Fig. 8.1
Shows an EM autoradiogram revealing tritiated thymidine grains (TT) present in the nuclei of transplanted human RPE in owl monkey retina several days after surgery
We then sought to reattach the photoreceptors over the transplanted area by working within a bleb detachment of the neural retina in rabbits and monkeys [4]. The bleb detachment was produced by jet stream force from the transplant micro-pipette. Dissociated, labeled RPE cells were injected over the host RPE. Such transplants survived and phagocytized outer segments. These results prompted us [5] and Turner’s group [6] to use RPE transplantation to treat the Royal College of Surgeons (RCS) strain of rats known to have a defect preventing their RPE from phagocytizing outer segments leading to photoreceptor degeneration. Transplantation of normal RPE did prevent this degeneration from occurring in the area where the transplants were located. Electron microscopy revealed that the transplants contained phagosomes and therefore capable of phagocytosis. This result proved that transplantation of RPE could stop the progression of a degenerative retinal disease(Fig. 8.2).
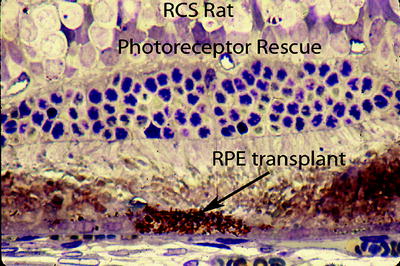

Fig. 8.2
Shows how transplanted normal RPE (arrow) can rescue an adjacent group of photoreceptors from degenerating in the RCS rat
This success prompted the idea that RPE transplantation might have a useful impact on choroidal neovascularization choroidal neovascularization (CNV) that occurs in age-related macular degeneration (AMD). At that time attempts were being made to surgically remove CNV membranes from the macula, but this produced a loss of the adjacent host RPE that was being removed simultaneously. We [7–9] and others [10–12] tried to restore this RPE layer by transplantation after removal of a CNV membrane using either cultured patches of fetal human RPE or dissociated cells. Although some patients maintained foveal function after such surgery, this result was transient lasting less than a month at most (Fig. 8.3).
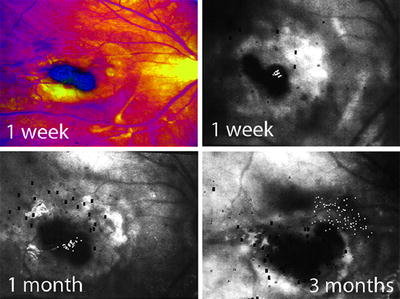

Fig. 8.3
Shows fundus photographs (upper left) and scanning laser micro-perimetry (upper right) of the macular area at 1 week after transplantation of a fetal human RPE patch following removal of a neovascular membrane. The small white spots show light detection over the fovea, the dark spots show scotomas. The lower photographs show how foveation is lost at 3 months
There was a consensus among those using this methodology that host/graft rejection was destroying the transplant. It is interesting that not all such transplants degenerated, however. Figure 8.4 shows an RPE transplant retinal pigment epithelium that slid away from an area of geographic atrophy after transplantation and relocated under a vessel adjacent to the optic nerve where it has remained unchanged for at least 3 years (Fig. 8.4). We have found similar results with human RPE patch transplants to monkey retina. When we transplant a patch to the fovea area versus the peripheral retina, we found a greater chance that a rejection-like picture occurred in foveal transplants (Fig. 8.5). Some RPE xenografts can survive for long periods of time without rejection. We found that foveal transplants are more prone to rejection than peripheral ones [13].
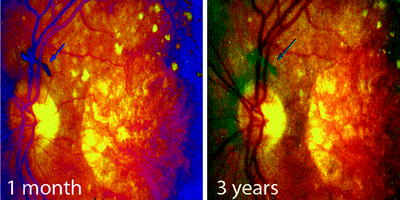
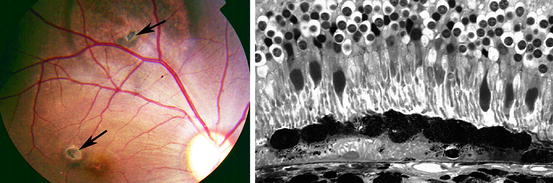

Fig. 8.4
Shows a transplanted patch of heterologous human fetal RPE which floated away after being transplanted to the macula of a patient with geographic atrophy and remained unchanged (arrows) for at least 3 years

Fig. 8.5
Shows on the left a fundus photograph of a rhesus monkey that received two human fetal RPE xenografts, one in the periphery (upper arrow) and the other in the fovea (lower arrow). The demarcation line of the detachment is larger in the periphery. On the right is an example of such a xenograft, more peripherally located that shows no sign of rejection in monkey retina 5 months after surgery. This transplant and host photoreceptors survive even though the transplant rests on the host RPE layer
Rejection of RPE transplants in the RCS rat has also not been very obvious. There is only one report of host/graft rejection of heterologous RPE transplants in the RCS rat, and this was atypical, being humoral rather than cellular [14]. Therefore the poor success of RPE transplantation may not be due only to rejection but to other factors such as surgical technique.
Autologous RPE Transplants
A new approach emerged that eliminated the problem of host/graft rejection retinal pigment epithelium by excising a patch of peripheral RPE together with the choroid from the patient’s own retina and transposing this patch to the macular area after a CNV membrane had been surgically removed [15–18]. What is remarkable about this method is that the choroidal vessels in the transplanted patch re-vascularize [19]. But this method has some drawbacks. One is that the host’s peripheral RPE patch is senescent and probably less viable than embryonic tissue. The second is that it requires two surgical procedures, the removal of the peripheral RPE patch with its choroid and the macula surgery. Another consideration is the difficulty of working within a macular bleb detachment, which is now being altered by a new surgical technique that exposes the macula.
Exposing the Macula
Surgical manipulation within a bleb detachment is awkward. It restricts the microsurgery, obscures the visibility, and tears the paramacular neural retinal opening needed to enter the bleb detachment. An improvement has been introduced to facilitate such surgery, which allows better access to the RPE by folding the neural retina away from the macula. This approach uses 1800 retinotomy at the temporal ora serrata in order to fold the neural retina nasally exposing the macular RPE (Fig. 8.6). I have often considered this approach to be advantageous for macular transplantation and surgery. This approach has now been used in human subjects by groups in Italy [20] and China (in press). This allows the surgeon to use both hands in removing a CNV membrane and/or a degenerate RPE layer, in dissecting a peripheral RPE-choroid patch and in placing it properly in the macula.
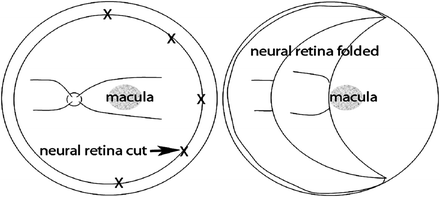

Fig. 8.6
Shows how the neural retina is cut at the ora serrata (crosses) and folded nasally to expose the macula
Removing Degenerate RPE
Reconstructing a new RPE layer should ideally include removing any degenerate RPE cells and if necessary, any CNV membrane. Exposing the macula RPE is important because it allows visualization of the RPE layer directly. With such visibility, the host’s macula RPE can be removed more easily as it was using the “open sky” procedure in the owl monkey. Wiping may be more traumatic than using more precisely controlled methods such as an ultra-sonic or a femto-laser probe. Because the femto-laser’s pulses are so brief, they do not heat up adjacent tissue keeping their ablation effects localized. Better control of the debridement of the host RPE might be facilitated by robotic surgery where movements of a few microns are possible.
Delivery of the Transplant
Delivering a mono-layer patch of RPE with the proper orientation and flatness presents another problem for transplantation. One method suggested, but not pursued consistently, is encasing a segment of cultured cells in a gel that is rigid at room temperature but fluid at body temperature. This could facilitate delivering an undamaged, flat transplant with appropriate polarity. If the sclera port were too small to introduce the patch, it could be delivered in separate segments. A supporting scaffold, natural or artificial, may be required to facilitate RPE cell delivery to the eye. Research to improve the biomimetic properties of such materials is being pursued [21–26]. A 3-dimensional scaffold may be effective in growing a 3-dimensional structure such as the entire retina but for therapeutic transplantation a 2-dimensional RPE monolayer seems more appropriate. All manipulations would be facilitated by exposing the macula RPE and Bruch’s membrane by folding of the neural retina nasally. Such exposure might even allow transferring an un-encased patch of RPE using a micro-spatula. But reattachment of a folded neural retina is a drawback to the proposed surgery since it is a large detachment including the macula. But if one is attempting to reconstruct a blind fovea, it might be worthwhile.
Iris Pigment Epithelium
Iris epithelium is closely related to RPE and is readily available by biopsy from potential recipients. But research by several groups [27–29] has not achieved success therapeutically even though it eliminates host/graft rejection. It indicates that there are other factors than rejection affecting epithelial transplantation, such as surgical technique, full exposure and preparation of the site, establishment of the proper flatness of the transplant as a monolayer, the virility of the transplant and its ability to interact in many unique ways with the photoreceptor layer.
RPE Derived from Stem Cells
The concept of using embryonic stem cells to treat disease has been complicated politically because it implied the use and destruction of human fetuses. Embryonic RPE cells have an advantage compared to adult RPE, however, in being very viable in culture and lacking any of the senescent changes that accumulate in adult RPE. Nevertheless they are heterologus and therefore subject to rejection. In 2006 a new era in stem cell research occurred with the demonstration that adult differentiated cells could be induced to become pluripotential by transducing them with unique combinations of transcription factors, Oct3/4, Sox2, Myc, and Klf4 [30], and these pluripotent cells could be further transformed into tissue specific cells such as RPE. This breakthrough meant that human embryos were unnecessary for obtaining stem cells and autologous cells could be obtained from the recipient obviating host/graft rejection although host/graft rejection may still occur [31]. These adult-induced pluripotential cells express similar genes to embryonic stem cells [32]. Recent reports indicate that differentiated adult fibroblasts can be transformed directly into neural cells by also using a unique combination of transcription factors but without going through a prior pluripotential stage [33–35]. Such transformed fibroblasts can be cultured, transformed into RPE, and tested for the presence of RPE-specific proteins, such as RPE 65, bestrophin 1, CRALP. These transformed cells would be pristine new without the waste products that accumulate in senescent RPE. Such cells could be easily cultured providing the option of genetically engineering them in vitro to express proteins that counteract genetic defects or which are trophic factors that promote survival [36–38].
Prophylactic RPE Transplantation
Will RPE transplantation continue to evolve and become a therapeutic method to treat blinding degenerations such as AMD? Optimists think it can but to do so it has to be performed before there is massive destruction of the photoreceptors in the fovea, as occurs in the late complication of CNV or geographic atrophy. This would then require prophylactic surgery while the patient still has foveal vision, which is the ultimate challenge for this methodology. This cannot be done at present but there is continued research trying to improve it so the method cannot be discarded. What may supersede the simple replacement of degenerate with healthy RPE, however, is the possibility to reconstruct the fovea after total loss of the photoreceptors has occurred by transplanting new photoreceptors especially cones, together with new RPE. This would allow the reconstruction of the fovea to take place in an already blind eye making any potential failure trivial. This may be the most promising future of cell transplantation in the retina.
Photoreceptor Transplantation
By comparison with RPE transplantation successful transplantation of photoreceptors is much more difficult. But it is sensational since it could restore sight to a blind eye rather than merely saving residual sight, the hope of RPE transplantation. Because it is so sensational it has had a complex history of exaggeration and confabulation. The major difficulty with the approach stems from a key problem, the inability of such transplants to form synapses with host neurons which is essential for proper visual function. There are several reasons for this problem, one obvious and the others arcane. The obvious problem is to obtain photoreceptors devoid of their contacts with their own second order neurons, which block any contacts of the receptors with host second order neurons. The arcane problems involve our inability to control and direct synapse formation from photoreceptors to natural second order retinal neurons.
Whole Retinal Sheets
Many ways have evolved in the many attempts to transplant photoreceptors. One has been to use a sheet of neural retina that is placed between the host RPE layer and the host neural retina [39]. This creates two retinas, one from the transplant and the other from the host, the latter usually with either degenerate or absent photoreceptors. The hope has been that the transplant will extend neural processes that can make synapses with second and/or third order neurons in the degenerate host retina, which could provide a functional connection from transplanted photoreceptors to host ganglion cells and ultimately the brain. Those championing this method have evidence that such synapses can form between the two retinas [40, 41]. Attempts using this method have been tried in both animals and blind human subjects with reports of success. But the approach has not been taken up by the ophthalmic community, undoubtedly because of its relatively minimal effects on vision. The number of synapses that form between these two retinas must not be plentiful enough for any useful vision. This approach seems to be an awkward way to restore retinal function because it does not try to reconstruct the retina in the natural way. Connecting what is the ganglion cell layer of the transplant with the outer nuclear layer of the host retina seems less rational than trying to connect transplanted isolated photoreceptors to their logical second order neurons, bipolar and horizontal cells.
Transplantation of Retinal Micro-aggregate
Small micro-aggregates from mature retina or from 3 to 4 days old mice, an age when photoreceptors are just developing outer segments, have been used as transplants [42–44]. Some of these micro-aggregates contain photoreceptors separated from their second order neurons making them potentially able to form new synaptic contacts with host bipolar cells but such synaptic reconnections have been difficult to find[46]. We have transplanted micro-aggregates into the subretinal space of rd mutant mice, at a stage where these mice have lost all of their rods and most of their cones. In early studies we only labeled the donor rods. In later ones both the donor rods and the host rod bipolar cells were labeled [45, 46] (Fig. 8.7). Transplanted, undifferentiated photoreceptors develop normal outer segments which survive for long periods of time, perhaps years, in the degenerate mouse retina. We have learned much from these experiments.
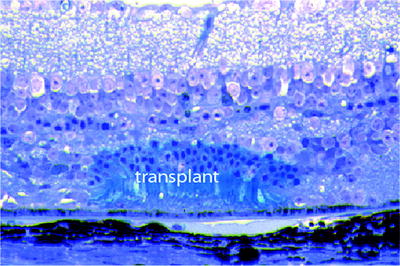

Fig. 8.7
Murine photoreceptors in a micro-aggregate labeled with the lacZ reporter gene (blue transplant) and transplanted to the subretinal space of an rd mouse where all of the host rods and most of the cones have degenerated. This transplantation occurred 11 months previously, and there is no evidence of host/graft rejection
Outer segments only develop if they are oriented in the proper direction, i.e. with the outer segments contacting the RPE layer. Second, the external limiting membrane remains a significant obstacle that blocks contacts between the transplant and host second order neurons. Third and most important we have been unable to detect many synaptic contacts between labeled donor rods and labeled host rod bipolar cells by electron microscopy. Figure 8.8 shows a rare example of lacZ reaction particles labeling a transplanted rod spherule in an adult rd mouse retina in which only the host rod bipolar cells were also labeled. There is a lacZ particle present on the postsynaptic side of this synapse implying that it belongs to a host rod bipolar cell which suggests that synaptic communication exists between the host and the transplant via a canonical synapse. But this is our only good example among many attempts. We also found membrane-to-membrane contacts between labeled rods and labeled rod bipolar cells, which could allow ephaptic transmission between transplant and host, i.e. K+ release from the rod could depolarize the host bipolar cell and generate a signal between the transplant and the host retina. Classic synaptic transmission between donor rods and rod bipolar cells that were labeled to be recognizable at the electron microscopic level was extremely rare.


Fig. 8.8
LacZ labeled rod spherule transplanted to the subretina of an rd mouse whose rod bipolar where also labeled by LacZ. The lower two arrows indicate Lac Z reaction particles within the spherule. One label (uppermost arrow) is in a post-synaptic structure implying that it is a host rod bipolar cell
The one shown in Fig. 8.8 is the only convincing sample of such an event we found. The rarity of either canonical synaptic as well as ephaptic contacts between the transplant and host retina indicates that such occurrences are too rare with current techniques. Fourth, we have not seen host/graft rejection, which implies that such neural tissue within the subretinal space may be tolerated or perhaps not detected by the immune system, although there is evidence against such a conclusion [47–49]. It is interesting that the latter study, which transplanted neural progenitor cells from humans into pigs, used laser photocoagulation to promote integration. The rejection encountered might be due to the prior laser treatment that could cause a considerable local inflammatory reaction. It is our impression that rejection is variable and can depend on the local inflammation produced by the surgery. In experiments with subretinal injection of viral solutions that led consistently to cellular rejection, immune-suppression for only a month prevented rejection permanently [50] suggesting that after the initial inflammatory response to the retinal surgery dissipates, the foreign material within the subretinal space may no longer be detected by the immune system.
Transplantation of Dissociated Photoreceptors
Townes-Anderson et al. [51] first reported a method to dissociate isolated rods. We have used this method to isolate photoreceptors for transplantation in rats [52] and mice [42, 43, 52]. Mature as well as progenitor photoreceptors can survive when transplanted to these retinas [53]. Recently cell suspensions of enzymatically dissociated retinas of 1- to 4-day-old mice have been used to obtain isolated photoreceptor cells with similar success [54]. Using a similar approach [55] one group concluded that such transplanted photoreceptors seem to integrate more consistently into the outer nuclear layer and showed more evidence of functional communication between transplant and host retina. The evidence for synaptic mediated function, the key challenge in this field, has been examined by both immunohistochemical and functional methods; the latter including pupillary responses and electrical field potentials from the ganglion cell layer. The ganglion cell recordings indicated increased activity and greater sensitivity in mice with retinal degeneration that had received such transplants than control mice. Curiously, prenatal and later postnatal transplants were less effective. The overall gain in visual function was small, however, most likely because the numbers of integrated and communicating transplanted photoreceptors were few. Attempts are being made to increase the amount of integration by disrupting the blocking outer limiting membrane [56, 57], by enzymatically degrading the inhibitory extracellular matrix (ECM) and cell adhesion molecules, such as CD44 and neurocan [38, 58, 59], by immune suppression [48, 49] by anti-apoptotic treatment of donor cells [38], by enrichment of labeled cells by flow cytometry [60] or by magnetic-assisted cell sorting [61]. The latter method appears to be the most successful in increasing the integration of young rods enzymatically disassociated from normal murine retina using magnetic beads with antibodies to a surface protein, called cluster of differentiation (CD73) expressed in young rods. This procedure significantly increased the number of transplanted rods integrating into murine retina. This method has the advantage of not requiring genetic modification of the photoreceptors in order to uniquely detect and concentrate them.
Retinal Progenitor Cells
This method involves selecting so-called progenitor cells that exist in the young murine retina especially at the ciliary margin and the optic nerve head [62]. Such cells are undifferentiated, express developmental markers, and can be distinguished by their organization in cultures. Transplantation of such cells into degenerate mouse retina shows that they can express photoreceptor proteins and improve visual performance in behavioral tests of vision. Visual improvement could also be due to trophic influences the transplant exerts on the residual host photoreceptors. This approach has a handicap that the progenitor cells are heterologous and therefore subject to rejection. This method is being eclipsed by recent attempts to transform the host’s own differentiated cells into pluripotential cells that should eliminate host/graft rejection.
Transforming Fibroblasts into Photoreceptors
Takahashi and Yamanaka’s demonstration [30] that differentiated cells can be re-programmed into pluripotential cells has had vast confirmation and now involves a variety of techniques [63–77]. This has influenced photoreceptor transplantation [78, 79]. The latter have transformed fibroblasts into pluripotential cells and selected cells that expressed visual proteins for transplantation into the subretinal space of degenerate murine retinas. They obtained integration and expression of photoreceptor genes in these transplants and in addition evidence of visually evoked responses from the mice. This offers the opportunity to take skin biopsies from patients with genetic defects that lead to retinal degenerations and produce photoreceptors that express the deleterious mutation in vitro, which can facilitate studying the pathogenesis of such diseases. The approach is being intensely pursued. It is possible to use only one and not several viral vectors to reprogram these cells [63] or to eliminate the viral vector completely by using nucleo-fection of a polycistronic construct co-expressing Oct4, Sox2, Klf4, and Myc [80] or only one transcription factor [66].
< div class='tao-gold-member'>
Only gold members can continue reading. Log In or Register to continue
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


