Fig. 9.1
(a) A 48-year-old female with severe inflammatory TED. (b) Same patient following rehabilitative orbital decompression surgery
Orbital decompression appears successful in improving visual functioning in a majority of patients with the rate of visual recovery correlating to the duration of optic neuropathy [29, 42]. Even in long standing optic neuropathy (>6 months) improvements in visual functioning can be seen. For CON with evidence of apical crowding on imaging (Fig. 9.2), orbital decompression is frequently combined with steroids or radiotherapy [41].
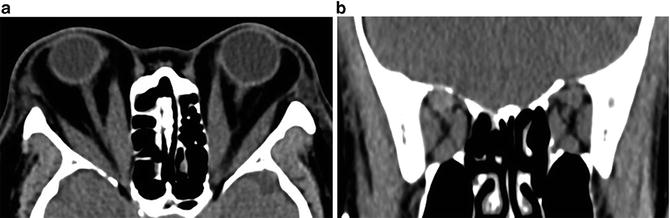

Fig. 9.2
(a) Axial CT of patient with severe thyroid eye disease demonstrating proptosis, extraocular muscle enlargement, optic nerve stretch, and fat infiltration. (b) Coronal CT of same patient
Decompression surgery may involve removal of a combination of the lateral wall, medial wall, orbital fat, and orbital floor [44]. In apical compression an adequate decompression should include the posterior medial wall and ethmoid air cells [44]. Although single wall decompression for CON has been described, recurrent optic neuropathy after single wall decompression has been reported [45]. Our approach for apical compression includes maximal decompression to both expand the orbital apex and also reduce proptosis. This approach avoids subsequent decompression for residual proptosis or recurrent CON.
Rituximab and Biological Therapies
RTX is FDA approved in non-Hodgkin’s lymphoma, chronic lymphocytic leukemia, rheumatoid arthritis (RA), Wegener’s granulomatosis (granulomatosis with polyangiitis), and microscopic polyangiitis but has been used off-label in various autoimmune disorders. RTX is a chimeric mouse-human monoclonal antibody that targets CD20, a human B lymphocyte-specific antigen expressed on more than 95 % of B cells from the stages of immature to mature B cells, as well as memory B cells but not on antibody-producing plasma cells [46]. As a consequence, therapy with RTX removes B lymphocytes and short-lived plasma cells (Fig. 9.3), but not long-lived plasma cells. Thus antibody production is maintained over a long period of time and immunoglobulin (Ig) levels tend not to change despite peripheral B cell depletion [47, 48]. RTX depletes more than 95 % of mature B cells in blood and primary lymphoid organs after 2 days by a single RTX treatment in mice. In humans, the mechanism of action of RTX in the treatment of autoimmune diseases remains unclear: response to treatment does not always correlate with complete B cell depletion and RTX might only indirectly affect autoantibody production [49, 50]. Clinical trials of RTX in RA have shown that it is effective in ameliorating the symptoms with improvement usually seen within 8–16 weeks and persisting for the duration of B cell depletion (typically 16–24 weeks) [51]. The rationale for RTX use in TED includes potential blockade of pathogenic autoantibody generation, depletion of B cell antigen-presentation, and inflammatory cytokine production. However an uncontrolled study demonstrated that in patients with TED, RTX does not affect TRAb titers, suggesting that its efficacy may result from its effect on B cell functions other than that of antibody production [52] or another mechanism entirely.
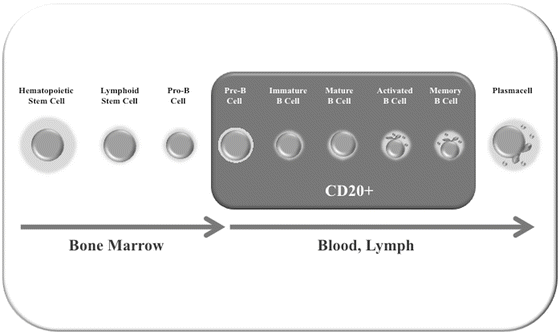

Fig. 9.3
Expression of the CD 20 antigen during B cell differentiation
Infusion related reactions are the most frequently reported side effects of RTX [53]. Most patients experience minor allergic reactions during the first RTX infusion, because the drug is a humanized mAb. Reactions are tempered by anti-histamine and low dose hydrocortisone prior to infusion and by slowing the rate of IV infusion during the first hour. Major acute infusion reactions, caused by inflammatory cytokine release and complement activation, may be present in about 10 % of patients and are usually reversible.
RTX has been reported to increase the risk of infections, especially Hepatitis B reactivation. The increased risk correlates with dose and duration and occurs more frequently in patients with neoplasia [54, 55]. A recent review of over 3,000 patients with rheumatoid arthritis showed comparable serious infection rates in those treated with RTX versus placebo plus methotrexate (MTX) [56]. No increased risk of malignancy over time was observed. Progressive multifocal leukoencephalopathy (PML) has been reported in patients receiving RTX, but predominantly in patients with systemic lupus erythematosus (SLE) [57]. More than 40 % of PML cases occurred in patients with SLE, including those on relatively minimal immunosuppression, suggesting that SLE itself may also predispose to PML development [58].
The effects of RTX in patients with active and severe TED have been studied in 13 patients (Table 9.1) in non-controlled studies, although successful therapy has been described in another 30 patients with active moderate-severe TED. One patient with acute CON received two doses of 1,000 mg RTX, 2 weeks apart. A significant but transient clinical response was seen, with the restoration of normal vision for 16 weeks until B cell return in the peripheral blood, at which time visual deterioration recurred [59]. RTX re-treatment resulted in only transient improvement necessitating orbital decompression. In the patient’s orbital tissues incomplete B cell depletion was noted, with relatively abundant CD19 + 5+ lymphocytes, which usually characterize early repopulation and autoreactive B cell clones [60].
Table 9.1
Characteristics of patients with severe Graves’ orbitopathy treated with rituximab
Study (No. of reference) | No. of patients treated | RTX dose | §CAS before therapy | CAS after therapy (16 weeks) | Severity after #RTX | Side effects | GO relapse |
|---|---|---|---|---|---|---|---|
Salvi et al. [39] | 1 | 1 g twice with 2 week interval + 1 g at 23 w | 7 | 2 | Transient improvement of visual acuity | Minor, infusion related | No |
Khanna et al. [40] | 6 | 1 g twice with 2 week interval | 5.5 | 1.3 | All improved | 2 (minor), 1 (major, cardiac death, likely unrelated to therapy) | No |
Krassas et al. [43] | 1 | 1 g twice with 2 week interval | 7 | 7 | Worsened | n.r. | Yes |
Salvi et al. [44] | 3 | 100 mg single dose | 5.3 | 1.6 | All improved | 2 (major but transient) | No |
Mitchell et al. [42] | 5 | 500 mg or 1 g at 2 week interval | 6.5 | 3 | 6 pts. improved 3 pt. unchanged | 4 (minor) | No |
The therapeutic efficacy of RTX in active, severe TED unresponsive to glucocorticoids was reported in a series of six patients [45]. RTX therapy was associated with significant decrease of the CAS at 8 weeks and 6 months. Four of these patients improved visual acuity to pre-morbid values, but had no improvement of extraocular motility or proptosis. In this study, 1,000 mg IV RTX was administered 2 weeks apart in combination with steroid therapy, which was subsequently tapered off without relapse of inflammation. Minor side effects were described in two patients, but a fatal cardiac event, not attributed to RTX, occurred in one patient. TED improvement and stabilization was associated with abundant peripheral CD25 cells (T regs), which have been reported to be predictive of successful RTX therapy in RA [61]. RTX treatment was studied in a series of patients with steroid-refractory TED, of whom five had CON. The dose of RTX was varied from 500 to 1,000 mg every 2 weeks, based on attainment of peripheral B cell depletion [62]. All the patients in this series improved their NOSPECS score from grade 6 to grade 4. Failure of RTX in preventing active TED progression to CON was described in one patient who was also unresponsive to IV MP [62]. Proposed reasons for therapeutic failure include: RTX was administered too late in the disease course or RTX might have induced increased soft tissue edema and apical optic nerve compression, as a consequence of orbital cytokine release. This effect has been reported in two patients with TED, who experienced transient visual deterioration within an hour from RTX infusion, but spontaneous recovery to normal visual acuity 3–4 h later [63].
Although prospective studies designed to assess the effectiveness of RTX in patients with severe TED are needed, the data available suggest a potential benefit in some. The evidence is mainly based on studies conducted in patients with active, moderate-severe disease, which show that RTX is acting as a disease modifying agent. Recent preliminary data of a randomized controlled trial in these patients seem to show that RTX is more effective than steroids in the inactivation of TED and in preventing relapses of disease, usually observed in 20–30 % [64]. Further data are needed before considering RTX treatment suitable for the most severe forms of disease.
Other biological therapies used in TED include etanercept and infliximab, but data on clinical efficacy is limited [65]. The biologic agent, tocilizumab has shown early promise in a recent retrospective series of TED patients refractive to IV steroid therapy [66]. Tocilizumab is an FDA approved humanized monoclonal IgG1 antibody to the interleukin-6 (IL-6) receptor for use in active moderate to severe rheumatoid arthritis. Of eighteen patients treated, 14 had a CAS score greater than 6, one had optic neuropathy, and one failed previous biological therapy. CAS improved in all 18 patients, proptosis reduced in 13/18, and ocular motility improved in 15/18. The patients with CON improved without the need for orbital decompression. These positive preliminary results were achieved despite half the patients being active smokers, but further clinical trials are warranted [66].
Conclusion
Severe TED management typically requires a multidisciplinary approach involving endocrinologists, orbital surgeons, strabismus specialists, and neuro-ophthalmologists. After severe sight-threatening orbitopathy has been controlled, rehabilitation with decompression, strabismus, and eyelid surgery can restore function, aesthetics, and quality of life [67, 68].
References
1.
Kalmann R, Mourits MP, van der Pol JP, Koornneef L. Coronal approach for rehabilitative orbital decompression in Graves’ ophthalmopathy. Br J Ophthalmol. 1997;81(1):41–5.PubMedCentralPubMedCrossRef
2.
Burch HB, Wartofsky L. Graves’ ophthalmopathy: current concepts regarding pathogenesis and management. Endocr Rev. 1993;14(6):747.PubMed
3.
4.
Dolman PJ, Rootman J. Predictors of disease severity in thyroid-related orbitopathy. (chap 18) Orbital disease. Present status and future challenges. Boca Raton: Taylor and Francis; 2005.
5.
Rundle FF. Management of exophthalmos and related ocular changes in Graves’ disease. Metabolism. 1957;6(1):36–48.PubMed
6.
7.
Bartley GB, Fatourechi V, Kadrmas EF, Jacobsen SJ, Ilstrup DM, Garrity JA, Gorman CA. The incidence of Graves’ ophthalmopathy in Olmsted County, Minnesota. Am J Ophthalmol. 1995;120(4):511.PubMedCrossRef
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


