | Anti-VEGF | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MTH County | MTH HRR | Other HRR | MTH County | MTH HRR | Other HRR | MTH County | MTH HRR | Other HRR | MTH County | MTH HRR | Other HRR | |
| 1992 | 19.3 | 16.3 | 17.6 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 1993 | 16.7 | 18.5 | 22.5 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 1994 | 15.8 b | 19.7 | 25.9 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 1995 | 19.3 | 18.8 | 22.5 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 1996 | 18.2 | 14.9 | 20.0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 1997 | 17.8 | 18.4 | 17.6 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 1998 | 14.4 | 18.1 | 19.4 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 1999 | 15.0 b | 24.4 | 23.9 | 1.7 | 0.8 | 1.6 | 0 | 0 | 0 | 0 | 0 | 0 |
| 2000 | 22.0 | 20.7 | 20.8 | 12.9 | 17.3 | 16.6 | 0 | 0 | 0 | 0 | 0 | 0 |
| 2001 | 11.3 | 12.1 | 10.6 | 26.8 | 22.5 | 27.4 | 0 | 0 | 0.2 | 0 | 0 | 0 |
| 2002 | 7.9 a | 10.5 | 13.7 | 29.6 | 24.0 | 29.6 | 0.5 | 1.0 | 0.9 | 0 | 0 | 0 |
| 2003 | 6.4 a | 12.7 | 11.7 | 26.5 | 30.9 | 30.1 | 2.7 | 1.2 b | 4.0 | 0 | 0 | 0 |
| 2004 | 8.0 | 9.1 | 10.2 | 31.9 | 38.9 | 38.7 | 6.7 | 8.3 | 7.8 | 3.8 | 2.1 b | 5.1 |
| 2005 | 5.2 | 9.4 | 7.8 | 26.5 | 31.9 | 29.5 | 6.2 | 8.2 | 8.7 | 22.0 a | 25.0 | 28.3 |
| 2006 | 2.5 b | 2.8 b | 5.1 | 11.5 | 10.3 | 10.4 | 3.5 | 2.0 a | 3.6 | 40.2 b | 45.0 b | 50.9 |
| 2007 | 2.4 | 2.7 | 3.0 | 5.2 | 5.5 | 5.9 | 1.5 | 0.9 | 1.3 | 55.3 | 58.2 | 59.8 |
| 2008 | 0.9 a | 2.1 | 2.1 | 3.7 a | 3.5 b | 6.1 | 0.6 | 0.6 | 0.8 | 55.6 a | 60.8 | 61.9 |
| 2009 | 3.1 | 2.6 | 2.1 | 2.9 | 2.2 | 3.1 | 1.0 | 0.5 | 1.4 | 55.6 b | 62.6 | 65.3 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


