Fig. 10.1
An example of national registry data from the Australian Corneal Graft Registry (http://hdl.handle.net/2328/25860). Long-term survival of penetrating keratoplasty, lamellar keratoplasty and limbal allografts (Reproduced with permission of the Australian Corneal Graft Registry)
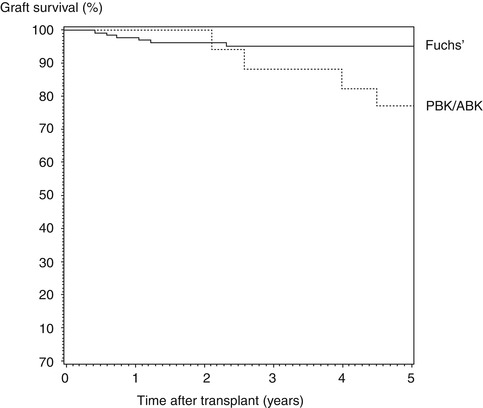
Fig. 10.2
An example of single-centre registry data from the Cornea Research Foundation of America (www.cornea.org) showing survival of endothelial keratoplasty for Fuchs endothelial dystrophy and pseudophakic/aphakic bullous keratopathy (Reproduced from Price et al. [19], with permission of Elsevier)
Setting Up a Registry
Registry design, extent and mode of operation will be influenced by multiple factors ranging from individual surgeon preferences through to the availability of funding and appropriate infrastructure at local, regional and national levels. There is no ‘best’ way to organize a corneal transplant registry, which could be achieved through professional or academic organizations as well as with government support. Keys to success include: active surgeon involvement and commitment to help define the clinical questions to be answered (which are likely to change with time) and to provide clinical data on their patients, expert statistical advice at all stages, staff to support and maintain the registry and an appropriate platform for the storage and analysis of clinical follow-up data. Other considerations will include the ethical and regulatory environments existing in different countries, for example, patient confidentiality, data protection and issues of consent for the storage of data for the greater good rather than being specifically linked to the treatment of an individual patient.
The three aforementioned national registries reflect these varying circumstances and, as a result, operate in different ways; for example, the Australian Corneal Graft Registry (ACGR) (http://hdl.handle.net/2328/25860), the oldest and largest of the three, which is based in an academic department, has been collecting data since May 1985 and can report very long-term survival data extending for more than 20 years [25]. The Swedish and UK registries both limit the follow-up of transplants to 2 and 5 years, respectively [2, 6]; however, longer-term follow-up studies are possible by collecting additional data on selected groups of patients rather than through continual collection of follow-up data on all patients without limit [4]. Not surprisingly, loss to follow-up, especially among the older patient population, becomes an increasingly important factor with extended follow-up times. Kaplan-Meier survival curves are used extensively for the analysis of graft survival [1]. Unfortunately, the right-hand side of Kaplan-Meier survival curves becomes increasingly less reliable at longer postoperative times as the numbers of transplants at risk declines through loss to follow-up and graft failures; moreover, the assumption that transplants lost to follow-up would have behaved in the same way as those available for examination becomes ever more uncertain.
In Sweden, there is substantial government support available for clinical registries across a range of clinical specialties. EyeNet Sweden (www.eyenetsweden.se), which was established in 2003, hosts the Swedish Cataract Registry [3] and the Swedish Cornea Registry (www.cornea.nu), which began collecting data in 1996. (NB The EyeNet website includes advice, in English, on setting up quality registers.) The Swedish registry collects data at two time points, viz., at the time of transplant and at 2 years postoperatively. In 2007, the Swedish Cornea Registry abandoned paper-based data submission and implemented a web-based application through EyeNet with direct online data entry by each individual clinic. This greatly reduced the risk of transcription errors and improved the accuracy of the data held in the registry. The participating clinics, which include transplant units in Denmark and Norway, can access their own data on line and compare their outcomes and activity directly with national data.
The UK Transplant Registry is also supported by government funding. It is maintained by NHS Blood and Transplant (NHSBT) and holds national outcome data both for solid organ and corneal transplants. Data are collected at the time of surgery and then at 1, 2 and 5 years postoperatively using standardized follow-up forms; however, a move to online input of data is, at the time of writing, under discussion. This registry has the added benefit in that it includes donor and eye bank information for each cornea transplanted. As a result, it enables robust traceability between donors and recipients for the purposes of investigating serious adverse events and reactions in corneal transplant recipients, thus meeting the regulatory requirements set out in the EU Tissues and Cells Directive (2004/23/EC) and its accompanying Commission Directives (2006/17/EC and 2006/86/EC) (http://ec.europa.eu).
Uses of Data from Corneal Transplant Registries
Graft Survival
Because of their size, national registries provide a broader perspective than single-centre studies and can provide information not necessarily available through other means, for example, the routine monitoring and analysis of rare events and complications including primary graft failure and postoperative endophthalmitis. Registry data can be used to monitor transplant activity and outcomes, trends and patient demographics, which are not just of interest to surgeons but are important for informing the development of healthcare policies and resource planning. The data are an invaluable resource for clinical research, enabling large-scale studies that improve our understanding of the factors that influence corneal transplant outcomes. Indeed, registry data can help identify questions that would be best answered by controlled clinical trials and provide supporting information for their planning and design.
Graft survivals reported in the registries are very similar (e.g. see Table 10.1 comparing Australian and UK data). All three registries have confirmed that the indication for transplantation, preoperative risk factors, such as vascularization and glaucoma, and postoperative complications, such as rejection, are the major factors influencing graft survival after penetrating keratoplasty (PK) [2, 6, 23, 25]. Registries also provide information relevant to the postoperative management of graft patients; for example, studies using UK Transplant Registry data have demonstrated that long-term topical steroid use reduces the risk of graft failure after PK for pseudophakic bullous keratopathy [20] and that oral antiviral treatment is more effective than topical treatment for reducing the risk of graft failure in patients undergoing PK for herpetic keratitis [10] (Fig. 10.3). The ACGR has also highlighted the negative impact of reversed rejection episodes on long-term graft survival: at 10 years, overall survival of transplants that have experienced no rejection is 68 % compared with just 35 % for those that have suffered one or more rejection episodes [8, 25].
Table 10.1
Five-year graft (PK) survival by indication from the Australian Corneal Graft Registry (ACGR) and UK Transplant Registry (UKTR)
5-year graft survival | ||
|---|---|---|
Indication | ACGR (%) | UKTR (%) |
Keratoconus | 95 | 93 |
Corneal dystrophy | 82 | 80 |
Bullous keratopathy | 56 | 59 |

Fig. 10.3
UK Transplant Registry data showing the influence of oral vs. topical antiviral medication on 5-year graft survival (Reproduced from Goodfellow et al. [10], with permission of Nature Publishing Group)
Visual Outcome
The majority of corneal transplants are performed to improve a patient’s vision. It is therefore of value to be able to use registry data to assess outcomes in terms of vision rather than simply graft survival. The Swedish Cornea Transplant Registry was started in 1996 with the principal aim of reporting visual outcomes [6] (Fig. 10.4). These analyses have shown the dependence on indication of the expectations for postoperative visual rehabilitation, principally visual acuity (VA). While >80 % of patients had a preoperative VA ≤0.2 across all indications, almost 80 % of patients with grafts for keratoconus achieved a VA of ≥0.5 at 2 years after surgery compared with just over 50 % of grafts for Fuchs and only 20 % for bullous keratopathy. These results do not take into account the increasing occurrence of co-morbidity, such as retinal disease, in the older patients; however, this information is collected by the Swedish registry and can be included as a variable in analyses of visual outcome. Interestingly, for regrafts, the respective percentages of grafts achieving ≥0.5 VA for keratoconus and Fuchs were somewhat reduced at 55 and 19 %, respectively, compared with first grafts, while the outcome for bullous keratopathy regrafts was less affected but still reduced to about 10 % [5].
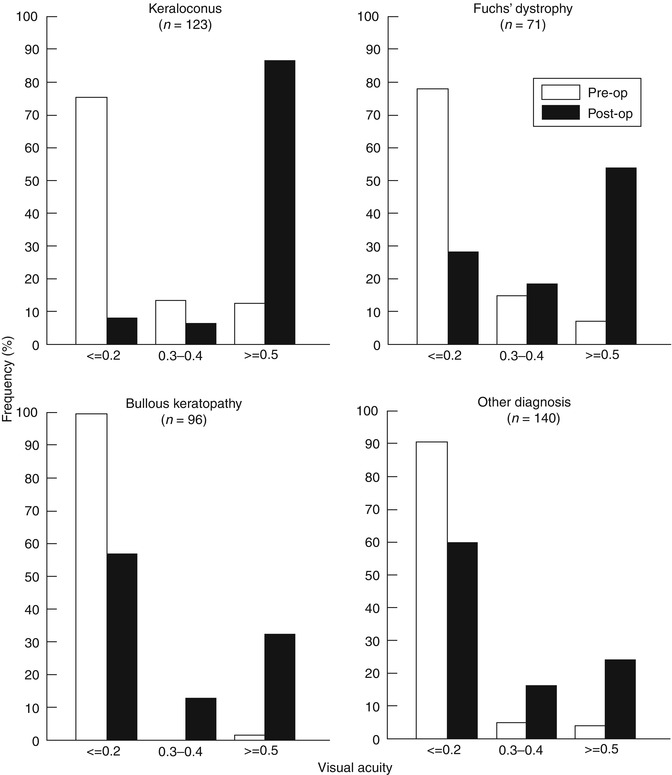

Fig. 10.4
Visual outcome data from the Swedish Corneal Transplant Registry showing percentages of grafts achieving VAs of ≤0.2, >0.2 and <0.5, and ≥0.5 preoperatively and at 2-year postoperative for keratoconus, Fuchs endothelial dystrophy, bullous keratopathy and ‘other’ indications (Reproduced from Claesson et al. [6], with permission of BMJ Publishing Group)
Patient-Reported Outcome Measures (PROM)
Registries and single-centre studies typically focus on clinical outcome measures (COM), such as graft survival, complications (e.g. rejection episodes) and visual outcome. There are, however, few studies of the impact of corneal transplantation on self-assessed, patient-reported outcome measures (PROM). These aim to determine improvement or otherwise in visual disability as perceived by transplant recipients. An early study from the ACGR looked at this important aspect of corneal transplant outcome [24], but little attention has since been paid to this area. The Swedish Cataract Register has conducted widespread studies on PROMs and compared them to COMs [14]. These studies have used the Catquest 9-SF visual disability instrument, which consists of just 9 questions that patients complete before and after surgery [13] (Table 10.2). The answers are resolved by Rasch analysis into a single score of visual disability (high scores equate to greater disability), which are amenable to parametric statistical analysis. A study is currently underway in Sweden to complete the validation of Catquest 9-SF for corneal transplant recipients and to apply this visual disability instrument to determine the factors that most influence PROMs as opposed to COMs. Patients will be asked to complete the questionnaire before surgery and at 2 years after surgery (Claesson M, personal communication, 2014).
Table 10.2




Catquest 9-SF visual disability questionnaire
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


