Nutritional and Environmental Influences on Risk for Cataract
Allen Taylor
The number of associations between nutriture and eye lens cataract has burgeoned in the past decade, inspired in part by early studies regarding antioxidant properties of nutrients.1 Such studies include laboratory, clinical, and epidemiologic investigations, as well as human intervention trials. Because this volume has as its focus relationships between nutritional and environmental influences on risk for age-related eye diseases, data regarding associations between nutriture and eye health are given the most thorough treatment. For a review of data regarding animal or cell-free studies, readers can refer to other recent summaries and the rich body of pioneering work which is, of necessity, given limited coverage here.2,3,4,5,6,7,8,9,10,11
CATARACT AS A PUBLIC HEALTH ISSUE
Cataract is one of the major causes of blindness throughout the world.12,13,14 In the United States, the prevalence of visually significant cataract increases from approximately 5% at age 65 to around 50% for persons older than 75 years of age.15,16,17 In the United States and much of the developed world, cataract surgery, albeit costly, is readily available and routinely successful in restoring sight. In less-developed countries, such as India,18 China,19 and Kenya,20 cataracts are more common and develop earlier in life than in more-developed countries. For example, for persons age 60 and older, cataract with low vision or aphakia (i.e., absence of the lens, which usually is the result of cataract extraction) is approximately five times more common in India than in the United States.17,18 The impact of cataract on impaired vision is much greater in less-developed countries, where more than 90% of the cases of blindness and visual impairment are found14,21,22,23,24,25 and where there is a dearth of ophthalmologists to perform lens extractions.
Given both the extent of disability caused by age-related cataract and its costs, $5 to $6 billion per year26 (Congressional Testimony of S.J. Ryan, May 5, 1993) in the United States, it is urgent that we elucidate causes of cataract and identify strategies to slow the development of this disorder. It is estimated that a delay in cataract formation of approximately 10 years would reduce the prevalence of visually disabling cataract by approximately 45%.12 Such a delay would enhance the quality of life for much of the world’s older population and substantially reduce the economic burden due to cataract-related disability and cataract surgery. It is such data that provide the impetus for this research.
AGE-RELATED CHANGES IN LENS FUNCTION
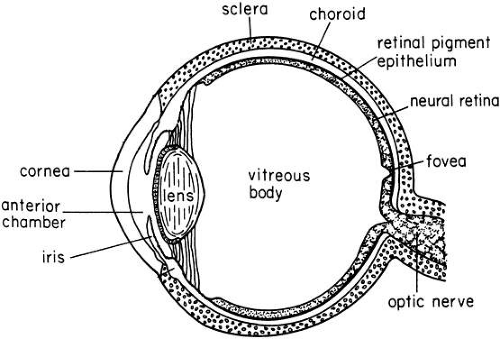
The primary function of the eye lens is to collect and focus light on the retina (Figs. 1 and 2a). To do so, it must remain clear throughout life. The lens is located posterior to the cornea and iris and receives nutriture from the aqueous humor. Although the clarity of the lens frequently is inter-preted as indicative of an absence of structure, the lens is exquisitely organized. A single layer of epithelial cells is found directly under the anterior surface of the collagenous membrane in which it is encapsulated (see Fig. 2b). The epithelial cells at the germinative region divide, migrate posteriorly, and differentiate into lens fibers. As their primary gene products, the fibers elaborate the predominant proteins of the lens, called crystallins. They also lose their organelles. New cells are formed throughout life, but older cells usually are not lost. Instead, they are compressed into the center or nucleus of the lens. There is a coincident dehydration of the proteins and of the lens itself. Consequently, protein concentrations rise to hundreds of milligrams per milliliter.27 Together with other age-related modifications of the protein (noted below) and other constituents, these changes result in a less-flexible lens with limited accommodative capability.
In addition, as the lens ages, the proteins are photo-oxidatively damaged, aggregate, and accumulate in lens opacities. Dysfunction of the lens due to opacification is called cataract. The term age-related cataract is used to distinguish lens opacification associated with old age from opacification associated with other causes, such as congenital and metabolic disorders or trauma.28
CLINICAL FEATURES IN CATARACT
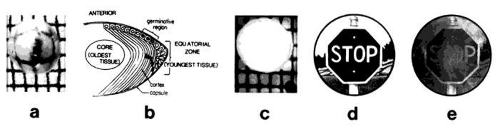
There are several systems for evaluating and grading cataracts. Most of these use an assessment of extent, or density, and location of the opacity.29 Usually evaluated are opacities in the posterior subcapsular, nuclear, cortical, and multiple (mixed) locations (see Fig. 2b). However, it is not established that cataract at each location has completely different etiology. Coloration or brunescence also is quantified, since these diminish visual function (see Figs. 2c-e).30,31
HIGH-ENERGY RADIATION, OXIDATION, SMOKING, AND FAILURE OF PRIMARY AND SECONDARY DEFENSE SYSTEMS
Only a brief introduction to some of these topics is offered here. The solid mass of the lens is about 98% protein. These proteins undergo minimal turnover as the lens ages. Accordingly, on aging, they are subject to the chronic stresses of exposure to light or other high-energy radiation and oxygen. Several, if not all, of these insults cause oxidative damage to lens constituents, and this damage is thought to be causally related to cataractogenesis. A schematic summary of insults and protective species, along with a proposal of their interactions, is indicated in Figure 3.
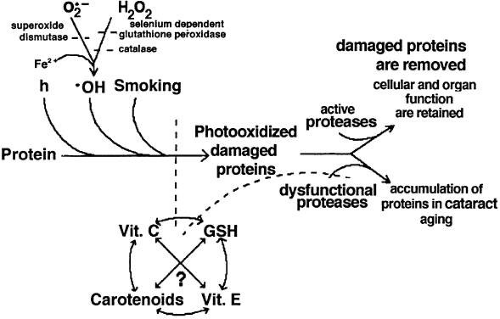 Fig. 3. Proposed interaction between lens proteins, oxidants, light, smoking, antioxidants, antioxidant enzymes, and proteases. Lens proteins are extremely long lived. Lens proteins are subject to alteration by light and various forms of oxygen. They are protected indirectly by antioxidant enzymes: superoxide dismutase, catalase, and glutathione reductase/peroxidase. These enzymes convert active oxygen to less-damaging species. Direct protection is offered by antioxidants: glutathione (GSH), ascorbate (vita-min C), tocopherol (vitamin E), and carotinoids. Levels of reduced and oxidized forms of some, but perhaps not all (?), of these molecules are determined by interaction among the three and with the environment.166,167,168,169,170,171 Proteins that are damaged may accumulate and precipitate in cataract if there is insufficient proteolytic capability. When the proteolytic capability is sufficient, obsolete and damaged proteins may be reduced to their constituent amino acids. On aging, some of the eye antioxidant supplies are diminished, antioxidant enzymes inactivated, and proteases less active. This appears to be related to the accumulation, aggregation, and eventual precipitation in cataractous opacities of damaged proteins. (Taylor A, Dorey CK, Nowell T Jr. Oxidative stress and ascorbate in relation to risk for cataract and age-related maculopathy. In Packer L, Fuchs J [eds]: Vitamin C in Health and Disease. New York: Marcel Dekker, 1997.) |
LIGHT EXPOSURE AS A RISK FACTOR FOR CATARACT
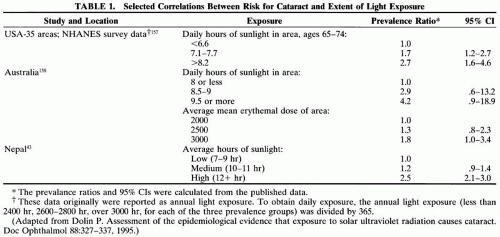
Various epidemiologic studies show associations between elevated risk of various forms of cataract and exposure to higher intensities of incident or reflected ultraviolet light or both (Table 1).15,32,33,34,35,36,37,38,39,40 Greater light exposure was (weakly) associated with an increased risk for cortical opacity in Chesapeake Bay watermen32 and in men (but not women) in Wisconsin.36 Light-related risk for cataract alsowas increased among Italians but not among resi-dents of Massachusetts.37 Risk for posterior subcapsular cataract was weakly related to light exposure in Chesapeake Bay watermen and (nonsignificantly) in residents of Wisconsin. Other studies (Massachusetts and Italy) did not find associations between posterior subcapsular cataract risk and light exposure. Nuclear cataract appears unrelated to risk for cataract in most studies.
Geographic data provide some support for purported relationships between light exposure and cataract risk.41 Persons living closer to the equator42 and living at higher elevations appear to have anelevated risk of various forms of cataract.35,38,39,40,41,43 Indeed, one of the strongest predictors of cataract surgery likelihood in a Medicare beneficiary is a person’s latitude of residence.40 Although not a uniform observation,2,44,45 these epidemiologic data have been corroborated or anticipated by exposure of squirrels to ultraviolet light in vivo46 and in many experiments in vitro6,7,8,42,47,48,49,50,51,52 and references cited within. As an aggregate, the latter references indicate that exposure of lens constituents to various wavelengths of light results in alterations that are quite similar to those found in cataract.
HIGH-ENERGY RADIATION AS A RISK FACTOR FOR CATARACT
Cataractogenesis is also clearly related to exposure to high-energy radiation. Taylor and associates showed a dose-response relationship betweenx-irradiation and risk for cataract in rats.53 In a study with 99 patients, the 89 who received whole-body irradiation (10 g) had cataract develop in less than 4 years.54 The 10 patients who were treated for aplastic anemia and did not receive radiation treatment did not show evidence of cataractogenesis.
EXPOSURE TO HIGH LEVELS OF OXYGEN AS A RISK FACTOR FOR CATARACT
Perhaps the clearest causal association between oxidative stress and cataract comes from experiences involving elevated levels of oxygen. Nuclear cataract was observed in patients treated with hyperbaric oxygen therapy,55 and markedly elevated levels of mature cataract were observed in mice that survived exposure to 100% oxygen twice weekly for 3 hours.56 A decline in glutathione (GSH) and an increase in glutathione disulfide (oxidative changes normally related to aging or cataract) also were noted. A higher incidence of cataract was noted in lenses exposed to hyperbaric oxygen in vitro,57 and Giblin also noted very early stages of cataract in guinea pigs exposed to hyperbaric oxygen.58 However, there was difficulty in repeating these results (Taylor and coworkers, unpublished data). Oxidative damage to membrane lipids in fiber cells also is associated with lens opacities.59
SMOKING AS A RISK FACTOR FOR CATARACT
Smoking and tobacco chewing appear to induce oxidative stress and have been associated with both diminished levels of antioxidants, ascorbate, and carotenoids60,61,62,63,64,65 and with enhanced cataract at a younger age.66,67,68,69 Of interest are the following recent observations:
CELLULAR ANTIOXIDANTS AS PRIMARY DEFENSES AGAINST LENS DAMAGE
Protection against photo-oxidative insult can be conceived as due to two interrelated processes. Primary defenses offer protection of proteins and other constituents by lens antioxidants and antioxidant enzymes (see Fig. 3). Secondary defenses include proteolytic and repair processes, which degrade and eliminate damaged proteins and other biomolecules in a timely fashion.71
The major aqueous antioxidants in the lens are ascorbate72 and GSH.3,73,74,75 Both are present in the lens at millimolar concentrations.76,77,78 Ascorbate is probably the most effective, least toxic antioxidant identified in mammalian systems.79,80 Interest in the function of ascorbate in the lens was prompted by teleologic arguments, which suggested age-related compromises in ascorbate and compromises in lens function might be related. Thus, the following observations were noted:
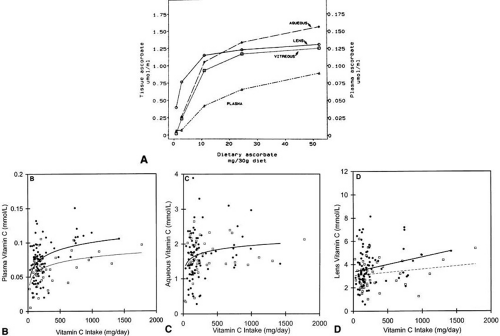
The lens and aqueous concentrate ascorbate greater than 10-fold the level was found in guinea pig and human plasma72,73,81,82 (Fig. 4).
In the lens core (see Fig. 2b), the oldest part of the lens, and the region involved in much senile cataract, the concentration of ascorbate is only 25% of the surrounding cortex.83
Lens ascorbate concentrations are lower in cataract than in the normal lens.84
These data suggest either that there is age-related depletion of ascorbate in the lens or that the bioavailability of this compound changes with age. Enthusiasm for nutrient antioxidants has been fueled by observations that ocular levels of ascorbate are related to dietary intake in humans and animals that require exogenous ascorbate (see Fig. 4).72,81,82 Thus, the concentration of vitamin C in the lens was increased with dietary supplements beyond levels achieved in persons who already consumed more than two times the recommended daily allowance (60 mg/day) for vitamin C.72,73
Feeding elevated ascorbate delayed progress of, or prevented galactose cataract in guinea pigs85 and rats,86 selenite-induced cataracts in rats,87 lens opacification in GSH-depleted chick embryos,88 and delayed UV-induced protein and protease damage in guinea pig lenses.7,8,9,89 Increasing lens ascorbate concentrations by only twofold is associated with protection against cataract-like damage.8
Because ascorbate is a carbohydrate, it is biochemically plausible that vitamin C induces damage in the lens in vivo.90,91 However, there currently are no data to support this as a medical concern. Mice fed 8% of the weight of their diet as ascorbate did not develop cataract.92 If glycation induces pathologic lens damage, antiglycating agents such as phenacylthiazoliums may be useful.93 It is interesting that comparable compounds have been tried as anticataractogens and were assumed to act as reducing cysteine prodrug agents.94
GSH levels are several-fold the levels found in whole blood and orders of magnitude greater than the concentration observed in the plasma. GSHlevels also diminish in the older and cataractouslens.74 There have been several attempts to exploitthe reducing capabilities of GSH. Injection ofGSH-OMe was associated with delayed buthioninesulfoxamine-induced95 and naphthalene cataract.74,96,97 Preliminary evidence from studies with galactose-induced cataract also indicates some advantage of maintaining elevated GSH status in rats.76 However, it is not clear that feeding GSH is associated with higher ocular levels of this antioxidant.76 Other compounds, such as pantetheine, which also includes sulfhydryls, are under investigation as anticataractogenic agents.98 However, the efficacy of this compound in later-life cataract remains to be established.99
Pharmacologic opportunities are suggested by observations that incorporating the industrial antioxidant 0.4% butylated hydroxytoluene in diets of galactose-fed (50% of diet) rats diminished prevalence of cataract.100
Tocopherols and carotenoids are lipid-soluble antioxidants101,102 with probable roles in maintaining membrane integrity103 and GSH recycling.104 Concentrations of tocopherol in the whole lens are in the micrometer range105 (Table 2), but it appears that lens and dietary levels of tocopherol are unrelated.106 Because most of the compound is found in the membranes, particularly in the younger tissues (Taylor and coworkers, unpublished data), the concentrations can be orders of magnitude higher. Age-related changes in levels of tocopherol and carotenoids have not been documented. Tocopherol is reported to be effective in delaying a variety of induced cataracts in animals, including galactose28,107,108 and aminotriazole-induced cataracts in rabbits.109
Elevated carotenoid intake frequently is associated with health benefits. However, little experimental work has been done regarding lens changes in response to variations in levels of this nutrient. It is intriguing that β-carotene levels in the human lenses are limited105 (see Table 2). Instead, major lens carotenoids are lutein/zeaxanthin. These also are the major carotenoids in the macula.110 Also present are retinol and retinol ester, and tocopherols. In beef, β-carotene occasionally was observed in lenses. This apparent quixotic appearance of the β-carotene appears to be caused by seasonal and dietary availability.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree