Multifocal, Accommodative, and Innovative Intraocular Lenses
Robert J. Schechter
Vinay Gutti
Marjan Farid
Widespread use of intraocular lenses (IOL) has changed the face of cataract surgery in the past decades. Optic rehabilitation of the patient after surgery has eliminated the handicaps and inconveniences of spectacle and contact lens correction. The conventional IOL, however, is a fixed-focus device currently not capable of accommodation-like changes in shape or power. It is also a single-focus device, restricting the practitioner to select only a single power for a particular patient. Technologies that would allow a greater range of visual focus from near to distance would be of interest to practitioner and patient alike. Currently, two general approaches to this problem are available. One is an IOL that can change its effective power, either by modifying its shape, index of refraction, or position in the eye (accommodating or pseudoaccommodating). The other is an IOL that incorporates more than one focal plane in its imaging (multifocal). The multifocal IOL differs from bifocal or multifocal optical correction of spectacles in that there is no movement of the IOL perpendicular to the optical axis. Thus, within limits, the multiple foci are imaged onto the retina at the same time.
Cataract surgery and implantation of intraocular lenses (IOLs) is becoming more of a refractive procedure where clinical outcome and patient satisfaction are measured not only by visual acuity but also the quality of vision. As a result, the development and continued innovations in IOL design technology and materials are considered some of the most important ophthalmologic advances in recent decades. These innovations as well as the use of sophisticated formulas to calculate the corrective prescription power of the lens and improved surgical technique have resulted in the realistic possibility of patients seeing across a wide range of distances without the need for spectacles or contact lenses with minimal postimplantation recovery or adaptation time.
Currently available IOLs can be differentiated based on their optic design. Nearly a decade ago the first multifocal lens (Array, AMO) was approved for use by the U.S. Food and Drug Administration (FDA). Modifications to the multifocal optics as well as materials have resulted in several second-generation multifocal lenses. In addition, accommodative IOLs are entering the market, and several IOLs with innovative optics are in various stages of development. Noteworthy in the changing face of refractive and cataract surgery was the finding of the 2004 survey by the U.S. members of the International Society of Refractive Surgery of the American Academy of Ophthalmology1: For the first time, a total of 8% of respondents believed it was acceptable to perform bilateral phakic IOL implantation at the same surgical visit.
OPTICS OF MULTIFOCAL, ACCOMMODATIVE, AND INNOVATIVE IOLS
Historically, conventional IOLs were fixed-focus devices that required the practitioner to select only a single distance power for a particular patient. These devices were not capable of accommodative-like changes in shape or power and, although patients had good distance vision, spectacles were typically needed for near vision activities.
In 1997 the FDA approved the Array SA40 N multifocal IOL, an event that heralded a new era in the field of presbyopic correction. The innovative design of the multifocal Array lens provided patients with distance visual acuity similar to that of traditional monofocal lenses with the added benefit of better near vision and depth of focus2,3,4,5 and reduced need for spectacles.6,7
In recent years, modifications to the multifocal design platform established by the Array lens have led to the release of several second-generation multifocal IOLs available in the United States (Table 1) and others in other countries. Albeit having different optics and designs, these multifocal lenses are similar in that they are designed to increase the patient’s range of focus to include objects at near, intermediate, and far distances.
Table 1. Design Features of Selected Multifocal and Accommodative IOLs | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||
The mechanism of diffractive lenses is based on the Huygens-Fresnel principle.1 This states that every point of a wavefront can be thought of as being its own source of secondary so-called wavelets, subsequently spreading in a spherical distribution. The amplitude of the optic field beyond this point is simply the sum of all these wavelets. When a portion of a wavefront encounters an obstacle, a region of the wavefront is altered in amplitude or phase, and the various segments of the wavefront that propagate beyond the obstacle interfere and cause a diffractive pattern. A regular array of obstacles that has the effect of producing periodic alterations in the emerging wavefront is termed a diffraction grating. As the spacing between the diffractive elements decreases, the spread in the diffractive pattern increases. By placing the diffractive microstructures in concentric zones and decreasing the distance between the zones as they get further from the center, a so-called Fresnel zone plate is produced that can produce optic foci. Thus, the distance power is the combined optic power of the anterior and posterior lens surfaces and the zero order of diffraction, whereas the near power is the combined power of the anterior and posterior surfaces and the first order of diffraction.
Diffraction can be interpreted as a spreading of wavefronts. If the scattered waves are in phase, they interfere constructively; if they are out of phase, they interfere destructively. By assigning appropriate dimensions for the 20 to 30 concentric zones on the posterior lens surface, approximately 41% of the light is in phase and focused for near vision, about another 41% is in phase and focused for distance vision, and the remaining 18% is lost.2 This diffractive optic effect is present at all points (or more precisely, at all sufficiently large areas) of the lens. This means that the multifocal performance of this lens should be unaffected by decentration, pupil eccentricity or deformation, or pupil size; distance and near vision are possible if any part of the lens optic is present behind the pupil.
Conversely, refractive lenses use only differing areas of refractive power to achieve their multifocality. They function by providing annular zones of different refractive power to provide appropriate focus for objects near and far. These lenses may have discrete zones of a near or distant power; thus they may be said to be bifocal. Others may have aspheric surfaces with transition zones resulting in powers that may vary continuously between a minimum for distance and a maximum for the strongest near add. These may be said to be multifocal. Refractive bifocal or multifocal IOLs can be affected by pupil size and decentration, to a greater or lesser degree depending on the size, location, and number of refractive zones. Postoperative pupil size might be a factor of importance in selecting which lens to use. One team investigated whether they could predict postoperative pupil size from preoperative factors.3 They concluded that they could not predict the pupil size with sufficient consistency to ensure a good match between pupil diameter and the zone sizes of various multifocal IOLs. There are now multiple different approaches to create “multifocality” or its equivalent in IOLs (Table 1).
The optics of newer multifocal lenses produce multiple foci using either refractive (ReZoom, AMO, Santa Ana, CA) or diffractive principles, with some incorporating apodized diffractive (AcrySof ReSTOR, Alcon Laboratories Inc., Fort Worth, TX) technologies. The concept of apodization is that of a gradual transition of optical properties from the center of a lens to the edge. Refractive or diffractive optic designs offer the ability to manage light delivered to the retina, distributing the appropriate amount of light to near and distant focal points without mechanical movement of the lens. Other multifocal IOL technologies include aspheric lenses (Tecnis ZM001, AMO) that reduce spherical aberrations of the eye and improve functional vision in varying light conditions.
There is another approach to correct the problem of the monofocality of the standard IOL. Accommodative lenses, such as the Crystalens, Synchrony, and the 1CU incorporate mechanical designs that allow movement of the optic and focusing ability through a range of distances.
Other techniques are in development and involve using a thermoplastic acrylic gel that unfolds into a full-size flexible lens that fills the capsular bag when in the aqueous environment at body temperature.8 This lens will be further discussed later in the chapter. Another technology being explored is a light-adjustable lens composed of a macromer matrix. The desired curvature or power can be adjusted, and then the matrix polymerizes and solidifies under ultraviolet radiation.9,10
CHARACTERISTICS AND COMPARISONS OF MULTIFOCAL AND ACCOMMODATIVE IOLS
MULTIFOCAL IOLS
Multifocal IOLs have at least two coaxial dioptric powers, usually separated by a dioptric interval, such as 4 diopters (D), to provide a refractive dioptric interval, such as 3 D, at the spectacle plane. On the retina, these powers produce two superimposed images of any observed object. Even under the best circumstances, one image will be sharply focused and the other blurred by the defocus aberration. For example, a black line on white paper will appear surrounded by a narrow grey ribbon. This simultaneous projection of in- and out-of-focus images of the same object on the retina, however, produces the reduction in modulation transfer function historically noted with multifocal IOL implantation.11 Retina or neural processing of these out-of-focus images can lead to the patients’ perception of halos around bright images at nighttime.12 The contrast between dark and light is enhanced under scotopic conditions, and thus the haloes are more pronounced at nighttime. The lower optical quality in comparison with monofocal IOLs results as a lower contrast sensitivity in patients implanted with the multifocal IOL. This perception of haloes and glare tends to decrease over time with neural adaptation.
Overall, the inherent optical effects of multifocal IOLs, compared with monofocal IOLs, generally results in similar distance visual acuity with increased near and intermediate visual acuity. This was noted in a Cochrane Review article13 of eight randomized, controlled trials. Review of the trials found that unaided distance acuity was similar in the those implanted with multifocal or monofocal lenses (Peto odds ratio [OR] 1.21, 95% confidence interval [CI] 0.75–1.96) with no statistical difference between multifocal IOL and monofocals with respect to the proportion of participants achieving 6/6 best corrected visual acuity (OR 1.43, 95% CI 0.99–2.09). In those with implanted multifocal lenses, unaided near vision was improved as compared with those with monofocal lenses implanted, and a higher percentage of patients with multifocal implants reported total freedom from spectacles (OR for spectacle dependence 0.15, 95% CI 0.11–0.22). Glare and halos were reported in those with multifocal lenses implanted.
As compared with the first-generation multifocal IOLs, the optics of newer multifocal IOLs attempt to mitigate many of the optical trade-offs by directing different amounts of the refracted light on the different foci, with distance-dominant multifocal IOLs providing higher contrast sensitivity for distance focus and lower contrast sensitivity for near focus with the contrary occurring in near-dominant multifocal IOLs. Additionally, newer designs draw on the pupil to drive different amounts of light on the different foci depending on pupil dilation.
ARRAY
Optic Characteristics
The Array multifocal IOL (Fig. 1) is a distance-dominant, simultaneous-vision, zonal-progressive lens. It combines a posterior refractive surface with multiple anterior aspheric refractive rings, or zones, of continuously varying power. Beginning with the 2.1-mm central zone, distance power is placed centrally in each odd ring (1, 3, and 5). The power gradually increases toward the periphery of the odd-numbered rings to form a smooth transition with the even zones that emphasize near vision with a 3.5-D add. This provides continuous refractive power across the lens to minimize dependence on pupil size. As compared with a monofocal IOL, the Array directs 42% (miosis) to 73% (mydriasis) of light onto the distance focus.14
Clinical Studies
Numerous studies have compared the safety and visual acuity of the Array with traditional monofocal lenses and found the two groups to be at least comparable6,15,16,17 and others reporting better near vision and greater depth of focus with the Array than with monofocal lenses.2,3,4,5 Several studies have also reported a less frequent use of spectacles for near vision activities in those having the Array implant than in those with a monofocal lens implant6,16,17 and fewer limitations in their overall ability to perform social and daily functioning activities without eyeglasses.6,7 Studies evaluating patient satisfaction and visual acuity following simultaneous bilateral implantation of the Array18 were comparable to those seen after sequential surgery.19
REZOOM
Optic Characteristics
Modifications to the refractive multifocal design platform of the Array multifocal IOL led to the development and, in March 2005, the FDA approval of the ReZoom multifocal IOL. The ReZoom is considered a second-generation multifocal IOL in that its refractive multifocal design was enhanced to improve optical performance while providing good distance, intermediate, and near visual acuity (Fig. 2). The ReZoom is a flexible three-piece lens with an overall diameter of 13 mm, a 6-mm diameter lens optic, and polymethylmethacrylate (PMMA) haptics that are in a modified C configuration. It is designed to be implanted in the capsular bag with minimal risk of decentration.
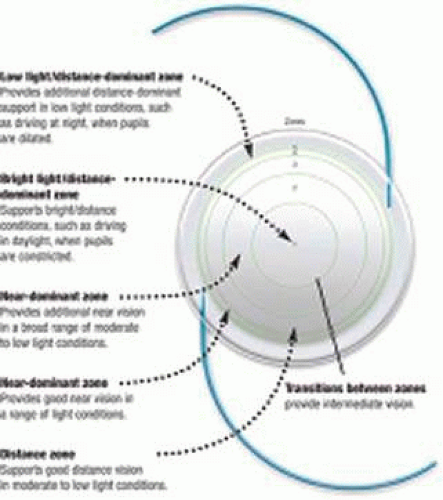
As with the Array, the ReZoom lens has a zonal-progressive design in which the five concentric refractive zones allow for alternating distance and near vision with zones 1 (the central zone), 3, and 5 being distance dominant and zones 2 and 4 being near dominant (Figs. 3 and 4). Aspheric transitions between the zones provide balanced intermediate vision. The refractive surface of the ReZoom lens is on a hydrophobic acrylic platform that incorporates the OptiEdge design.
 Figure 4. Schematic diagram of the refractive zones of the ReZoom IOL. Courtesy of American Medical Optics. |
Although the inherent design mimics that of the Array, the ReZoom multifocal IOL incorporates Balanced View Optics technology that manipulates light distribution across distance and near vision zones to reduce symptoms of dysphotopsia in low-light situations. The aspheric transition zone provides intermediate vision in all zones, even if the distance and near powers form slightly out-of-focus retinal images.
Selection of the ReZoom lens should be targeted to emmetropia, and any necessary error in the refractive target should be on the side of slight hyperopia (±0.25 D).27 The goal of the lens power calculation should be to achieve all of the benefits of near through distance vision for the aphakic patient. Patients should be plano to slightly hyperopic to provide good near vision and good distance vision for driving.27
In presbyopic correction, the add power must augment the distance correction to bring the near point within the reading range. A too-high add power will result in a near point that is too close and the range of focus will be reduced. Theoretically, +4.0 D of add power will yield approximately +3.0 D add power in the spectacle plane, resulting in a near point of 33 cm (14 in).27 The near-dominant zones 2 and 4 in the ReZoom lens provide +3.5 D of add power at the IOL plane for near vision, yielding approximately +2.57 D add power in the spectacle plane.27 This correction translates to a near point of 39 cm (16 in).27 The +3.5 D add power of the ReZoom IOL provides sufficient power for good functional near vision; it also provides a more usable working distance at near and an opportunity for better intermediate vision than a multifocal IOL with a higher add power.27
Clinical Studies
Clinical studies28,29 and comparative trials involving monofocal30,31 as well as other multifocal lenses21,22,23,25,32,33 have demonstrated the ability of the refractive multifocal ReZoom lens to provide good quality distance, intermediate, and near vision and improved patient quality of life (Table 2).
Table 2. Summary of Results of Selected Studies Comparing Intraocular Lenses (IOLs) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
In comparison with the Array multifocal lens, one retrospective chart analysis21 reported significantly better near vision with the ReZoom, and several prospective studies22,23,25 have reported comparable or better visual performance with less photic phenomena in those with the ReZoom implant. In retrospective chart analysis,21 more than 75% of 464 patients had refractive lens surgery and received a zonal multifocal Array lens implant, with 12 patients having the ReZoom implant. The lenses performed comparably at far, with significant differences favoring the ReZoom lens noted at near. The mean uncorrected near visual acuity was J 2.56 versus J 2.0 among those with the Array and ReZoom, respectively, (p <0.05). The analysis also evaluated 656 patients who had refractive cataract surgery and found a mean uncorrected near visual acuity of J 3.64 in the 644 with Array implant versus J 2.0 among the 12 patients with the ReZoom implant (p <0.05).
In a prospective study,22 60 patients had bilateral implant with either the ReZoom or the Array lens. At 6 months after implantation, all patients expressed satisfaction with the surgical results. Distance vision was similar in the two groups; however, 80% of those with the ReZoom implant (24/30) were spectacle independent compared with 60% of those with the Array implant. Of those with the ReZoom implant, 80% reported no glares or halos compared with 40% of those with the Array implant. In another study,23 visual acuity, photic phenomena, and defocus acuity curves were compared between the ReZoom and Array lenses. Similar defocus acuity curves were observed between the lens implant groups, indicating good near and intermediate vision. Those having the ReZoom implant reported spectacle independence at distance (100%), intermediate (95%), and at near (71%). The ReZoom group also reported a lower frequency of photic phenomena (i.e., halos and starbursts) when compared with those implanted with the Array.
Several studies have compared the refractive ReZoom multifocal IOL with the diffractive ReSTOR multifocal IOL. Visual efficacy was compared among 51 patients having bilateral implant with ReZoom, ReSTOR, or the AcrySof monofocal IOL.32 At 3 months, the uncorrected distance visual acuity was 20/40 in 88% of both multifocal groups compared with 94% in the monofocal group. Uncorrected near vision acuity was 20/40 in 88% of the ReZoom, 94% of the ReSTOR, and 23% of the monofocal patients. Uncorrected intermediate vision acuity was 20/40 in 64% of ReZoom, 41% of ReSTOR, and 23% of monofocal patients. Contrast sensitivity was lower in the eyes with multifocal implants.
In a three-arm study that also included the Tecnis aspheric multifocal lens, 30 patients received bilateral implants with the ReZoom, ReSTOR, or the Tecnis IOL.33 Patients in all groups had similar outcomes for distance acuity, depth-of-focus, quality-of-life assessments, and contrast sensitivity, the latter of which was slightly decreased in all groups. Although similar, comparisons between the diffractive and refractive lens groups found that those patients having implants with diffractive models (i.e., Tecnis or ReSTOR) performed slightly better at near vision and scored slightly higher on a quality-of-life questionnaire than those having implants with the refractive IOL.
Implantation of the ReZoom instead of the Tecnis may be better in those with pupils larger than 5 mm, whereas the Tecnis may be better in those with pupils smaller than 5 mm, according to recent findings.34 In the study, 31 eyes of 18 patients (mean age 45 years, range 24–65) with spherical equivalent refraction range of -2.41 ± 6.57 D had the Tecnis implant and 19 eyes of 12 patients (mean age 56 years, range 31–81) and spherical equivalent refraction range of -0.78 ± 4.77 D had the ReZoom implant. Preimplantation and postimplantation visual acuities were similar in the groups. Glare and halo occurred in three with the Tecnis implant, with these patients having dim light pupil size ≥ mm or greater. Glare and halo occurred in one patient with the ReZoom implant who had a dim light pupil size of 7 mm.
Emerging data suggest that implantation of the refractive ReZoom lens in one eye and a diffractive multifocal IOL (ReSTOR or Tecnis) in the contralateral eye provides excellent range of visual function.35,36,37,38,39 IOL implantation selection may be based on assessment of the patient’s visual needs: For those who spend more time reading or performing other near vision tasks, the ReZoom refractive IOL is implanted in the nondominant eye and the ReSTOR IOL implanted in the fellow eye, and visa versa for those with more need for intermediate vision.38 In the largest study to date,35 investigators compared visual function in 39 patients with the ReZoom implanted in the nondominant eye and the ReSTOR in the dominant eye versus 55 patients bilateral ReSTOR implantation. Both groups were found to have similar bilateral near vision (J 1.06 vs. J 1.0 in the ReZoom/ReSTOR and ReSTOR/ReSTOR groups, respectively).35 Of note, the combination implantation group (ReZoom/ReSTOR) exhibited better bilateral intermediate vision (J 2.39) compared with bilateral ReSTOR group (J 3.81, p = 0.0001 for the comparison). In an assessment of unilateral ReZoom versus ReSTOR eyes, a significant difference in intermediate vision favoring the ReZoom lens was observed (J 3.03 vs. J 4.21, p = 0.0001) with no difference in near vision (J 1.56 vs. J 1.33). Overall complaints regarding intermediate vision were less frequent in the ReZoom/ReSTOR group versus the bilateral ReSTOR group (2.6% vs. 25%).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree