Miscellaneous Abnormalities of the Fundus
Neal H. Atebara
This chapter covers disease entities caused by compressing or stretching mechanical forces on the choroid, Bruch membrane, and retinal pigment epithelium (RPE). Chorioretinal folds are undulations of the choroid, Bruch membrane, and the RPE, caused by compressive forces on these tissues or contraction of the underlying sclera. Lacquer cracks are seen in pathologic myopic degeneration, when the stretching effects of a severely enlarged globe cause disruptions in the RPE, Bruch membrane, and choriocapillaris. Angioid streaks are cracklike defects in the Bruch membrane that result from its calcification and fragmentation secondary to various disease conditions. Choroidal ruptures from blunt trauma are due to rapid anteroposterior compression of the globe, resulting in stretching of the choroid, Bruch membrane, and RPE. Tears of the RPE layer can be seen uncommonly in a setting of pigment epithelial detachments secondary to age-related macular degeneration. The pathophysiology, clinical presentation, and associated disorders of these five disease entities are discussed in this chapter.
Chorioretinal Folds
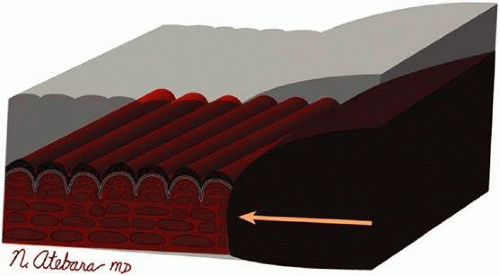
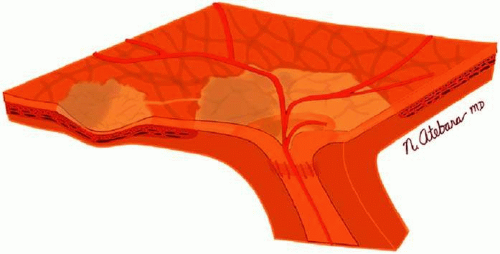
Chorioretinal folds are undulations of the choroid, Bruch membrane, and RPE. The overlying retina may show some minor degree of wrinkling, but only as a secondary effect of distortion of the underlying choroidal-Bruch-RPE complex. Whereas the Bruch membrane is a relatively inelastic tissue, the choroid is composed of a plexus of blood vessels and interstitial tissue that is more easily compressed and distorted (Fig. 36-1). Any mechanical force (such as an intraocular or orbital tumor or a scleral buckle) that sufficiently distorts the choroid will eventually cause the Bruch membrane to be thrown into folds because of its inelasticity. Nettleship1 first described this condition in 1884, and in 1915 Birch-Hirschfeld and Siegfried2 histopathologically demonstrated folds in the retina and choroid of a patient with an orbital tumor. Although an orbital mass remains a common cause of chorioretinal folds, many other orbital and ocular conditions have been associated with this fundus finding.3,4,5,6,7,8,9 They can also occur in otherwise normal eyes and might easily be overlooked during cursory fundus examination. Chorioretinal folds should be distinguished from retinal folds, which occur more commonly and usually reflect tractional forces acting on the retina.10
Opthalmoscopy
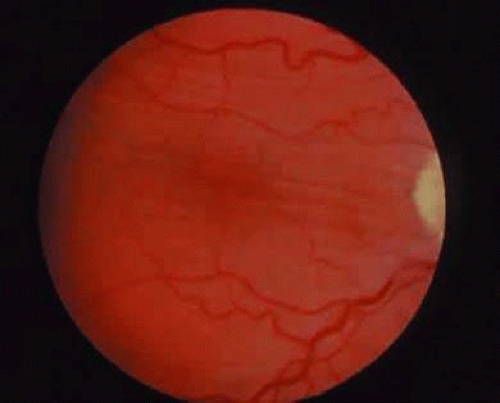
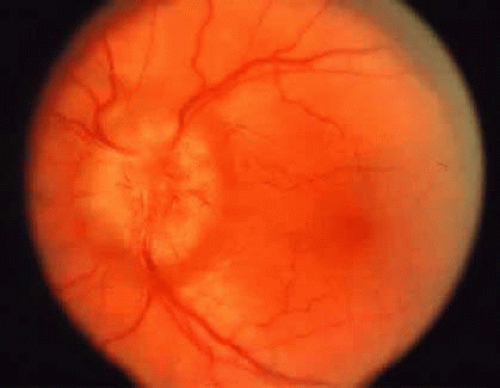
Chorioretinal folds are seen as alternating light and dark striae in the posterior pole, giving the fundus a corrugated appearance (Fig. 36-2).11 The darker striae correspond to the troughs of the folds where the RPE is viewed on edge. The lighter-colored striae correspond with the ridges, where the RPE layer is stretched thin. The width, length, and number of folds are variable. The folds are usually roughly parallel, except in hypotony (in which case the folds are often multidirectional), in a contracted choroidal neovascular membrane (in which case they may be radial), and in optic disc swelling (in which case they may be concentric). Careful biomicroscopic evaluation is helpful in establishing the level of the abnormality in the fundus. The overlying retina is usually uninvolved in isolated chorioretinal folds, although parallel retinal folds can occasionally occur.12
Chorioretinal folds should be distinguished from retinal folds, which are usually thinner, translucent, and radiating from a point of a visible pathologic process in the retina. Retinal folds are more superficial than chorioretinal folds. On careful biomicroscopic examination, the underlying choroid can be seen to be uninvolved in isolated cases of retinal folds. Retinal folds usually reflect tractional forces acting on the neurosensory retina, typically at its surface—the internal limiting membrane. The most common causes of retinal folds are idiopathic epiretinal membranes, diabetic fibrovascular membranes, and proliferative vitreoretinopathy associated with retinal detachment.
Fluorescein Angiography
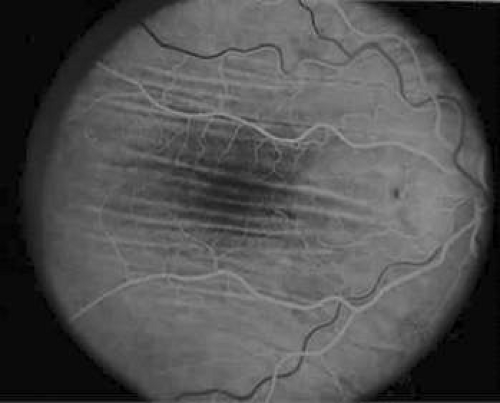
Norton13 described the angiographic appearance of chorioretinal folds in 1969. In the arterial or early laminar venous phase of the angiogram, alternating lines of hyperfluorescence and hypofluorescence become apparent (Fig. 36-3). These fade in the late recirculation phase of the angiogram without leakage. The hyperfluorescent streaks correspond to the peaks of the chorioretinal folds, and the hypofluorescent streaks correspond to the valleys. Histopathologic evidence suggests that attenuation of the RPE at the peaks results in hyperfluorescence; compaction of the RPE layer in the valleys and partial collapse of the underlying choriocapillaris results in relative hypofluorescence.14
Because the RPE and choroidal are unaffected by retinal folds, the fluorescein angiographic appearance is usually normal, except for distortion of the retinal vessels or mild leakage, which may occur with epiretinal membranes.
Etiology
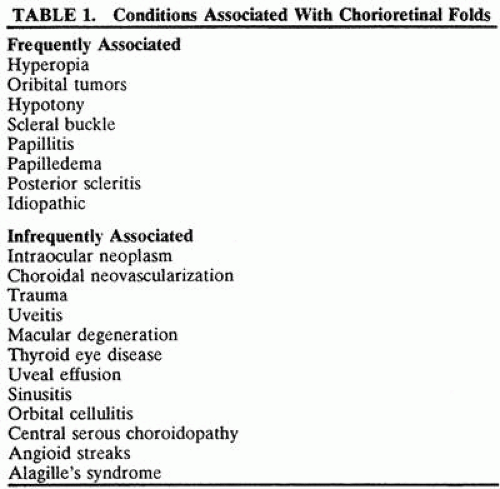
The differential diagnosis of chorioretinal folds is extensive (Table 36-1). The more common causes include orbital tumors, hypotony, papilledema or papillitis, posterior scleritis, hyperopia, and previous scleral surgery. Uncommon causes include intraocular tumor, choroidal neovascularization, trauma, uveitis, thyroid eye disease, uveal effusion, sinusitis, orbital cellulitis, central serous choroidopathy, angioid streaks, and Alagille syndrome.3,4,5,6,7,8,9 Idiopathic chorioretinal folds are sometimes encountered as an incidental finding in an otherwise normal eye.
The mechanism by which chorioretinal folds are produced probably relates to mechanical forces that compress the spongy choroid, thereby throwing the relatively inelastic Bruch membrane into folds. Choroidal tumors (choroidal melanomas, metastatic choroidal carcinomas) may produce chorioretinal folds at the margins of the tumor.13,15,16,17 These folds are produced by mechanical displacement of the surrounding choroid by the expanding tumor in conjunction with choroidal vascular engorgement. As the choroid is compressed laterally, the relatively inelastic Bruch membrane is thrown into folds (Fig. 36-4). These folds roughly follow the shape of the tumor’s margin.
Optic disc swelling (papillitis, papilledema) may cause chorioretinal folds in a similar fashion, where expansion of the optic nerve at its entrance into the eye causes compression of the surrounding choroid (Fig. 36-5).4 The folds are sometimes seen concentric to the optic nerve in the meridians of greatest nerve swelling, often nasally.
Orbital tumors may produce chorioretinal folds through scleral deformation and choroidal congestion. It is nteresting that scleral indentation alone does not produce chorioretinal folds—it therefore appears that scleral edema, scleral shortening, choroidal congestion, and possibly other factors are necessary for the formation of chorioretinal folds.
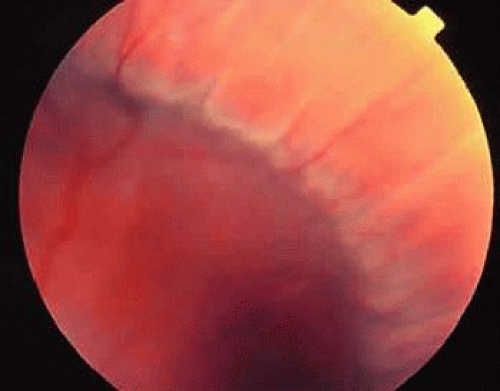
Scleral buckles placed for the repair of retinal detachment can cause chorioretinal folds. These folds are seen perpendicular to the buckle, and they may be exacerbated by the presence of a serous choroidal detachment (Fig. 36-6).
Intraocular hypotony results from a wound leak or excessive filtration after glaucoma surgery. Chorioretinal folds may be seen in this condition. The folds may tend to radiate outward from the optic disc, may be concentric with the optic disc, or they may be randomly oriented. These folds may result from a combination of scleral contraction due to the low intraocular pressure, scleral edema, choroidal congestion, and optic disc swelling (Fig. 36-7).
Clinical Correlation
Chorioretinal folds occur in patients at any age and of either gender. In general, symptoms are related to the cause of the folds. Blurring of vision is a frequent presenting symptom.18,19 Proptosis and diplopia can also occur if an orbital mass is present. Increasing hypermetropia or astigmatism and distortion of the Amsler grid are frequently present.
Examination of a patient with chorioretinal folds should include careful assessment of ocular motility and exophthalmometry. In addition to fluorescein angiography, which will confirm the diagnosis, computed tomography or magnetic resonance imaging of the orbits should be performed in all patients in whom the cause of the folds is not otherwise apparent.20 Treatment of the underlying condition usually results in resolution of the folds, although this may occur gradually.21
Myopic Degeneration
Although myopia may result from excessive refractive power, it is the expansion of the scleral shell resulting in axial elongation that causes the fundus abnormalities and retinal complications associated with pathologic myopia. The fundus findings of myopia do not strictly correlate with the degree of refractive error, but the likelihood of the appearance of pathologic changes in the fundus clearly increases in eyes with greater than -6 to -8 diopters of myopia.22 Therefore, in clinical practice, myopia is often classified as physiologic when the refractive error is less than -6 diopters and pathologic when the refractive error is -6 diopters or more.
The precise causative role of environment versus genetics in myopia is not clear, although in many cases there appears to be a strong genetic association in pathologic myopia. Pathologic myopia may be seen in various genetically determined diseases such as Marfan syndrome, Ehlers-Danlos syndrome, and Stickler syndrome. The sclera in familial cases of pathologic myopia show collagen that is thinner and more loosely woven than normal,23 and it has been hypothesized that this leads to sclera that is structurally weak and thus subject to stretching in response to intraocular pressure.24
Some fundus findings related to expansion of the scleral shell include temporal peripapillary atrophic crescent, patches of RPE atrophy, lacquer cracks, and posterior staphyloma. Subretinal hemorrhage, choroidal neovascularization, and Foerster-Fuchs spots (pigmented involuted choroidal neovascularization) may also be seen. White without pressure, lattice degeneration, paving-stone degeneration, and posterior vitreous detachment are seen more commonly in highly myopic eyes than in the normal population.25
Physiologic Myopia
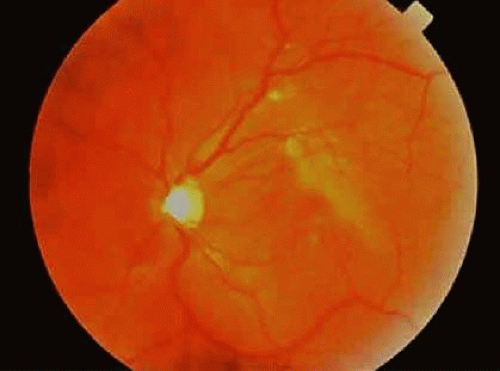
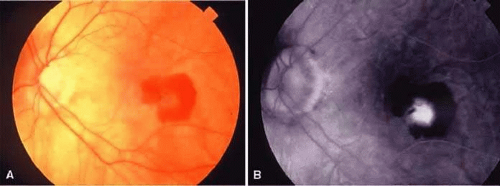
The fundus finding that is most commonly seen with low to moderate degrees of myopia (less than -6 diopters) is a scleral or choroidal crescent located adjacent to the optic nerve head (Fig. 36-8).26 As the globe expands, the retina, RPE, Bruch membrane, and choroid are pulled away from the optic nerve head, resulting in the scleral crescent (Fig. 36-9). In this area, the sclera is clearly visible given the absence of the RPE, Bruch membrane, and choroid, which are usually found at the temporal aspect of the disc.27,28 In physiologic myopia, its width rarely exceeds one third of a disc diameter. Stenstrom and other investigators29,30,31,32 showed that the presence of a peripapillary crescent correlated with increasing axial length but not total refractive power (corneal plus lens power) of the eye.
In patients with higher degrees of myopia, more substantial fundus changes may occur. The enlarged temporal crescent’s width may exceed one third of a disc diameter. A crescent light finding known as the Weiss-Otto reflex33 may become apparent on the nasal aspect of the disc. This reflex results from concentric “piling up” of the nasal retina and choroid, and it is best appreciated in young persons.
Pathologic Myopia
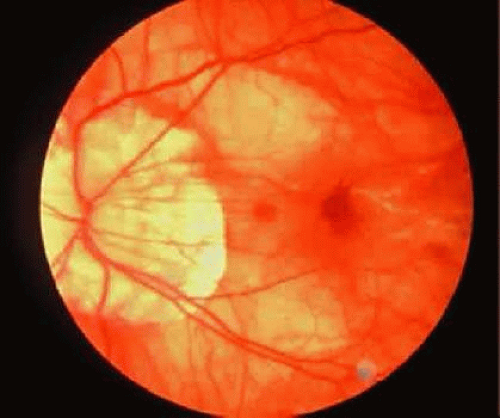
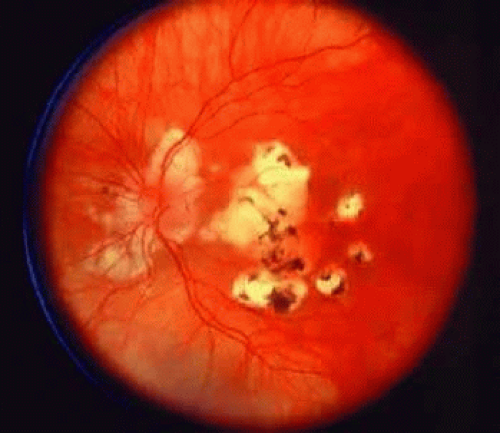
In pathologic myopia, the RPE is stretched and attenuated throughout the fundus. This results in a pale-appearing fundus with increased visualization of the choroidal vessels.22,34,35 In progressive disease, the RPE may become atrophic in patches surrounding the optic disc and in the macula. Loss of RPE function inevitably results in photoreceptor damage, and macular involvement often results in significant loss of central visual acuity. Secondary pigmentary changes may be seen in these patches of RPE atrophy (Fig. 36-10).
If there is sufficient tangential stretch, lacquer cracks may occur. These are areas of fissuring in the RPE, Bruch membrane, and choriocapillaris,36 with a histopathologic appearance similar to that seen in angioid streaks and traumatic choroidal ruptures. Funduscopically, they appear as yellow-white lines of variable caliber found deep in the retina, principally in the posterior pole.37,38 They form a distinctive reticular pattern with connections to the temporal scleral crescent (Fig. 8), frequently appearing in the third or fourth decades of life. They are often multiple, and normal large choroidal vessels may be seen traversing them. Lacquer cracks extending through the macula may result in poor central visual acuity.
Normal development of the sclera and choroid partially depends on the presence of normal overlying RPE. In pathologic myopia, the sclera and choroid may be thinned secondarily, resulting in marked ectasia and outpouching in the posterior pole, called posterior staphyloma (Fig. 36-11). These areas may be large, occupying the entire posterior pole. Although the retina and RPE are thinner in these areas, visual function may be relatively normal except for the distortion caused by the outpouching. In other cases, there is markedly reduced visual acuity due to atrophy of the RPE and choriocapillaris. Posterior staphylomas most often involve the macula and optic nerve.
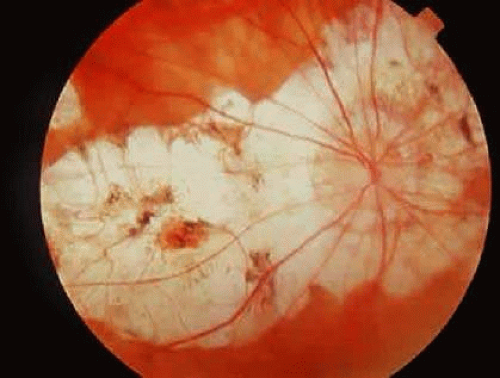 Fig. 36-11. Photograph of a pathologically myopic eye with large staphylomas of the optic nerve and macula. |
Spontaneous subretinal hemorrhage can occur in the macula of the pathologically myopic eye. The origin of the bleeding is not certain, although it probably arises from the choriocapillaris, and the hemorrhage is sometimes first noticed immediately after the appearance of a break in the Bruch membrane. Subretinal hemorrhages can be an isolated finding with a benign clinical course if choroidal neovascularization or a lacquer crack is not involved. Blood often resorbs over a period of weeks to months, and vision may return to its former level.
Choroidal neovascularization may occur secondary to a defect in the Bruch membrane from a lacquer crack or a patch of RPE atrophy (Fig. 36-12).38 The presence of myopic choroidal neovascularization is not necessarily as ominous as it is with other conditions, such as age-related macular degeneration.38,39,40,41,42,43 Overall, up to 50% of patients with choroidal neovascularization may see spontaneous improvement or stabilization of vision. The reason for this finding is that myopic choroidal neovascular membranes often demonstrate a self-limited course, regressing spontaneously. The long-term prognosis for these patients, however, is still guarded because of recurrent subretinal hemorrhage and degeneration of the RPE and choriocapillaris.
The sequelae of the myopic choroidal neovascular membrane is a round or elliptical black lesion known as a Fuchs spot, which may be seen in up to 10% of patients with pathologic myopia, usually in the macula.44,45 This lesion is sharply circumscribed, slightly elevated, and of variable size. Histopathologic examination reveals marked RPE hyperplasia in an area where a choroidal neovascular membrane has undergone spontaneous involution. Overlying fibrosis may impart a gray, yellow, green, or red coloration to the lesion; with time, the spot becomes less distinct with a surrounding halo of atrophy. Like lacquer cracks, Fuchs spots are a poor prognostic sign, with the development of subsequent widespread chorioretinal degeneration being the rule.43
Extensive peripheral retinal changes are invariably present in pathologic myopia.26,46 Benign changes include paving-stone degeneration and the area’s turning white without pressure. Posterior vitreous detachment tends to occur at an earlier age, there is a higher incidence of lattice degeneration, and the retina is relatively thin compared with the emmetrope. Not surprisingly, the risk of retinal tears and retinal detachment, sometimes bilateral, is significantly higher than in the emmetropic population. Round and multiple breaks characterize a myopic retinal detachment.47,48,49 Scleral ectasia can make surgical repair of retinal detachment more challenging.50,51
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree