Neoplasms of the Temporal Bone & Skull Base: Introduction
The skull base includes the frontal bone, the sphenoid bone, the temporal bone, and the occipital bone. Tumors of the temporal bone and skull base tend to arise in one of three locations: (1) the mastoid or middle ear, (2) the jugular foramen, or (3) the petroclival junction or petrous apex. Tumors of the cerebellopontine angle and Meckel cave are not considered in this chapter (see Chapter 61, Nonacoustic Lesions of the Cerebellopontine Angle). Tumors arising within the skull base are rare and usually cause few symptoms until they grow to a size in which they begin to affect cranial nerves. Table 65–1 lists the various skull base neoplasms and their imaging characteristics.
| Neoplasm | Most Common Site of Origin in Skull Base | CT | T1-Weighted MRI | T2-Weighted MRI | Contrast Enhancement |
|---|---|---|---|---|---|
| Paraganglioma | Jugular foramen and middle ear | Bone destruction | Intermediate, with flow voids | High, with flow voids | Strong |
| Facial nerve schwannoma | Geniculate ganglion | Smooth remodeling and dilation of the surrounding bone of the facial canal | Intermediate | Intermediate | Strong; follows the course of the facial nerve |
| Geniculate hemangioma | Geniculate ganglion | Erosion of surrounding bone with bony spicules within tumor | Intermediate | High | Strong |
| Leukemia, lymphoma, and plasmacytoma | Petrous apex | Lytic lesion | Low | Intermediate | Moderate |
| Langerhans cell histiocytosis | Mastoid | Irregular bone destruction; may have other skull lesions as well | Intermediate | High | Moderate |
| Chondrosarcoma | Petroclival junction | Bone destruction, but can produce calcium matrix in 50% of tumors | Intermediate | High | Moderate or mixed |
| Chordoma | Clivus | Bone destruction, but can have bone remnants within it | Intermediate, with some areas of low signal representing mucus | High | Moderate or mixed |
| Meningioma | Posterior face of temporal bone | Surrounding hyperostosis and intratumoral calcification | Intermediate | Intermediate | Strong; characteristic dural tail |
| Intralabyrinthine schwannoma | Within the inner ear | Mass within labyrinth; no bone erosion | Low | Intermediate | Strong |
| Schwannoma of jugular foramen | Jugular foramen and middle ear | Soft tissue mass posterior to jugular bulb; mild, smooth bone erosion | Low | Intermediate | Strong |
| Rhabdomyosarcoma | Anywhere, predominant tumor of children | Bone destruction | Intermediate | High | Strong |
| Osteosarcoma | Anywhere | Either lytic osteoblastic or osteolytic; may have concentric rings of calcium | Intermediate | High | Strong |
| Fibrosarcoma | Anywhere | Bone destruction | Intermediate | High | Strong |
| Adenoma | Middle ear | Middle ear soft tissue mass; no bone erosion | Low | Intermediate | Strong |
| Endolymphatic sac tumor | Posterior face of temporal bone | Bone destruction and erosion of otic capsule | Mixed, due to localized areas of mucus | Mixed | Strong |
| Carcinoma | Middle ear | Bone destruction | Intermediate | Intermediate | Strong |
| Metastatic disease | Petrous apex and internal auditory canal (IAC) | Lytic lesion | Intermediate | High | Strong |
The majority of skull base tumors are benign and can typically be successfully managed by an otolaryngologist specialized in neurotology and skull base surgery. Surgical approaches to these three areas are numerous, and the nomenclature is confusing. To remove a lesion of the middle ear or mastoid, a mastoidectomy through a postauricular incision or a middle ear exploration through the ear canal is usually adequate. Tumors of the jugular foramen require a postauricular incision that extends down into the upper neck. A mastoidectomy is performed along with skeletonization of the facial nerve, the sigmoid sinus, and the jugular bulb. One classic approach to the jugular foramen is the Fisch Type A approach (Figure 65–1). This involves dissecting the facial nerve out of its bony canal and rerouting it anteriorly. Permanent facial paresis or synkinesis can occur. Closure of the ear canal is also part of the Fisch Type A approach, which leaves the patient with a maximal conductive hearing loss. However, newer approaches are available that may permit adequate exposure of the jugular foramen without requiring facial nerve rerouting and closure of the ear canal. Finally, tumors of the petroclival junction and petrous apex require either a middle fossatranspetrous approach with removal of the petrous apex bone (Kawase triangle, Figure 65–2) or a combined subtemporal-retrolabyrinthine approach (Figure 65–3). The Fisch Type B and C approaches can also be used to access the petroclival junction and can be extended all the way to the nasopharynx, orbital apex, and cavernous sinus. Surgical strategies are chosen by the skull base surgeon based on approaching the tumor with enough exposure to perform a complete and safe resection while minimizing neurologic morbidity.
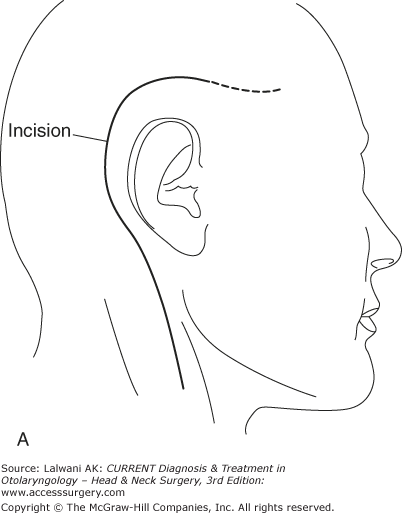
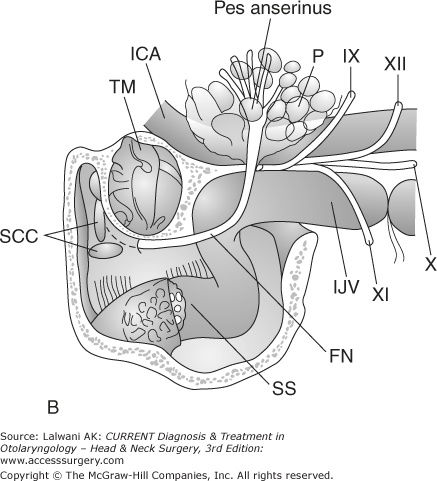
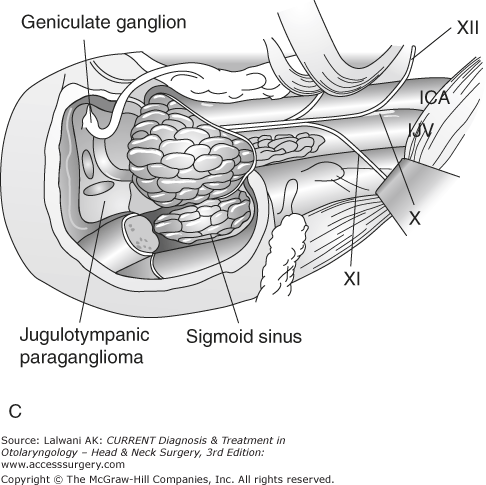
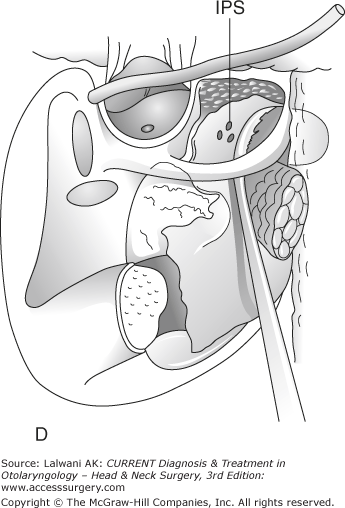
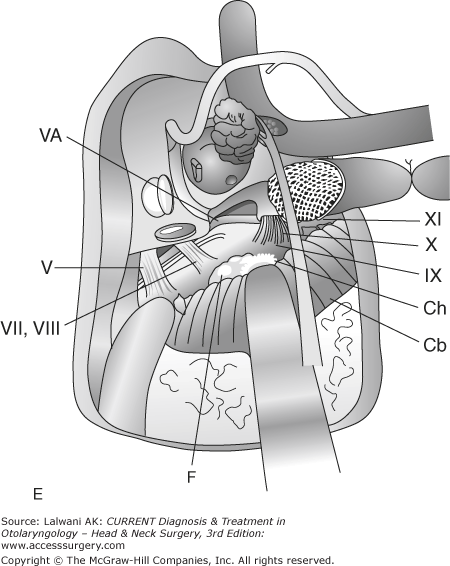
Figure 65–1.
Surgical resection of a large jugulotympanic paraganglioma (Fisch Type A approach). (A) The incision runs from above the ear down into the upper neck. (B) The external auditory canal is oversewn and the pinna reflected anteriorly, exposing the facial nerve, middle ear, parotid gland, internal carotid artery, and internal jugular vein. (C) The facial nerve is rerouted out of its bony canal and transposed anteriorly. (D) The sigmoid sinus is occluded superiorly and the internal jugular vein is ligated inferiorly; the tumor is then removed from the jugular foramen. Although the classic Fisch Type A approach involves closure of the external auditory canal and rerouting of the facial nerve, these procedures are not often required to resect even large jugular foramen tumors as shown in this example. ICA, internal carotid artery; TM, tympanic membrane; SCC, semicircular canals; P, parotid gland; SS, sigmoid sinus; FN, facial nerve; IJV, internal jugular vein. Roman numerals indicate cranial nerves; IPS, inferior petrosal sinus. (E) If necessary, any remaining tumor is removed off from the internal carotid artery or cerebellopontine angle. Fisch Type B and C approaches (not shown) are not used to approach the jugular foramen, but instead are used to approach tumors of the infratemporal fossa, petroclival junction, and nasopharynx. VA, vertical artery; CH, choroid plexus; CB, cerebellum.
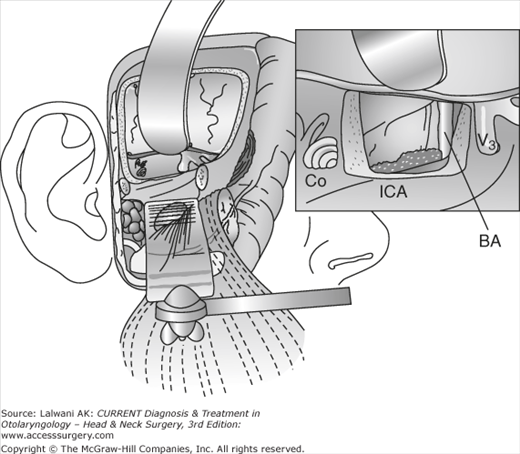
Figure 65–2.
Middle fossa-transpetrous approach for resection of petrous apex tumors. These tumors can grow to involve the petroclival junction, around foramen lacerum, and extend posteriorly into the ventral brainstem and superiorly into the temporal lobe. They can be approached by a middle fossa craniotomy with drill-out of the anterior petrous apex (Kawase triangle). Good visualization of the petroclival junction, as well as the anterior brainstem, is obtained by this approach. Co, cochlea; BA, basilar artery; ICA, internal carotid artery.
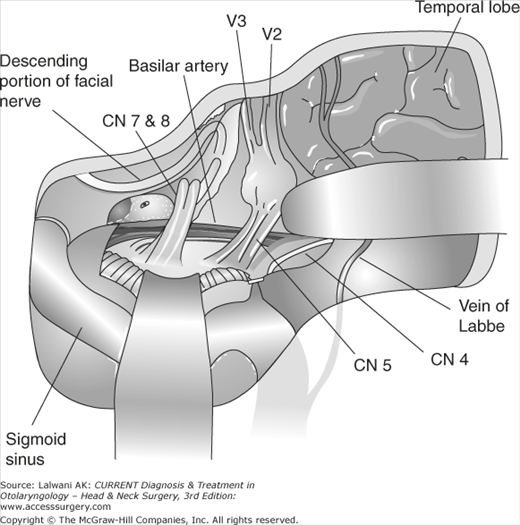
Figure 65–3.
Combined subtemporal-retrolabyrinthine approach for resection of tumors of the petroclival junction. This involves opening both the posterior and middle cranial fossa dura, with division of the tentorium. Excellent exposure of the entire brainstem from the posterior circle of Willis to the jugular foramen is obtained.
It is also important to consider the roles of conservative observation, particularly in patients who are elderly and have tumors that are typically slow growing. Stereotactic radiation must also be considered as a viable treatment option to slow or stop tumor growth. Modern techniques of delivering radiation appear to have excellent tumor control rates and side effect profiles, although the long-term consequences have yet to be fully elucidated.
Paragangliomas
Paragangliomas (or glomus tumors) are tumors of paraganglionic tissue, which originally derive from the migration of neural crest cells during fetal development. These tissue rests are distributed predominantly throughout the middle ear, the jugular foramen, the vagus nerve, and the carotid body, but are also found in the upper mediastinum and the retroperitoneum. These cell clusters are innervated by the parasympathetic nervous system and function as chemoreceptors for circulatory regulation.
The most common paraganglioma is the carotid body tumor. A well-known, but rare, paraganglioma is the pheochromocytoma. Within the temporal bone, there are two main types of paraganglioma: glomus tympanicum and glomus jugulare. Glomus tympanicum tumors arise within the middle ear, from paraganglionic cell rests associated with branches of cranial nerves IX and X (the glossopharyngeal and vagus nerves, respectively), which run over the promontory. Together these nerves are called the tympanic plexus, which consists of the Jacobsen nerve (a branch of CN IX) and the Arnold nerve (a branch of CN X). Glomus jugulare tumors arise within the jugular foramen from cell rests associated with cranial nerves IX, X, and XI. Glomus vagale tumors are just below the temporal bone in the upper neck and may invade the skull base as well.
Paragangliomas are most common in white populations. They typically occur in the fourth or fifth decades of life, although they can be identified at any age. Paragangliomas are slow-growing tumors, and metastases are extremely rare. They grow by spreading along the paths of least resistance. Within the skull base, they tend to extend through fissures and foramina, vascular channels, and air-cell tract lines. Paragangliomas also demonstrate locally aggressive behavior with bone destruction and the invasion of soft tissue. They can extend from the temporal bone down into the upper neck.
Approximately 1% of paragangliomas display functionally significant catecholamine secretion similar to a pheochromocytoma. Pathologically, the chief cell is the cell of origin of the tumor and contains acetylcholine, catecholamines, and serotonin. Classic findings are clusters of chief cells, termed Zellballen, with a rich vascular plexus throughout the entire tumor. Indeed, these tumors are highly vascular and may bleed substantially during surgical excision.
The overall incidence of multiple lesions is about 10% in sporadic tumors. There is a 1–2% incidence of bilateral glomus jugulare tumors and a 7% incidence of an associated carotid body tumor. Paragangliomas can be based on germline mutations and can be hereditary. Mutations in the mitochondrial complex II genes SDHB, SDHC, and SDHD cause hereditary paragangliomas. In addition, another form of the disease has an autosomal dominant mode of transmission, and the causative genetic defect has been localized to two separate loci: 1lq13.1 and 11q22–23. Patients with hereditary disease display a much higher incidence of synchronous paraganglioma, approximately 25–35%.
Paragangliomas are also associated with phakomatoses (neurologic diseases with cutaneous manifestations). These include von Recklinghausen neurofibromatosis, Sturge–Weber syndrome, tuberous sclerosis, and von Hippel–Lindau disease. In addition, they can be associated with multiple endocrine neoplasia Type I syndrome.
There are two main classification schemes for paragangliomas of the temporal bone: Fisch and Glasscock–Jackson.
The Fisch classification includes four main categories: (1) Type A (tumors limited to the middle ear), (2) Type B (tumors limited to the tympanomastoid area), (3) Type C (tumors extending into the petrous apex), and (4) Type D (tumors with intracranial extension).
The classification scheme of Glasscock and Jackson differentiates between glomus tympanicum and glomus jugulare tumors.
For glomus tympanicum neoplasms, this staging system includes (1) Type I (small masses limited to the promontory of the middle ear), (2) Type II (tumors filling the middle ear space), (3) Type III (tumors filling the middle ear and mastoid), and (4) Type IV (tumors extending into the external auditory canal or around the internal carotid artery).
