Nongranulomatous Inflammation: Endophthalmitis, Panophthalmitis, and Sequelae
Russell W. Read MD, PhD
Ehud Zamir MD
Narsing A. Rao MD
Introduction
Inflammation can be defined as a series of responses mounted by an organism to a stimulus interpreted to be hazardous or undesirable. The stimulus may be extrinsic to the organism, such as an invading microorganism, or may be intrinsic, such as that produced by the clearing of internal debris from cell death. This response, mounted by what is collectively termed the immune system, functions to contain and destroy the stimulus; effect repair of any damage created; and then ultimately to turn itself off to prevent further damage. Many different components make up the immune system, including cellular elements such as neutrophils, lymphocytes, and macrophages, and soluble elements such as cytokines, complement, and antibodies. Each of these components has unique but interdependent functions within the immune system. For example, cytokines (small proteins which serve as intercellular signals) such as IL-2 are produced by leukocytes to activate immune cells known as T lymphocytes to prime them for action against the detected threat. Other cytokines, termed chemokines, act to attract additional cellular elements to the area of interest, with these recruited cells then themselves producing additional cytokines, as well as antibodies, which act to contain and destroy the stimulus. All of these components are required for normal functioning of the immune system.
Cellular Components of Inflammation
The inflammatory response is dependent on the actions of many different cell types. These cells express varied surface markers that serve to identify them and allow them to interact with other cells to perform their various functions.
Major Histocompatibility Complex
The Major Histocompatibility Complex (MHC) is a region of genes that encode for the production of small proteins found on the surface of cells. These proteins serve to present antigen in a recognizable form to other effector cells of the immune system, such as T lymphocytes. The term MHC derives from the original description of these molecules in relation to their function in the rejection of transplanted tissues. The MHC system is made up of three subclasses: I, II, and III. All are located on chromosome 6. MHC Class I molecules are found on all nucleated cells. This molecule class functions in the regulation of the actions of CD8+ cytotoxic T cells, especially in the defense against viral infection. Viral antigen present within a cell is processed and presented on the cell surface in association with an MHC class I molecule. This presentation serves to activate CD8+ T cells (see description below) to destroy the infected cell, thus preventing further viral production. MHC class II molecules are present only on certain cells of the immune system, such as macrophages and other antigen presenting cells. Processed antigen is presented in association with MHC class II molecules to CD4+ helper T cells. MHC Class III molecules consist of complement and are not true histocompatibility markers. These molecules play a role in the recruitment of neutrophils and other inflammatory cells and in the amplification of the inflammatory process.
The MHC system in humans is termed the human leukocyte antigen (HLA) system, as these molecules were first discovered on the surface of leukocytes. In the human, MHC Class I antigens consist of HLA types A, B, and C. MHC Class II molecules consist of HLA type D. It is now known that HLA molecules play an important role in an organism’s susceptibility to certain inflammatory diseases. The exact mechanism by which HLA molecules predispose an individual to disease is not known, but several theories exist and are summarized in Table 3.1.
Table 3.1. Proposed Mechanisms of HLA Disease Associations | ||
|---|---|---|
|
Inflammatory Cell Types
The hallmark of acute inflammation is the polymorphonuclear leukocyte or neutrophil. The most numerous of the circulating leukocytes, neutrophils live an estimated 2 to 3 days in circulation. Their primary function is in phagocytosis of foreign material with destruction of the ingested material. Neutrophils may also release their granules and oxygen free radicals, causing damage external to the cell. Neutrophils are attracted by C5a (an activated complement component), IL-8, fibrin breakdown products, and neutrophil chemotactic factor.1 Neutrophils are slowed in their transit through the blood stream via a process called sticking and rolling, which is mediated by L-selectin. Once arrested at a site where chemotactic factor concentration gradients stimulate movement across the vessel wall, this is accomplished via the CD11b/CD18 adhesion complex.2
Small immune cells termed lymphocytes are the hallmark of chronic inflammatory infiltrates, though they may be present from the onset of inflammation. Lymphocytes are divided into B and T types as well as natural killer (NK) cells. Following their production in the bone marrow from pluripotent stem cells, B and T lymphocytes undergo differentiation in the fetal liver/adult bone marrow and thymus respectively. T cells are characterized by the presence of a T cell receptor (TCR) on their surface, of which two main types exist. TCR-1 cells (5% to 10% of circulating T cells) function primarily as cytotoxic cells. TCR-2 cells (90% to 95% of circulating T cells) can be further divided into those which carry the CD4 surface marker and recognize antigen in the context of MHC Class II molecules; and those which carry the CD8 marker and recognize antigen in association with MHC Class I molecules. The CD4 positive subset of T cell, also known as the helper T cell, can again be further subdivided. For many years the classic paradigm of CD4 T helper cell division consisted of a dichotomy of Th1 and Th2 type cells. Th1 cells produce IL-2 and interferon- and promote the functions of cellular immunity. Th2 cells produce IL-4, IL-5, IL-6, and IL-10, which help regulate the functions of macrophages and stimulate B cells to produce antibodies (humoral immunity).1,3 Recent work has shown that a third type of T helper cell exists, the Th17 cell, characterized by the production of IL17 and IL22. Choice of which pathway a naive T cell will follow is determined by the environment in which that cell is first presented its antigen. T cells primed in the presence of IL12, interferon gamma, and IL27 are driven toward Th1 differentiation. Those exposed to IL4 and IL2 are driven toward Th2; and those exposed to IL6, transforming growth factor beta (TGF beta), and IL1 beta toward Th17. Each of these three subsets appears to be primarily active in differing disease states. Th1 and Th17 are involved in autoimmune disease. In their “normal” function, however, Th1 are active in eradicating intracellular pathogens, Th2 in helminthic infections, and Th17 in infections from microorganisms such as Klebsiella pneumoniae and Bacteroides fragilis.4
B cells produce specific antibody directed against that cell’s cognate antigen, following stimulation by helper T cells in association with binding to that target antigen. Continued stimulation leads to their differentiation into plasma cells. As plasma cells produce antibodies, they may accumulate excess amounts within their cytoplasm, producing amorphous eosinophilic structures referred to as Russell bodies.
Mononuclear cells are widely distributed in the body, both in circulating form as well as resident tissue monocytes/macrophages such as Kupffer cells in the liver, alveolar macrophages in the lung, serosal macrophages, and microglia in the brain and retina. These cells provide important functions in the areas of antigen presentation and phagocytosis.1
In addition to the above cell types, eosinophils and basophils/mast cells participate in immune reactions as well, usually in relation to allergic stimuli or parasitic invasion (Fig. 3.1A).
Soluble Components of the Immune System
In addition to the cellular elements described above, the immune system achieves its goal of responding to adverse stimuli via production of numerous soluble factors. Proteins such as cytokines and chemokines signal between cells, some stimulating the system and others inhibiting it. Others such as complement serve both to attract the cellular elements as well as control invading organisms. As a complete discussion of the many soluble components of the immune system is beyond the scope of this text, Table 3.2 lists the sources and functions of some of the better-studied cytokines.
Table 3.2. Features of Selected Cytokines | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Immune Privilege in the Ocular Environment
In order to maintain its role as a functioning organ of sense, the eye has an absolute need for a clear visual axis, normal functioning of the delicate neural networks of the retina and optic nerve, and production of aqueous humor by the ciliary body. Therefore, while the ocular environment needs the protection of the immune system as detailed above, the eye cannot tolerate unchecked inflammation with its attendant destruction of tissue and repair by fibrosis. Evolutionary processes have thus driven the development of unique modifications to the immune system’s standard repertoire, as they relate to the eye.5 Briefly, tight junctions between the cells of the ciliary epithelium, retinal pigment epithelium, and retinal vascular endothelium inhibit penetration into the eye of molecules and cells from the circulation, thus creating a physical blood-ocular barrier.6,7,8,9,10 Lack of a lymphatic drainage system from the intraocular space results in the initial presentation of antigen derived from the eye within the spleen.5 In addition, the unique distribution of antigen presenting cells within the eye,11 presence of immunosuppressive factors within the aqueous humor,12 and the presence of cell surface molecules within the eye which modify the function of the immune system13 all contribute to a modified state of immunity. Initially described in relation to the anterior chamber, this modified immune state is known as anterior chamber associated immune deviation (ACAID). It has subsequently been found to be present in the vitreous cavity and subretinal space as well.5
Classification of Inflammation
Multiple systems of nomenclature have been developed to describe the various causes, patterns, and spectra of ocular inflammation. These classification schemes are variably based on the relationship to the organism’s environment (exogenous vs. endogenous); anatomical area of involvement (anterior, posterior; cornea, retina, iris, etc.); temporal course (sudden onset, insidious onset, acute, chronic); etiology (bacteria, viral, autoimmune, etc.); or specific histopathologic pattern (granulomatous or nongranulomatous; suppurative or nonsuppurative). In actual practice, a combination of these classifications is typically used.
Relation to Organism
Stimuli can be classified as exogenous (coming from outside the organism) or endogenous (coming from within the organism). Under this scheme, a septic emboli from an infected heart valve that lodges in the eye and produces a bacterial endophthalmitis would be considered endogenous, while an acute bacterial endophthalmitis occurring after cataract surgery would be termed exogenous, even though both might be caused by the same bacterial species. This scheme is useful in helping to emphasize whether the source is within the organism, prompting an evaluation to identify this source.
Anatomic
Here the portion of the eye that is primarily involved in the inflammatory process is used with the modifying suffix –itis to characterize the disease process. Thus involvement of the cornea with an inflammatory process secondary to herpes simplex is a keratitis, whereas the same viral organism, affecting the retina, becomes a herpetic retinitis (also known as acute retinal necrosis, which brings up the point that conditions may have multiple names, some developed before the actual etiology of the condition was fully elucidated). Similarly, one can have an iridocyclitis, vasculitis, papillitis, choroiditis, scleritis, myositis, and so on. When more than one anatomical structure is involved, one can combine anatomical terms, resulting in entities such as keratouveitis, sclerouveitis, or retinochoroiditis. The structure primarily involved is listed first, followed by those with secondary involvement. Further modifying terms such as multifocal, focal, or diffuse can also be used. When an inflammatory process involves an ocular cavity and its adjacent structures, the term endophthalmitis is used. When the inflammatory process extends to involve the outer coats of the eye, Tenon’s capsule, and orbit, the term panophthalmitis is used.14 While not necessarily infectious processes, panophthalmitis and endophthalmitis are most commonly used in this context. Exceptions such as phacoanaphylactic endophthalmitis, or “sterile” endophthalmitis exist.
Etiologic Agent
Inflammation within the eye may be induced by any number of sources, exogenous or endogenous, as detailed above. When due to an endogenous noninfectious condition, the actual process at work may not be known, or may be associated with a systemic condition or marker, such as the recurrent anterior uveitis associated with the seronegative spondyloarthropathies. When due to an infectious cause, any microbial agent can serve as the initiating organism, including bacteria, fungi, viruses, protozoa, and helminthes. It is obviously important to differentiate infectious from noninfectious causes, as the former are potentially curable with appropriate antimicrobial therapy.
Temporal Course and Histopathologic Pattern
When faced with a condition of uncertain etiology, samples of intraocular fluids, tissues such as iris, retina and choroid, or both can be obtained.15,16 It is in these situations that the ophthalmic pathologist can be of maximal benefit by establishing the pattern of inflammation present, as well as the presence of infectious agents. The advent of modern molecular biologic techniques such as in situ hybridization, the polymerase chain reaction (PCR), and in situ PCR now allows detection of specific DNA sequences from infectious organisms within tissue or fluid samples17,18 and is of increasing importance in ocular diagnosis.19
Somewhat simplistically, inflammation may be characterized by its time course and characteristic cellular makeup into acute inflammation and chronic inflammation. In acute inflammation, the onset of disease is typically sudden with rapid onset of pain, photophobia, and decreased visual acuity. Based on the histopathologic pattern, acute inflammation can be further divided into acute suppurative inflammation, such as in an infectious endophthalmitis due to virulent bacteria after cataract surgery; or acute nonsuppurative inflammation, such as in an HLA-B27 associated anterior uveitis (see Fig. 3.1B) or Behçet’s disease. Suppurative inflammation is characterized by the presence of neutrophils and their byproducts, collectively known as pus. Suppurative inflammation is very characteristic of acute bacterial infections, such as in acute postoperative bacterial endophthalmitis. Certain bacterial species such as staphylococci commonly produce this type of inflammation and are referred to as pyogenic (pus-producing) bacteria. Conversely, acute nonsuppurative inflammation is that which does not produce purulent material. The predominant cell type in acute nonsuppurative inflammation remains the neutrophil, but there is no formation of purulent material.
In contrast to acute inflammation, in chronic inflammation the onset of disease is typically slower, such as in a Propionibacterium acnes endophthalmitis, in which signs of persistent inflammation may not appear until several weeks following surgery. Whatever the etiology of the inflammation, when it persists for longer than 3 months (in the case of uveitis), it is termed chronic.20 Based again on histopathologic pattern, chronic inflammatory processes can be divided into the two broad categories of chronic granulomatous inflammation and chronic nongranulomatous inflammation. Granulomatous inflammation is typified by the formation of granulomas, with the presence of epithelioid histiocytes against a background of lymphocytes and plasma cells. Multinucleated giant cells may or may not be present. This tissue pattern is produced in response to certain bacterial or fungal infections such as Mycobacterium tuberculosis or Aspergillus, as well as noninfectious processes such as retained foreign bodies, lipid extrusion from a ruptured chalazion, sarcoidosis, and others. This histopathologic pattern of inflammation is discussed in Volume 3, Chapter 4. Chronic nongranulomatous inflammation thus is defined by the lack of these findings, with the predominate feature being an infiltration of lymphocytes and plasma cells.
Uveitis
Uvea is derived from the Latin uva, meaning grape, because of the resemblance of removed uveal tissue to that of a peeled red grape. Thus strictly speaking, uveitis refers only to inflammation of the uveal tract, which is composed of the iris, ciliary body, and choroid. In common practice, however, uveitis is used to refer to any form of intraocular inflammatory disease, including that of nonuveal tissues such as the retinal vasculature. Uveitis may take many forms, from the sudden, explosive onset of an HLA-B27 associated anterior uveitis with hypopyon to the more smoldering course that can be seen in entities such as sarcoidosis. With anterior involvement, patients typically complain of a red, photophobic eye, associated with a decrease in visual acuity and discomfort around the involved eye. Examination in the acute stage reveals conjunctival injection and edema with the classically described sign of a ciliary flush, indicating increased blood flow thorough the ciliary body. The cornea may reveal edema and keratic precipitates. The anterior chamber reveals the presence of leukocytes, and possibly a hypopyon (see Fig. 3.1B). The outpouring of inflammatory mediators and fibrin may result in the formation of adhesions between the iris and the anterior lens capsule, termed posterior synechiae. These adhesions may progress to involve 360° of the pupil, producing a secluded pupil and subsequent iris bombé. The narrowing of the anterior chamber angle thus produced in association with the fibrin may result in peripheral anterior synechiae formation, worsening the degree of angle closure. Because of inflammation of the ciliary body, intraocular pressure is typically low in acute iridocyclitis, but with iris bombé formation and peripheral anterior synechiae, elevated intraocular pressure may result from chronic angle closure, leading to glaucomatous damage to the optic nerve. When involvement is primarily in the intermediate, posterior, or both segments, the complaint may be of blurred vision and “floaters.” Vague discomfort may be felt around the involved eye.
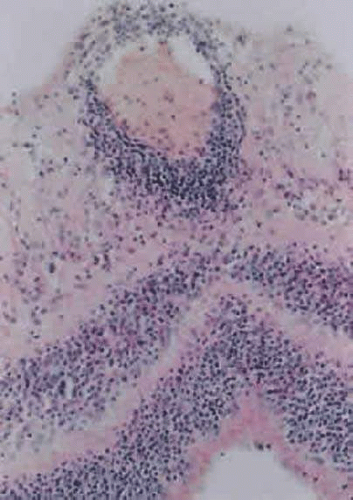
Acute nongranulomatous uveitis does not typically result in the need for enucleation or even biopsy, therefore histopathologic examination of human eyes in the acute stages of disease is rare. When enucleation is needed, it is usually at the end stage of the disease, when all attempts to preserve vision and/or comfort have failed. Thus, most histopathologic descriptions of human uveitis are from chronically inflamed eyes and show an infiltration with chronic inflammatory cells (Fig. 3.2). Most of the information regarding events in acute uveitis has been gained from the use of various animal models of uveitis. Several models exist, but all are based on a similar principle, that of bypassing the normally existing state of self-tolerance and stimulating an organism to attack its own ocular tissues. This is accomplished by the subcutaneous inoculation of various substances (including retinal S antigen, interphotoreceptor retinoid binding protein [IRBP], and melanin-associated proteins) into laboratory animals. A variable period of time later, depending on the specific model, uveitis develops. In experimental autoimmune anterior uveitis, inoculation of Lewis rats with melanin-associated proteins produces primarily anterior segment disease,21,22,23,24 with minimal retinal involvement. The most widely studied animal model of endogenous posterior uveitis is experimental autoimmune uveoretinitis (EAU), induced by the inoculation of either the retinal S antigen or IRBP into susceptible strains of animals. Other models of uveitis include bacterial product-induced intraocular inflammation. In endotoxin-induced uveitis, injection of bacterial endotoxins directly into the vitreous cavity or systemically produces inflammation localized primarily to the anterior segment in Lewis rats.25
Early events in EAU include expression of adhesion molecules on the surface of retinal vascular endothelium, allowing the adherence of circulating leukocytes and subsequent infiltration into the retina.26,27 In Lewis rats, production of EAU by inoculation with retinal S antigen produces an acute cellular infiltrate composed of neutrophils that primarily affects the retina (Fig. 3.3) and anterior segment.28 T cells are necessary for the development of EAU,29,30 with various studies demonstrating the uveitogenic potential of Th1, Th2, and Th17 cells.30 There is likely a genetic susceptibility to the development of EAU,31 which may help explain occurrence of uveitis in only certain individuals. Once initiated, much of the damage resulting from EAU, and most likely in human uveitis, is due to the production of numerous oxygen-derived radicals (superoxide, hydrogen peroxide, hydroxyl radicals, peroxynitrite, and others), which directly attack the photoreceptor layer, producing oxidative damage to these sensitive light-sensing cells.32,33,34
Representative Nongranulomatous Human Uveitic Entities in Which Pathology is Known
Behçet’s Disease
Behçet’s disease is a systemic inflammatory disease predominantly involving the skin, genitalia, and eyes, where it may manifest as a panuveitis. The disease is most commonly encountered in Japan and in the Mediterranean basin area, along the historic “silk route,” an ancient trade route that connected China and imperial Rome, initially developing between 100 BC and the fifth century AD. Disease characteristics and severity vary among patients from different geographical locations, specifically inside or outside the silk route. At least five different sets of diagnostic criteria have been proposed, therefore an international consensus classification was developed and published in 1991 (Table 3.3).35
Table 3.3. Behçet’s Disease Diagnostic Criteria | ||
|---|---|---|
|
It has been shown by polymerase chain reaction methods that the pathogenic gene may exist near the HLA-B region, probably between it and the TNF region.36 Selected human heat shock protein (HSP)-derived peptides (specifically from a 60 kd HSP), show some homology to microbial heat shock proteins and may act as epitopes for T-cell activation in Behçet’s disease.37,38,39,40,41 These human epitopes are uveitogenic when injected in Lewis rats with an adjuvant.41e is that repeated exposure to an antigen may result in positive selection of antigen-specific T cells. Through molecular mimicry mechanisms, these T cells may recognize human heat shock proteins, among other targets, as their autoantigens and therefore initiate an inflammatory response against them.39 An association with HLA-B51 is present, according to some reports, in 81% of Asian patients versus 13% of Caucasian patients.39 The pathogenic significance of HLA-B51 is unknown. Takemoto et al42 showed that B*510101 was associated with disease independent of ethnic group, suggesting that a pathogenic mechanism may be associated with this gene. The HLA-B51 phenotype itself seems to promote neutrophil hyperfunction, as evidenced by hyperactive chemotaxis of neutrophils from B51 positive patients.43 Similarly, the ability of neutrophils to generate superoxide seems to correlate with the presence of the B51 phenotype, regardless of the presence of Behçet’s disease.43 T lymphocytes of patients with Behçet’s disease have a lower threshold for production of interferon gamma than controls when exposed to low concentrations of staphylococcal enterotoxins. Thus, these patients have an antigen-inspecific T cell hyperreactivity, with abnormalities in signal transduction triggered by perturbation of T cell receptors, which may play a role in the pathogenesis of Behçet’s disease.44
One suggested mechanism for Behçet’s diseas
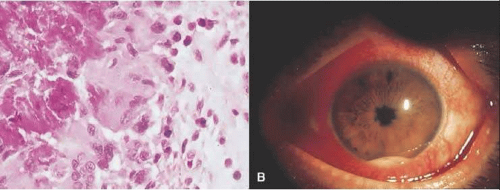
A Japanese national survey of Behçet’s disease has shown that ocular symptoms occur in 86% of patients with Behçet’s disease.45 Common clinical presentations include anterior uveitis with hypopyon (Fig. 3.4), vitreitis, retinal vasculitis (Figs. 3.5 and 3.6), and chronic ischemic optic neuropathy. The hypopyon in Behçet’s disease has been shown to be composed of neutrophils46 and, in its later stages, lymphocytes as well.47 The disease tends to have a remitting-relapsing clinical course and carries a high potential for visually devastating complications.
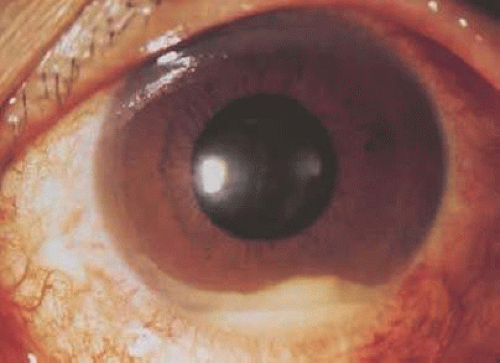 Figure 3.4. Hypopyon in patient with Behçet’s disease. (Photograph provided courtesy of Professor Masahiko Usui, Tokyo Medical University, Japan.) |
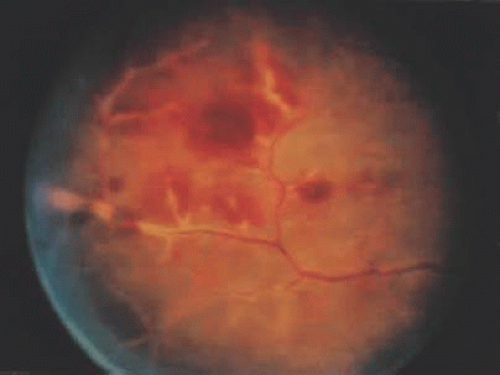
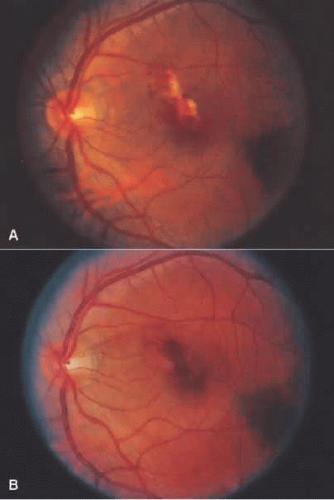 Figure 3.5. (A) Left fundus showing focal area of retinitis with hemorrhage in a patient with Behçet’s disease. (B) Resolution of retinitis after treatment with prednisone. |
Histopathological data about Behçet’s uveitis comes from a few pathological reports on enucleated, blind, painful eyes with chronic disease.48 Light microscopy usually shows a panuveitis with infiltrating polymorphonuclear leukocytes, lymphocytes, plasma cells, and macrophages (Fig. 3.7). Immunohistochemical studies have demonstrated a perivascular choroidal infiltration of activated CD4+, but not CD-8+, T-cells,49,50 as well as focal aggregates of B cells and plasma cells, with increased expression of cell adhesion molecules on vascular endothelial cells.48 These findings imply a mixture of cellular and humoral components in the pathogenesis of Behçet’s ocular vasculitis, where CD-4+ cells activate B cells, resulting in enhanced local production of antibodies and immune-complex deposition.48 However the nature of the antigen in the complex is not known.
Intermediate Uveitis
Intermediate uveitis, as manifested in the classic pars planitis subtype, is characterized by a chronic vitreitis with fibrosis over the inferior pars plana and peripheral retina. The inferior vitreous typically shows white aggregates of inflammatory cells or “snowballs.” Patients usually have minimal external inflammatory signs but report symptoms of floaters and reduced visual acuity, which result from both vitreous opacification and the often-accompanying cystoid macular edema. Other complications may include cataract, glaucoma, cyclitic membrane formation, peripheral retinal neovascularization, macular epiretinal membrane, and retinal detachment. Phthisis may follow in rare, severe cases. This idiopathic condition should be differentiated from other disease entities that may present similarly, such as peripheral toxocariasis, toxoplasmosis, sarcoidosis, and Lyme disease. There is a well-documented association with multiple sclerosis,51,52,53,54,55,56 as well as evidence for the association of both conditions with the HLA-DR15 allele, coding for one of the two HLA-DR2 subtypes.52,54,56
Pars planitis tends to occur in young adults, and generally runs a benign course. Most histopathological data, however, is derived from severely complicated cases that required enucleation, and therefore represent the severest consequences of this entity. The iris and ciliary body are usually reported to be minimally, if at all, inflamed.57,58,59,60 The anterior part of the choroid may reveal a lymphoid cell infiltration in some patients. In other patients with chronic, uncomplicated pars planitis, eyes obtained at autopsy showed no uveal inflammation.58
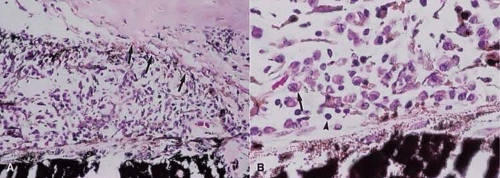
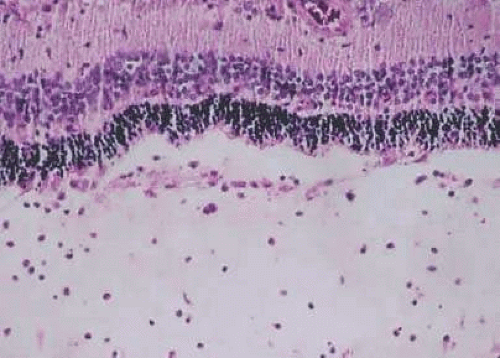
Much attention has been given to the histopathological nature of the “snowbanks,” which has been studied by both light and electron microscopy.57,59,60,61,62,63 These fibrovascular-glial proliferations are found over the inferior pars plana and peripheral retina. They are composed of dense connective tissue derived from condensed, collapsed vitreous, and are often vascularized. These vessels may reveal features of high endothelial venules (Fig. 3.8). Such endothelia are involved in the homing of activated lymphocytes59 and thus their presence in pars planitis specimens suggests that inflammatory cells are preferentially attracted to this area of the peripheral retina. The reason why high endothelial venules would be expressed in the peripheral retinal vasculature is not clear. The cellular elements of these membranes include fibroglial cells that are believed, based on their electron microscopic characteristics, to represent fibrous astrocytes, as well as nonpigmented ciliary epithelial cells, fibroblasts, and lymphocytes (Fig. 3.9).57,58,59,60,64,65 The vitreous is infiltrated by chronic inflammatory cells and despite the nongranulomatous nature of the inflammatory component in the “snowbanks,” vitreous snowballs have been reported to consist of epithelioid cell granulomas.57 The retina often shows a perivasculitis, cystoid macular edema (CME), and epiretinal membrane formation. Inflammation-induced contraction of the vitreous body may cause CME and, through breaks in the inner limiting membrane, predispose to epiretinal fibrous proliferation.58 However, prostaglandins and cytokines released from inflammatory cells may also play a role in the development of CME as well.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree