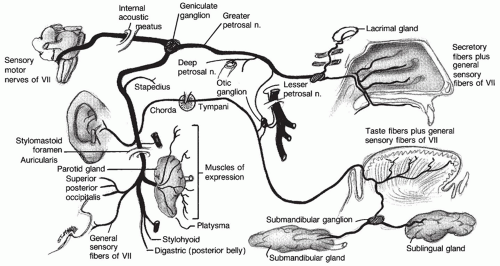
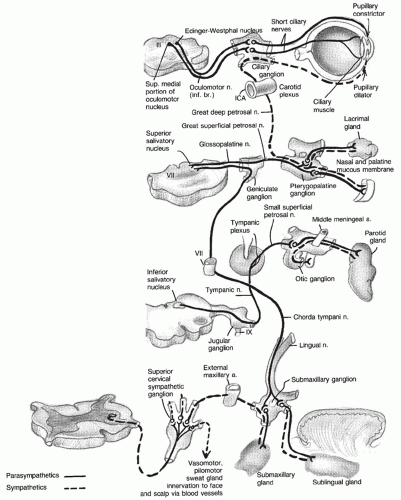
Abnormalities of blood flow to the central nervous system (CNS) are a common cause of morbidity and mortality across the world. In the United States, cerebrovascular disease is the third leading cause of death and the primary cause of long-term disability (
1). Stroke can be either hemorrhagic or ischemic in nature, with ischemic strokes being far more common and making up approximately 87% of all cerebrovascular events (
1). Clinical presentations and functional deficits can be quite variable depending on the area of the CNS involved. Permanent losses typically result from complete arterial occlusion or from hemorrhagic infarction. Transient effects may accompany arterial stenosis, vascular spasm, inadequate perfusion pressure, or reversed arterial flow with shunting such as subclavian steal syndrome.
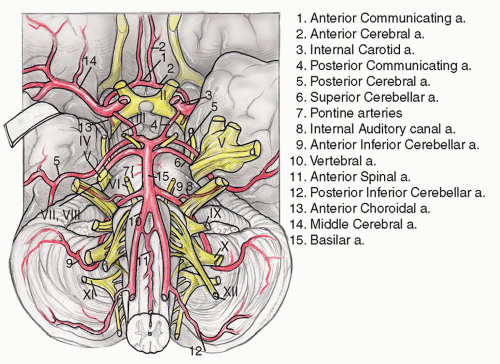
The blood supply to the inner ear, brainstem, and cerebellum arises from the vertebrobasilar system, thus vertigo is a common manifestation of cerebrovascular disease and can be caused by occlusion of any of the three major circumferential branches of the vertebral or basilar arteries: the posterior inferior cerebellar artery (PICA), the anterior inferior cerebellar artery (AICA), and the superior cerebellar artery (SCA) (
Fig. 13.3).
Vertebrobasilar Insufficiency
Vertebrobasilar insufficiency is a significant cause of vertigo in individuals over age 65 (
2). Vertigo due to vertebrobasilar insufficiency has a sudden onset, usually lasts several minutes, and is often associated with nausea and vomiting. Because of the presence of vertigo symptoms, the otolaryngologist may be called upon to make this diagnosis; however, vertigo symptoms alone are extremely uncommon and occur in less than 1% of patients (
3). Usually, multiple symptoms suggestive of central etiology are present which often include headache, diplopia, ataxia, numbness, weakness, or oropharyngeal dysfunction (
4). Repeated episodes of vertigo without other symptoms suggest another diagnosis.
Atherosclerosis of the subclavian, vertebral, or basilar arteries is often the underlying cause of vertebrobasilar insufficiency. The distribution of atherosclerosis differs according to race and gender (
5). European men tend to have
atherosclerosis at the origin of the vertebral arteries near the subclavian. Intercranial large-artery atherosclerosis is more common among African Americans, Asians, and women. Hypertension increases the risk of lipohyalinotic thickening of these vessels, which increases the risk of infarction.
If a patient is suspected to have had a stroke, a computed tomography (CT) scan without IV contrast should be ordered to rule out the possibility of a hemorrhagic event. Patients with ischemic strokes who present within three hours of the onset of symptoms should be considered for administration of intravenous tissue-type plasminogen activator (
6). However, recommendations in this area are rapidly evolving, and a neurologist familiar with stroke should treat acute patients.
In most patients with nonacute presentation, treatment consists of controlling risk factors such as diabetes, hypertension, and hyperlipidemia. In patients with symptomatic intracranial stenosis of 50% to 99%, aspirin is the treatment of choice. Anticoagulation with warfarin should be considered if the patient has previously had an embolic stroke, which is cardiac in origin (
2). Stenting of vertebral artery stenosis has been attempted with mixed results. Endarterectomy for extracranial vertebral artery disease has also been successfully performed, but indications for such procedures are still evolving.
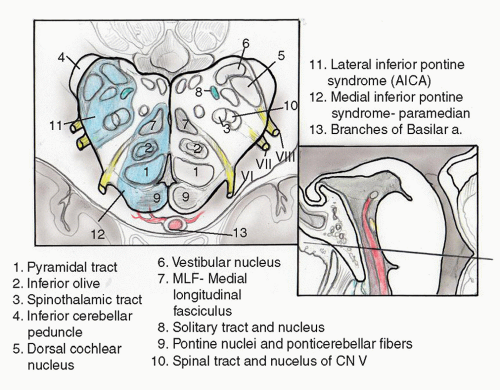
Lateral Medullary Syndrome
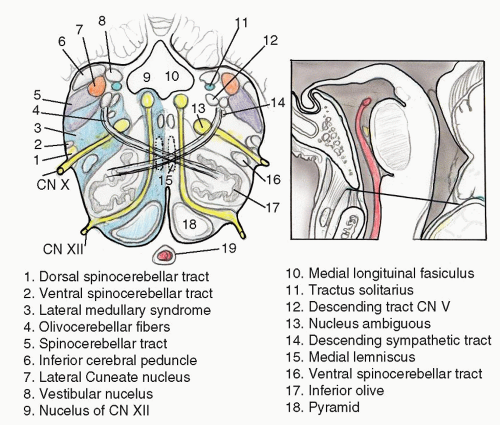
Lateral medullary syndrome (Wallenberg syndrome) is caused by an infarction involving a wedge of the dorsolateral medulla just posterior to the olive (
Fig. 13.4). The syndrome usually results from occlusion of the ipsilateral vertebral artery and rarely from occlusion of the PICA (
7). Classic presentation includes sensory deficits affecting the trunk and extremities on the opposite side of the infarct and sensory and motor deficits affecting the face and cranial nerves ipsilateral to the infarct. Characteristic symptoms include vertigo, ipsilateral facial pain, diplopia, dysphagia, and dysphonia. Neurologic examination may reveal an ipsilateral Horner syndrome, ipsilateral dysmetria, dysrhythmia, spontaneous nystagmus, and contralateral loss of pain and temperature sensation. Hearing is not preserved because the lesion is caudal to the cochlear nerve entry zone and cochlear nuclei.
Lateral Pontomedullary Syndrome
Occlusion of the AICA results in infarction of the dorsolateral pontomedullary region and the inferolateral cerebellum (
8). The middle cerebellar peduncle is typically the core of the affected territory (
Fig. 13.5). Because the labyrinthine artery originates from the AICA in 80% of patients, infarction of the membranous labyrinth is a common accompaniment. Severe vertigo, nausea, and vomiting are common initial symptoms. Other symptoms include ipsilateral hearing loss, tinnitus, facial paralysis, and cerebellar asynergy. Spontaneous nystagmus is common. Contralateral loss of pain and temperature sensation may be caused by crossed spinothalamic fibers. Onset of symptoms is acute followed by gradual improvement. Vertigo may persist longer than would be expected from a pure vestibular lesion due to damage of the central compensation mechanisms.
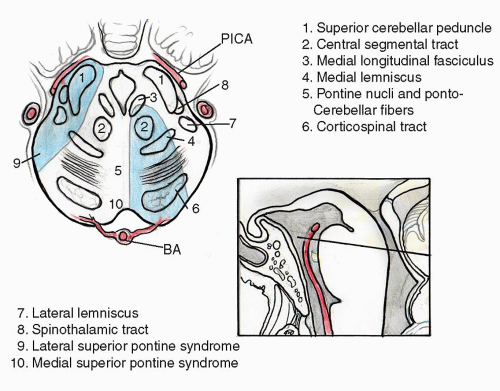
Midbrain Syndromes
Occlusion of the posterior cerebral artery (PCA) or its more distant branches can result in several brainstem syndromes that may present with cranial neuropathies (
Fig. 13.6).
Paramedian midbrain syndrome (Benedikt syndrome) involves the oculomotor fascicle, red nucleus, medial lemniscus, and dentatothalamic tract. An ipsilateral oculomotor nerve paresis with contralateral chorea or tremor secondary to red nucleus involvement is seen.
Weber syndrome similarly affects the oculomotor fascicle, but also involves the medial midbrain. Here, penetrating branches of the PCA supplying the cerebral peduncle are affected, leading to defects in the corticospinal and
corticobulbar tracts, which manifest as contralateral hemiparesis. Dorsal midbrain infarction leads to
Parinaud syndrome resulting in a tetrad of findings: (a) loss of upward gaze (with loss of smooth pursuit and saccades but with vestibuloocular reflex preservation), (b) large pupils with loss of light reflex, (c) convergence-retraction nystagmus, and (d) lid retraction. Parinaud syndrome is due to lesions affecting the posterior commissure and pretectal region, and can often be identified in an asymptomatic patient on physical examination.
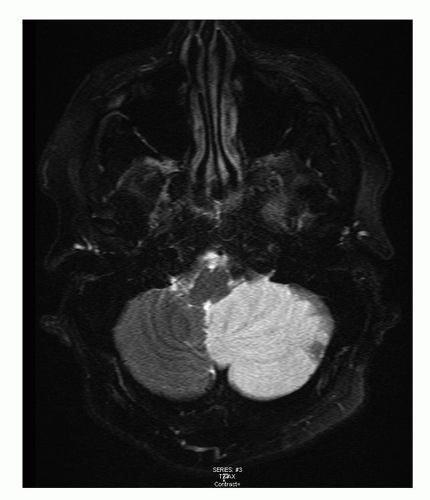
Cerebellar Infarction
Initial symptoms of cerebellar infarction are often non-specific and many include severe vertigo, vomiting, and ataxia (
9). This may be caused by occlusion of the vertebral artery, the posteroinferior cerebellar artery, the anterior inferocerebellar artery, or the SCA. Lack of typical brainstem signs may cause an incorrect diagnosis of an acute peripheral labyrinthine disorder. The key to the diagnosis is the presence of prominent cerebellar signs such as gait ataxia and paretic gaze nystagmus. An early CT scan will often miss posterior fossa ischemia. The diagnostic procedure of choice is magnetic resonance imaging (MRI,
Fig. 13.7). After a latent interval of 24 to 96 hours, some patients develop progressive brainstem dysfunction caused by compression by a swelling cerebellum. Progression to quadriplegia, coma, and death may follow, and early surgical decompression should be considered.
Cerebellar Hemorrhage
Hemorrhage into the cerebellum causes neurologic symptoms that often rapidly progress to coma and death (
10). The initial symptoms include severe vertigo, vomiting, and ataxia. Similar to cerebellar infarction, brainstem signs may not initially be present, which may cause the condition to be confused with a peripheral vestibular problem. Rapid diagnosis can be made with CT or MRI. Mortality with this condition remains near 25%, with a poor neurologic outcome in many who survive (
11). Better initial neurologic status at presentation is highly correlated with a favorable eventual outcome. The value and indications for surgical intervention remain controversial and likely depend on the size and location of the hematoma as well as the patient’s symptoms (
12).
Subarachnoid Hemorrhage
Subarachnoid hemorrhage (SAH) frequently occurs in young and middle-aged adults. Eighty-five percent are caused by aneurysms, most commonly saccular (berry) aneurysm rupture within the Circle of Willis (
13,
14). Cigarette smoking, age, hypertension, and genetic factors have all been associated with increased risk. Most cases occur before age 55. Presenting symptoms include a sudden explosive headache (“worst headache of one’s life”), nuchal rigidity, decreased consciousness, photophobia, meningismus, nausea, and vomiting. The fatality rate is near 50% with many dying prior to arrival at the hospital (
14), The seriousness of the condition necessitates a high clinical suspicion to ensure rapid diagnosis and management. Early CT scan is the diagnostic test of choice and may detect subarachnoid blood in 95% of patients if performed within 24 hours from time of rupture (
15). If CT is negative and clinical suspicion is high, lumbar puncture should be pursued. Cerebrospinal fluid (CSF) analysis classically shows xanthochromia. If SAH is present, cerebral angiography should be completed with management focused on eliminating the aneurysm from the circulation, which is often addressed using intravascular techniques. Subarachnoid blood is associated with high risk of seizure development, and early seizure prophylaxis should be considered.
Evaluation and Management
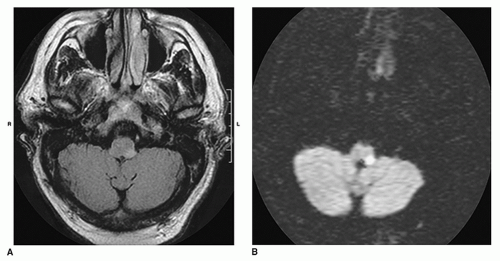
Acute spontaneous vertigo is a frequent reason for referral to an otolaryngologist. The possibility of a stroke must be considered, particularly if the patient has vascular
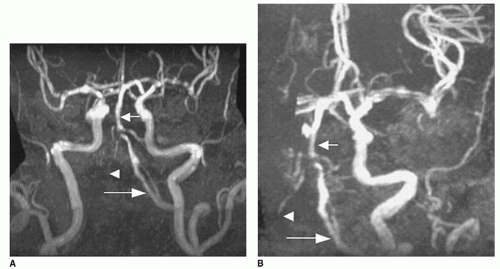
risk factors or other cranial neuropathies. If the history and examination suggest a vascular disorder, immediate neuroimaging is recommended. CT of the brain without contrast is often the first imaging test because it is widely available and shows intraparenchymal or subarachnoid blood. Hemorrhage can mimic any of the ischemic stroke syndromes and must be excluded with thin-section CT through the cerebellum, brainstem, and fourth ventricle. MRI and MRA are superior to CT for visualizing the vertebrobasilar vessels and their supplied structures since CT is often normal with cerebellar or brainstem infarction. MRI, on the other hand, detects ischemic strokes in the brainstem and cerebellum early on, as well as edema. Diffusion weighted imaging with MRI can now detect infarcts within the first hour of ischemia (
Fig. 13.8). MRA has begun to replace conventional angiography in certain circumstances. Vertebral artery dissection and vertebral or basilar stenosis/occlusion can generally be identified with MRA of the cervical vessels (
Fig. 13.9).