For glomus jugulare tumors, this staging system includes (1) Type I (small tumors involving the jugular bulb, the middle ear, and the mastoid), (2) Type II (tumors extending under the IAC), (3) Type III (tumors extending into the petrous apex), and (4) Type IV (tumors extending beyond the petrous apex into the clivus or infratemporal fossa).
The two most common presenting symptoms of a patient with a paraganglioma of the temporal bone are conductive hearing loss and pulsatile tinnitus. Patients may also complain of aural pain, facial nerve weakness, and a neck mass. The patient should be questioned as to symptoms of sympathetic discharge, which may represent a functionally secreting tumor, such as tachycardia, arrhythmias, flushing, or labile hypertension. Moreover, the patient should be queried about any symptoms of dysphagia or hoarseness, which may represent palsy of cranial nerves IX or X.
Physical examination demonstrates a reddish–bluish mass behind the eardrum. An aural polyp may be noted. There are two clinical signs associated with paraganglioma that can be identified during microscopic exam of the tympanic membrane: (1) brown sign is the cessation of tumor pulsation and tumor blanching with positive pressure using the pneumatic otoscope; and (2) Aquino sign is the blanching of the mass with manual compression of the ipsilateral carotid artery. A complete examination of the cranial nerves is indicated, with particular attention to cranial nerves VII (the facial nerve), VIII (the vestibulocochlear nerve), IX (the glossopharyngeal nerve), X (the vagus nerve), XI (the accessory nerve), and XII (the hypoglossal nerve).
Ascultation of the ear and upper neck can be performed with a stethoscope if a patient complains of pulsatile tinnitus. Pulsatile tinnitus from a vascular tumor gets louder and faster when the patient exercises. Therefore, having the patient jog in place or go up and down some steps facilitates the ability of the clinician to hear the bruit.
Patients with a suspected paraganglioma can be screened for a catecholamine secretion by collecting a patient’s urine for 24 hours and determining the vanillylmandelic acid and metanephrine levels. An audiogram will reveal conductive hearing loss if the middle ear space is invaded with tumor. If the inner ear is invaded, a sensorineural hearing loss will be found. Impedance audiometry will reveal a flat tympanogram if a middle ear mass is present and touches the eardrum. Occasionally, vascular pulsations may be noted on the tympanogram.
The importance of imaging paraganglioma of the skull base cannot be underestimated (Figure 65–4). Imaging is critical to delineate the extent of these tumors precisely. The first thing to determine is whether the jugular foramen is involved with the tumor. A glomus tympanicum is limited to the promontory and the mastoid though a glomus jugulare begins in the jugular foramen and extends superiorly into the middle ear and mastoid. In addition, the studies should be reviewed with careful attention to the middle ear, jugular foramen, and carotid bifurcation to look for a synchronous tumor.
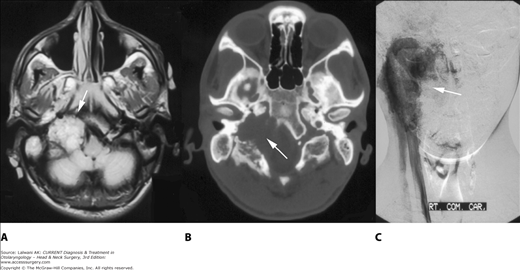
Figure 65–4.
Jugulotympanic paraganglioma. (A) Axial view, T1-weighted image MRI with gadolinium contrast demonstrates a large enhancing mass in the right jugular bulb extending intracranially into the cerebellopontine angle (arrow). (B) Computed tomography at the same level shows significant bone destruction of the temporal bone and clivus (arrow). (C) Coronalview angiogram demonstrates the highly vascular tumor extending from the skull base inferiorly into the upper neck. This mass was embolized prior to surgical excision.
CT scanning is useful to visualize the bony structures of the temporal bone. Of key importance is to evaluate the bone above the jugular bulb, the jugular plate. If the tumor is a glomus jugulare tumor that has extended into the middle ear cavity, this bone will be eroded. In contrast, if the tumor is a glomus tympanicum tumor, the bone surrounding the jugular bulb is usually intact. Inner ear or facial nerve involvement may also be noted. There may be a semicircular canal fistula or the tumor may be in close proximity to the fallopian canal, particularly along the vertical segment. The tumor may extend anterior to the IAC or along the petrous portion of the internal carotid artery. These findings may affect the planned surgical approach.
MRI studies are useful to identify whether there is intracranial extension of the tumor. MRI gives excellent soft tissue contrast resolution and permits delineation of the tumor from the brainstem, the cerebellum, and the cranial nerves. The tumor has intermediate signal intensity on T1-weighted MRI and high intensity on T2-weighted MRI. Both may demonstrate a speckled pattern within the tumor, termed as “salt and pepper” pattern. This pattern is due to flow voids from the large number of intratumoral blood vessels. The tumor enhances strongly with gadolinium contrast.
Magnetic resonance angiography can be used to evaluate for compression of the internal carotid artery. Magnetic resonance venography is useful to assess collateral circulation within the dural sinuses of the skull, since blood flow within the sigmoid sinus is often blocked by the tumor.
Angiography of glomus jugulare tumors is usually done 1 or 2 days before surgical excision. This permits definitive diagnosis of the tumor by visualizing the tumor blush characteristic of such highly vascular tumors. In addition, the feeding vessels can be identified and embolized to reduce blood loss during surgery. The typical feeding vessels for a glomus jugulare tumor are the ascending pharyngeal artery and the stylomastoid branch of the occipital artery. Glomus tympanicum tumors typically do not need to be embolized preoperatively because of their small size and easy accessibility.
The differential diagnosis of a patient with a middle ear mass includes otitis media, cholesterol granuloma, other types of middle ear neoplasms–including middle ear adenoma or carcinoma–a vascular anomaly such as a high-riding dehiscent jugular bulb, an aberrant carotid artery, or a persistent stapedial artery. Other temporal bone neoplasms that might involve the middle ear space include meningiomas, schwannomas or neuromas, adenomas, or endolymphatic sac tumors.
Progressive conductive hearing loss usually is the presenting symptom of patients with a temporal bone paraganglioma. This may occur directly if the tumor contacts the ossicular chain or indirectly if the tumor blocks the eustachian tube, producing a serous middle ear effusion. Sensorineural hearing loss is uncommon but can occur if the tumor erodes the dense otic capsule bone and invades the inner ear. Alternately, the tumor may extend intradurally and affect cranial nerve VIII in the cerebellopontine angle and IAC.
Paragangliomas of the temporal bone may cause facial nerve palsy (21%) by invading the nerve within the temporal bone. Usually, this occurs along the vertical portion of the nerve within the mastoid. Even if nerve function is unaffected, most glomus jugulare tumors grow to wrap around the facial nerve and erode its bony canal in this location. A microsurgical dissection of a dehiscent nerve surrounded by tumor is the norm and can be quite challenging. (A dehiscent nerve is one in which the bony canal surrounding the nerve has been eroded.)
If the jugular foramen is involved with the paraganglioma, the insidious onset of neuropathy of the lower cranial nerves (IX, X, and XI) ensues as they are slowly encroached upon by the tumor. Symptoms include dysphagia and aspiration, as the sensation to the pharynx (CN IX) and the larynx (CN X) is diminished. Also, hoarseness may be noted owing to vocal cord paralysis (CN X). It should be noted that rather than an isolated recurrent laryngeal nerve injury that causes vocal cord paralysis (such as with a Pancoast tumor), the jugular foramen syndrome includes a high vagal nerve injury. This is more severe because the combination of a lack of sensation to the upper larynx and vocal cord paralysis puts these patients at extremely high risk of aspiration. The paralysis of cranial nerve XI can be noted as weakness and atrophy of the sternocleidomastoid and trapezius muscles.
The hypoglossal nerve exits the skull base through the hypoglossal foramen in the occipital bone, anteroinferior to the jugular foramen. Large paragangliomas that extend inferiorly may affect the hypoglossal nerve. The patient may complain of worsening articulation, and the physical exam will demonstrate ipsilateral tongue atrophy, muscular fasciculations, and deviation to the affected side with protrusion.
The sympathetic nerves to the head run from the superior cervical ganglion up along the internal carotid artery into the skull base. Paragangliomas that envelop the petrous portion of the internal carotid artery may cause an ipsilateral Horner syndrome with ptosis, miosis, and ipsilateral facial flushing and sweating.
Large paragangliomas can affect other neurologic functions, depending on the tumor extension. Intradural tumors can grow within the cerebellopontine angle, producing cerebellar dysfunction and imbalance, brainstem compression, and even obstructive hydrocephalus. Tumors that grow superiorly or medially can affect other cranial nerves, causing diplopia (CN IV or VI), facial numbness or pain (CN V), or dry eye (the greater superficial petrosal branch of CN VII).
Observation with no treatment is reasonable in patients with minimal symptoms, particularly if they are older. Because glomus tumors are slow growing, serial MRI scans can be obtained, reserving surgery or radiation therapy for obvious tumor growth. This approach is less acceptable for younger patients in whom the tumor would be expected to grow substantially during their life span.
The role of radiation therapy in the management of paragangliomas is controversial. Radiation is thought to reduce the growth rate of these tumors; however, it does not eliminate viable tumor cells within the mass. Tumors have been known to recur even more than a decade after radiation therapy. Radiation therapy for paragangliomas of the temporal bone can be useful as a treatment for elderly patients with symptomatic tumors or for patients who are unwilling to undergo a surgical resection. Postoperative stereotactic radiation therapy may be used for patients in whom total tumor removal could not be achieved.
Microsurgical total tumor removal is the treatment of choice for most patients. Patients with functionally secreting tumors need to be alpha-blocked with phentolamine before and during surgical resection to prevent life-threatening hypertension as the alpha-adrenergic hormones are released with tumor manipulation.
The surgical approach for resection of paragangliomas of the temporal bone depends on the tumor extent. For a glomus tympanicum tumor that is limited to the middle ear cavity, a simple middle ear exploration through the ear canal may be all that is indicated. After raising the tympanic membrane, the tumor can be visualized on the promontory. It may then be cauterized with a bipolar cautery and removed. If the tumor is larger and extends into the mastoid air cells, a tympanomastoidectomy with an extended facial recess approach may be required. This is a standard mastoidectomy via a postauricular incision with sacrifice of the chorda tympani nerve to allow exposure of the middle ear and hypotympanum from the mastoid. A tumor extending medially to the facial nerve (the retrofacial air cells) can be resected after exposing the facial nerve along its vertical segment to prevent injury to it.
For glomus jugulare tumors, a larger surgical approach is required. One very important aspect during the removal of these tumors is delineation and preservation of the facial nerve. Unfortunately, the vertical segment of the facial nerve lies in the middle of the operative field, and the tumor is usually based directly behind it, wrapping around it. A tympanomastoid approach with an extended facial recess and complete skeletonization of the facial nerve to the stylomastoid foramen typically provides adequate exposure. If possible, the preservation of a thin layer of bone surrounding the facial nerve circumferentially is ideal to minimize risk to the facial nerve (the fallopian bridge technique). It is also important to extend the skin incision into the neck and identify the internal carotid artery and internal jugular vein. The sternocleidomastoid and digastric muscles are separated from the mastoid tip so that the great vessels can be followed up to the skull base. These vessels need to be controlled both proximally and distally to the tumor in case a great vessel rupture occurs.
The most important part of the surgery is resection of the jugular bulb. Superiorly, the sigmoid sinus is occluded in the mastoid cavity, inferior to the junction of the transverse sinus and sigmoid sinus because the vein of Labbé enters at that location. Occlusion of the vein of Labbé may cause venous infarction of the temporal lobe since it is the only vein draining this territory. Inferiorly, the internal jugular vein is divided and ligated in the neck. Next, the jugular bulb (the lateral wall of the sigmoid sinus and the tumor filling the sinus) is dissected from the posterior fossa dura and cranial nerves IX, X, and XI. There is usually substantial bleeding from the entry point of the inferior petrosal sinus to the jugular bulb during this process. It is important to quickly remove this tumor and pack this area with an absorbable knitted fabric (eg, Surgicel) to control the bleeding. After tumor removal, the mastoid cavity is often packed with fat harvested from the abdominal wall and closed in layers.
Large glomus jugulare tumors, which extend anteriorly along the internal carotid artery, typically require a larger infratemporal fossa surgical approach (Fisch Type A, see Figure 65–1
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree