When you’re lying awake With a dismal headache And repose is taboo’d by anxiety, I conceive you may use Any language you choose To indulge in without impropriety. “Iolanthe” by W.S. Gilbert
Headache is one of the most common presenting complaints for clinicians and encompasses a diverse spectrum of maladies. The International Headache Society (IHS) has revised their classification of headache diagnoses, and this is summarized in
Table 16-1.
1 This chapter addresses the neuro-ophthalmologic aspects of migraines and provides a brief review of other common headaches, facial and ocular pains.
Migraine is a periodic, paroxysmal, protean disorder whose prevalence varies by race and geographic region. In the United States, observed prevalence is lowest among Asian Americans, intermediate in African Americans, and highest among Whites, independent of demographic covariates.
2 Similarly, an international meta-analysis found that the prevalence was lowest in Africa and Asia and higher in Europe and Central/South America.
3 The highest estimates were found in North America.
In the United States, migraine affects an estimated 18% of women and 6% of men.
4,
5 It has been estimated that nearly one in four US households had someone with migraine.
6 Higher income and education levels are associated with lower rates of migraine.
2One of the first names for this disorder was the descriptive term
hemicrania; this was later contracted by the French in the 13th century to the word “migraine.” Although descriptions of migraine-like symptoms and conditions were found in Babylonian writings dating back to 3000 BC and in Egyptian papyri from 1550 BC, the first modern description of migraine and its possible causes is attributed to Thomas Willis about 300 years ago. Famous historical migraine sufferers included Julius Caesar, Emmanuel Kant, Alexander Pope, Isaac Newton, and Sigmund Freud. Throughout the 18th and 19th centuries, descriptions of the clinical phenomena and suggestions for therapy appeared in the writings of many prominent men in the medical professions. Edward’s masterful treatise
On Megrim, Sick Headache, and Some Allied Disorders (1873, as detailed by Sacks) is an unequaled description of the disorder.
7 Further detailed clinical descriptions are found in the writings of Gowers.
8In contemporary medicine, Dalessio, Goadsby, Raskin, Sacks, Silberstein, Lipton, Stewart, Saper, and Welch have all contributed significantly to the study of migraines. A constant tenet for all is that migraine cannot solely be defined by a unilateral (hemicranial) headache. “It is necessary to state that headache is never the sole symptom of a migraine, nor indeed is it the necessary feature of migraine attacks… Migraine is diagnosed by the entire history, not by physical findings or by the presence of headache alone.”
7,
9Unfortunately, many have limited their concept of migraine to a stereotyped syndrome of visual disturbance followed by unilateral throbbing headache, which can be diagnosed by the response to ergot preparations. In actuality, migraine includes several well-recognized syndromes as well as a variety of “equivalents” less commonly classified as migraine. The symptom-complexes or syndromes of migraine include migraine without aura, migraine with aura, ophthalmoplegic migraine, retinal migraine, as well as the others listed in
Table 16-1. The clinical features of migraine will be discussed according to the formal criteria published by the IHS in 2004.
1Other conditions and syndromes discussed in this chapter include cluster headache, trigeminal neuralgia, atypical facial pain, temporal arteritis, and the headaches produced by intracranial mass lesions, muscle contraction, trauma, vascular anomalies, and ocular lesions.
PATHOPHYSIOLOGY
Migraine is a neurovascular process.
22 It appears to be a polygenetic “channelopathy” of ion channels in the brain stem nuclei that modulate sensory input.
23 An abnormal fluctuation of ions leads to electrophysiologic changes that subsequently cause changes in cerebral blood flow (CBF). Initially, there is a brief decrease in CBF, followed by a rebound vasodilation and increased CBF for several minutes, then finally an extended period of decreased blood flow (several hours).
24 The initial electrophysiologic changes, termed CSD, are the likely basis of migraine aura.
25,
26 The headache was initially thought to be the solely result of painful vasodilation, but it is now thought to also be caused by activation of the trigeminovascular system (TGVS). The TGVS is superficial cortical and meningeal blood vessels innervated by the trigeminal nerve. It projects to the trigeminal caudal nucleus in the brain stem, which in turn projects to higher order pain centers, causing headache.
24 It is unclear if CSD also plays a role in activation of the TGVS.
The role of vascular phenomena in the migraine headache was recognized largely by the work of Wolff and colleagues.
27 Research suggests that the migraine attack starts as a neuronal phenomenon with secondary hemodynamic consequences.
28,
29,
30,
31,
32 Wolff divided the migraine attack into four phases: preheadache, headache, late headache, and postheadache. The preheadache phase is characterized by the constriction of certain blood vessels that supply the brain. Then, the beginning of the headache phase is characterized by vascular dilatation, particularly involving branches of the external carotid such as the temporal, occipital, and middle meningeal arteries. Local tenderness of the scalp ensues, and the scalp vessels may become rigid. The nature of the headache then changes from a pulsatile type to a more constant dull ache. Alleviation of the early headache phase with vasoconstrictors (e.g., ergotamine) supports vasodilation as a source of pain. However, vasodilatation may occur without pain, and additional factors are involved in the production of the headache. Local tissue changes take place (e.g., vessel edema, scalp swelling, and conjunctival chemosis) that may continue after vasodilatation has ceased.
Various substances have causative roles in the production of large and small vessel dilatation as well as local tissue changes. Among the substances most frequently cited are the kinins (neurokinin and bradykinin), acetylcholine, histamine, serotonin, and reserpine. Migraine, then, may result from dysfunction of brain stem or diencephalic nuclei involved in nociceptive modulation of afferents from the trigeminal vascular system.
28Positron emission tomography has detected activation in the brain stem during attacks of migraine.
33,
34 Sicuteri
35 hypothesized that the following sequence occurs: the initial event is a local release of catecholamines (with vasoconstriction and increased urinary excretion of vanillylmandelic acid); during subsequent reactive hyperemia serotonin is released (documented by plasma serotonin decrease
36 and increased urinary 5-hydroxy indoleacetic acid
37), presumably from platelets or mast cells, which sensitizes cranial pain receptors perhaps also affected by the kinins.
Additional evidence suggests that there are nervous system connections between the trigeminal ganglia and cerebral blood vessels, the TGVS.
38 Trigeminovascular neurons and their peripheral unmyelinated nerve fibers contain the neurotransmitter peptide, substance P. Stimulation of this system by various mechanisms would cause the release of substance P, which is postulated to increase vascular permeability and dilate cerebral blood vessels. The role of this system in the generation of human vascular headache may account for the effects of hormones or other circulating substances that change the receptive field properties of trigeminal ganglion cells.
Individuals prone to chemically induced headaches from ingestion of tyramine, alcohol, phenylethylamine, monosodium glutamate, nitroglycerine, wine, or chocolate also experience spontaneous headaches.
39 Extensive studies of the reactivity of blood vessels in migraine
40 and CBF
30,
32,
41,
42 suggest that abnormal vasomotor responses may be present in patients with migraine between, as well as during, migraine attacks.
There are several lines of indirect evidence that suggest a relationship between serotonin and migraine, making the understanding of the pharmacology of serotonin important for understanding the pharmacology of the new serotonin agonist in migraine therapy
11 The serotonin or 5-HT receptors consist of at least three distinct types of molecular structures: guanine nucleotide G proteincoupled receptors, ligand-gated ion channels, and transporters. At least five 5-HT
1 receptor subtypes are present in humans. Headaches resembling migraine can be triggered by serotonergic drugs such as reserpine (a 5-HT releaser and depleter) and m-chlorophenylpiperazine (a serotonin agonist).
43,
44Other metabolic and endocrine factors also influence migraine attacks. According to Friedman and Merritt,
45 80% of pregnant women previously prone to migraines either lose the headaches or experience improvement. However, Callaghan
46 found an increase in the severity of migraine in pregnancy. The use of oral contraceptives appears to increase the incidence and severity of migraine.
47,
48 Whitty et al.
49 felt that migraine might be precipitated by withdrawal of progesterone, whereas Somerville
50 found from a study of three women with regular menstrual migraine that their attacks were related to estradiol withdrawal rather than to decreasing levels of progesterone. Tyramine has also been invoked as a precipitating factor, especially in the so-called allergic migraine;
51 however, only approximately 5% of migraine subjects notice headaches precipitated by food. However, some patients are unusually sensitive to chocolate or alcohol, particularly red wines. Recent therapeutic trials with dietary therapy designed to avoid hypoglycemia suggest that glucose and/or insulin metabolism may play a role in the generation of vascular headache.
The role of trauma in the production or exacerbation of a preexisting migrainous tendency is still incompletely defined. Many individuals experience vascular headaches of the common migraine type after even minor head trauma.
52 A previously well-controlled migraineur can experience a recrudescence of prior symptomatology after a head trauma. Such exacerbations are usually short-lived with a return to the preinjury status in weeks to months. However, there are some patients who experience post-traumatic migraine headaches for years after a head injury. Other triggering events preceding migraine attacks include bright light, especially sunlight reflected from water, exercise or exertion, and high altitude. Vascular headache of the migraine type may also follow orgasm.
53 The role of stress is less clear. It appears more likely that migraine headache follows a period of psychological stress than occurring during the time of stress.
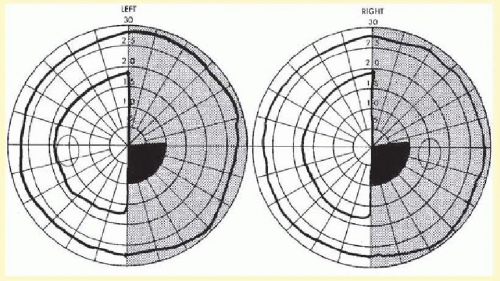
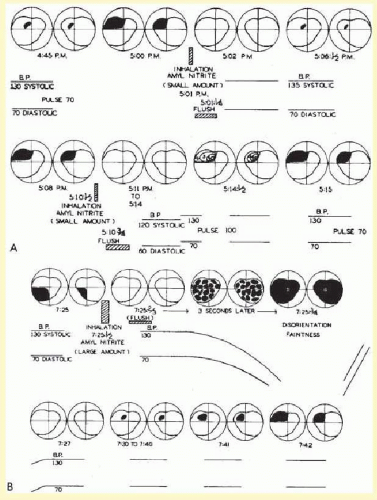
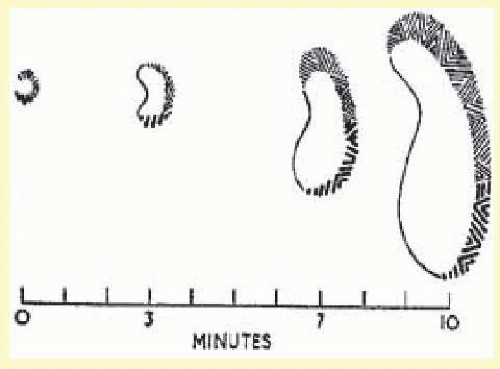
The pathophysiology of the migraine aura itself also has been studied extensively. Wolff showed that the use of a potent vasodilator, amyl nitrate, could abort the migraine scotoma (
Fig. 16.1), supporting the vasoconstrictor hypothesis. Milner
54 suggested that the scotomas of migraine and the neurophysiologic phenomenon, Leão spreading depression, may be related. The spreading depression progresses across the cortex at approximately 3 mm per minute, similar to the slow evolution of the visual phenomenon that had been detailed by Lashley,
55 and estimated to spread over the occipital cortex at a rate of 3 mm per minute.
It is currently believed that the aura of migraine may be the human counterpart of the animal phenomenon of Leão spreading depression.
18 The aura is characterized by a wave of decreased blood flow or oligemia passing across the cortex
30,
56,
57,
58 at a slow rate (2 to 6 mm per minute) consistent with the spread of the visual phenomenon through the visual cortex, as mentioned above.
19 There is a short phase of hyperemia preceding the oligemia that may be a correlate of the scintillating scatoma, also a characteristic of migraine with aura.
59 However, persistent oligemia is probably a response to depressed neuronal function and is present when the headache starts, as noted by Goadsby.
58,
60,
61 Such findings coupled with the direct evidence of adequate local oxygen supply
62 vitiate the theory that migraine is just a vascular headache.
61Three cardinal factors are important in the pathogenesis of migraine, according to Goadsby.
61 These include the cranial blood vessels, the trigeminal innervation of these vessels, and the reflex connection of the trigeminal system with a cranial parasympathetic outflow. The pain sensitive structures within the cranium, such as large blood vessels or the dura mater, are innervated by branches of the ophthalmic division of the trigeminal nerve,
63 and the posterior fossa structures are innervated by branches of C2 nerve roots.
64 Involvement of the ophthalmic division of the trigeminal nerve and the overlap with structures innervated by C2 explain the common distribution of the pain of migraine in the frontal and temporal regions, as well as involvement of parietal occipital and high cervical regions, by referred pain.
61 Peripherally, the trigeminal afferents are activated in migraine by the release of calcitonin gene-related peptide (CGRP) a vasodilator,
65 and while the mechanism of pain generation is not entirely clear, animal studies suggest that pain is caused by a sterile neurogenic inflammation in the dura mater.
66 This may, in part, explain the prevention of migraine pain by substances such as botulinum toxin type A, which inhibit the release of CGRP.
67 The pain may be a combination of an altered perception—as a result of peripheral or central sensitization—of craniovascular input that is not usually painful
61,
68 and the activation of feed-forward neurovascular dilator mechanism that is functioning specific to the first ophthalmic division of the trigeminal nerve.
69 Again, the effect of botulinum toxin type A in reducing migraine pain may interfere with this peripheral activation and, therefore, functions to provide peripheral desensitization.
67Eighty percent of migraineurs experience photophobia, or exacerbation of the headache by what is normally a tolerated level of light, during migraine attacks.
70 This photophobia is not a product of the traditional image forming pathway of the visual system, for example, the photoreceptors, retinal ganglion cells (RGCs), lateral geniculate nucleus, and visual cortex.
71Instead, it is a nonimage forming system of vision that originates in intrinsically photosensitive RGCs (ipRGCs) that appears to be the origin of photophobia. These ipRGCs contain the photoreceptor melanopsin and function without any synaptic input from the rods and cones.
71 Their peak spectral sensitivity lies in the blue range at approximately 480 nm.
72 The axons of these ipRGCs project via the optic nerve to the suprachiasmatic nucleus, intergeniculate leaflet, and the olivary pretectal nucleus.
72 It is through these projections that the melanopsin system mediates such nonimage forming visual functions like the pupillary light response and circadian rhythms. Both of circadian rhythm and pupillary light response are preserved in some blind patients. That is because the melanopsin-containing ipRGCs are spared in some blinding eye conditions, for example, mitochondrial optic neuropathies (Leber and dominant optic atrophy) and peripheral retinal degenerations (retinitis pigmentosa); however, they tend to be preferentially targeted in glaucoma.
72Likewise, migrainous photophobia is preserved in some blind patients. Noseda et al.
71 not only found that light modulated the activity of a subset of trigeminovascular thalamic neurons but also that most retinal projections to the thalamus consisted of axons of ipRGCs. They therefore posit that the melanopsin system carries the photic signal from the retina to the nociceptive thalamic TGVS, and thereby exacerbates migraine headache pain.
MIGRAINE WITHOUT AURA
The IHS classification has improved the diagnosis of headaches. It has also facilitated clinical research on migraine. In order to establish a diagnosis of migraine without aura, five attacks are needed (
Table 16-2). Each attack must last 4 to 72 hours and have two of the following four pain characteristics: unilateral location, pulsating quality, moderate to severe intensity, and aggravation by routine physical activity. In addition, the attacks must be associated with at least one of the following: nausea, vomiting, or photophobia and phonophobia. With these criteria, no single characteristic is mandatory for a diagnosis of migraine. A patient who has severe pain aggravated by routine activity, photophobia and phonophobia, meets these criteria as does the more typical patient with unilateral throbbing pain and nausea.
Migraine usually lasts several hours or the entire day. When the migraine persists for longer than 3 days, the term “status migrainosus” is used. Frequency of attacks varies widely from a few per lifetime to several per week.
11 The average migraineur experiences from one to three headaches per month.
5 A precise location ascribed to migraine, such as unilateral or temporal, is misleading, for as Wolff
40 wrote,
The sites of migraine are notably temporal, supraorbital, frontal, retrobulbar, parietal, postauricular, and occipital… They may as well occur in the malar region, in the upper and lower teeth, at the base of the nose, in the median wall of the orbit, in the neck, and in the region of the common carotid arteries and down as far as the tip of the shoulder.
The prodromes of common migraine are vague, preceding the attack by hours or days, and include psychic disturbances (such as depression or hypomania), gastrointestinal manifestations and changes in fluid balance. Usually the onset of the common migraine headache is unilateral, but the pain often becomes holocephalic. In an individual patient, the headache is commonly more prominent on a single side, with occasional or rare alternation. Some individuals always experience a unilateral headache, while in approximately one-third the headache is diffuse from onset. Traditionally, the character of the headache is described as throbbing, but this may be a feature only at onset, with the discomfort soon changing to a steady ache. The victim can often relieve unilateral headache by carotid artery or temporal artery compression, only to experience resurgence of the pain after release.
Nausea in some degree almost always accompanies common migraine. Vomiting can occur at the height of an attack, sometimes with relief of the headache, but more often heralds an intensifying phase of the episode, which continues for many minutes or hours. Usually the migraine sufferer becomes pallid and seeks seclusion, darkness, quiet, and a cold towel or ice bag for the head. Frequently at the time of nausea with vomiting, a diuretic phase with polyuria ensues, the consequence of fluid retention that occurred in the hours or days preceding the acute headache.
Ocular signs and symptoms may occur in common migraine, such as conjunctival injection, periorbital swelling, excessive tearing, foreign body sensation, and photophobia; however, these phenomena are more prominent in cluster headache.
MIGRAINE WITH AURA (CLASSIC)
The new term for classic migraine, that is migraine with aura, requires at least two attacks with any three of the following four features (
Table 16-3): one or more fully reversible aura symptoms, aura developing over a course of more than 4 minutes, aura lasting less than 60 minutes, and headache after aura within 60 minutes. Migraine with aura refers to a more well-defined clinical constellation than does migraine without aura. The episodes are characterized by definite prodrome or aura, which is usually a visual sensation; however, sometimes motor or other sensory phenomena precede the headache. The headaches of classic migraine tend to be more compact and intense, rarely lasting more than 12 hours; most often 2 to 3 hours.
Many general characteristics are shared by common and classic migraine. Both varieties affect men and women and can occur at any age, often seemingly triggered by a significant event such as puberty, school graduation, or marriage. A family history is usually present both in classic and common migraine, and there may be an earlier history of colic as a baby or car illness as a small child. The full history of a complete migraineur would include migraine with aura in the teens, migraine without aura with nausea and vomiting in the second and third decades, followed by simple periodic headache or isolated migrainous auras in later life.
Migraine with aura is subclassified into migraine with typical aura (homonymous visual disturbance, unilateral numbness or weakness, or aphasia), migraine with prolonged aura (or lasting longer than 60 minutes), FHM, basilar migraine, migraine without headache, and migraine with acute-onset aura.
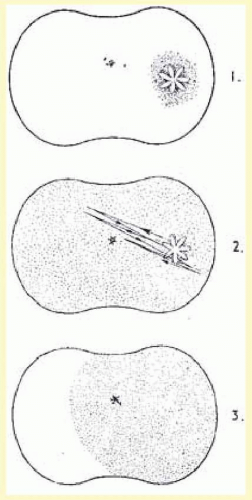
The primary feature of migraine with aura is the visual aura. Extensive reviews of this phenomenon are found throughout the literature.
7,
8,
40,
73,
74,
75 Although many variations occur, the following description by Richards
50 summarizes the most common type of visual phenomena (
Figs. 16.2 and
16.3):
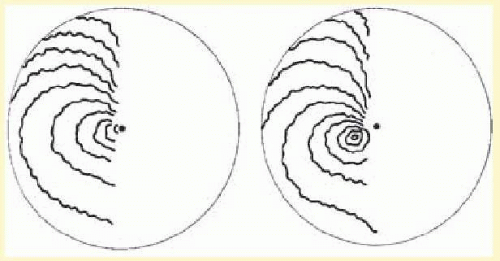
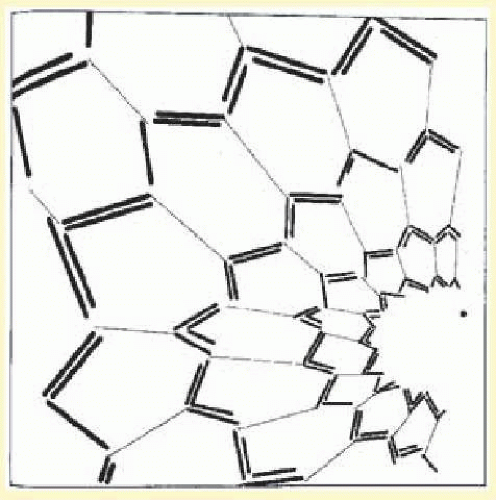
The visual disturbance usually precedes the headache … [it] begins near the center of the visual field as a small gray area with indefinite boundaries. If this area first appears during reading, as it often does, then the migraine is first noticed when words are lost in a region of “shaded darkness.” During the next few minutes the gray area slowly expands into a horseshoe with bright zigzag lines appearing at the expanding outer edge. These lines are small at first and grow as the blind area expands and moves outward toward the periphery of the visual field.
One important aspect of the visual disturbance just described is that it expands slowly over 10 to 20 minutes. The initial region of visual abnormality is most often near fixation and then, as described by Lashley,
55 with increase in size the disturbed area moves or “drifts” across the visual field so that its central margin withdraws from the macular region as its peripheral margin invades the temporal; the area may be totally blind (negative scotoma), amblyopic, or outlined by scintillations.
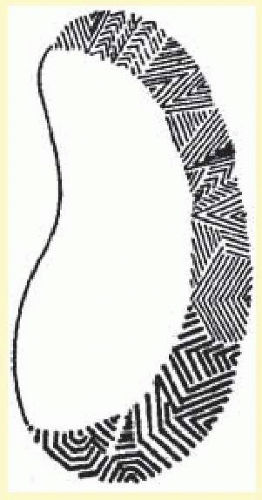
The scintillations surrounding the negative scotoma make “fortification” figures or spectrums, so called by the appearance of a “map of the bastions of a fortified town.”
55 The scintillations are brilliant, with the intensity of a bright fluorescent bulb flickering at a rate of 5 to 10 cycles per second (
Figs. 16.4 and
16.5).
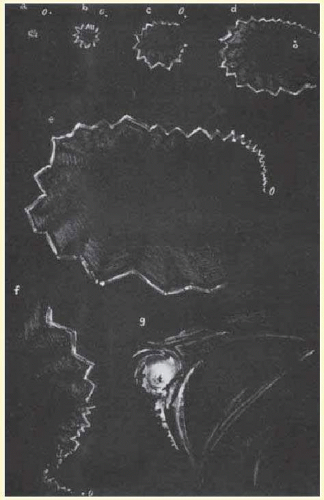
Gowers
76 was particularly impressed with the intensity of the visual sensation. Many migraine sufferers can precisely recall their own vivid visual experiences well enough to describe them precisely or even on occasion to paint them (
Figs. 16.6 and
16.7). Not all migraine visual disturbances begin near the fixation point; some patients consistently experience scotomas starting eccentrically in the visual field, and these sensations can appear alternately or simultaneously in both hemifields (
Fig. 16.8). Other less dramatic visual auras also occur: just the sensation of peripheral brightness or awareness of a rhythmicity or pulsating character in the intensity of the ambient light. The duration of these visual symptoms is measured in minutes rather than the brief few seconds of flashing, bright moving spots, or transient flickering phenomena characteristic of occipital epileptic discharges.
9,
76 Additional visual disturbances are categorized by Klee and Willanger,
74 consisting of metamorphopsia, diplopia, polyopia, and apparent movement of stationary objects. Variations in the scotomas of migraine, including their occurrence in patients with acquired blindness, are well described.
74,
77 The auras of migraine, although most commonly only visual, have many other associated manifestations, such as hemihypesthesias, perioral anesthesia, vertigo, and transient aphasia. The aura or prodromes of classic migraine may be precipitated by intense stimuli: bright lights, loud noises, head trauma, or the intake of certain foods in susceptible individuals.
In the usual sequence of migraine with aura the sensory prodrome precedes the onset of the headache (in accord with the traditional concept of vasoconstriction followed by vasodilatation). The visual disturbance rarely may have a simultaneous onset with headache or, once having disappeared, may recur following the onset of headache. Such unusual patterns, or strict unilaterality for all attacks, should increase suspicion of a mass lesion or vascular malformation. As opposed to definite periodicity with symptom-free intervals and predictable circumstances, as in migraine without aura, migraine with aura may occur “out of the blue” and in multiple attacks over a few days.
Migraine-with-aura attacks tend to diminish in the third and fourth decades. Although most migraine patients experience a stereotyped clinical pattern, there is a well-recognized group in which both classic and common migraine attacks are admixed.
78 Some patients with classic migraine may lose the headache component eventually and suffer only isolated auras thereafter. This monosymptomatic pattern stresses the importance of accurate history-taking when confronted by a patient with isolated visual phenomena (migraine dissociée). Haas
79 emphasized the occurrence of “migraine aura status.” The differential diagnosis should include consideration of vertebro-basilar transient ischemic attacks. Symptomatology that favors migraine has been reviewed by Fisher
80 and includes luminous visual images, build-up of images, progression from one aura to another, and benign outcome.