Nonpenetrating injuries are more common and cause more cases of visual impairment than penetrating injuries.
3 Contusion injuries lead to a rapid shortening of the globe’s anterior-posterior axis causing acute traction along the vitreoretinal interface and vitreous base.
Subsequent vitreous base contracture may lead to traumatic injury, vitreous hemorrhage, retinal tears or detachment, or traumatic macular hole formation. Contusion injury may cause the lens to subluxate or dislocate.
Subluxation or Dislocation of the Lens
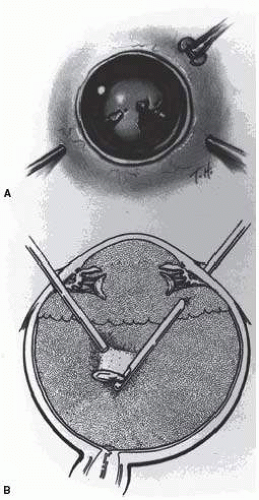
The presence of phakodenesis or iridodenesis on initial evaluation should alert the examining physician to suspect minor lens subluxation. Vitreous in the anterior chamber indicates that zonular rupture is present. In the absence of cataractous changes and related visual impairment, treatment is not indicated. A subluxated, cataractous lens can be removed by aspiration-irrigation or phacofragmentation through a limbal incision. However, a limbal approach may lead to posterior dislocation of the lens or lens fragments, vitreous prolapse and incarceration, and vitreous aspiration with resultant vitreous base traction and retinal tear formation. In addition, visualization of the anterior vitreous by coaxial illumination is poor compared with fiberoptic halogen or xenon endoillumination. These challenges to performing anterior segment surgery make pars plana vitrectomy and lensectomy a potentially safer alternative. Bimanual techniques permit fixation and simultaneous removal of the lens by the vitreous suction cutter, if the lens is soft, or phacofragmentation if it is sclerotic (
Fig. 66.1A). Posteriorly dislocated fragments can be removed safely, with minimal vitreous traction, using the same incisions and instruments (
Fig. 66.1B). With endoillumination, prolapsed and juxtalenticular vitreous is readily identified and excised with the vitrectomy probe.
Pars plana lensectomy is the preferred method for removing a completely dislocated lens. The technical aspects of dislocated lens and lens fragment removal are described elsewhere in these volumes.
Vitreous Hemorrhage
Blood in the vitreous may arise from tears in the iris, ciliary body, choroid, or retina. Hemorrhage from choroidal rupture accumulates beneath the neurosensory retina, and may then pass through the retina into the vitreous without necessarily causing a retinal break. Vitrectomy is indicated for vitreous hemorrhage caused by ocular contusion when a retinal detachment is suspected. Additional indications for vitrectomy in the setting of vitreous hemorrhage include suspicion of an undiagnosed scleral rupture, sudden additional loss of vision, retinal detachment detected through a window in the hemorrhage, retinal break or retinal detachment diagnosed by ultrasound, or lack of improvement after a reasonable period of observation.
Preoperative contact A-scan and B-scan ultrasonography is helpful for detecting posterior vitreous detachment and differentiating it from retinal detachment, though other clinical indicators should be assessed when such a distinction is being made. It can be misleading to rely
completely upon the accuracy of ultrasound. Tissue presumed to be the posterior hyaloid should be approached with caution until it can be identified with certainty.
A three-port vitrectomy technique is the standard approach for nonclearing vitreous hemorrhage. We prefer 23G or 25G, sutureless, transconjunctival vitrectomy if the blood is not particularly dense and no extremely peripheral pathology is suggested. If the blood is dense, and visualization of the infusion port is a concern, a 6-mm infusion port, an illuminated infusion port (preferably with a xenon light source), or an illuminated infusion instrument may be substituted for the standard 4-mm port to facilitate visualization, and larger 20G vitrectomy techniques are preferred. Assuming that the anterior chamber is fairly clear, a central core of opaque vitreous is initially removed, where the tips of the cutter and endoilluminator can be visualized and seen to be safely away from both the retina and the lens. The excision is carried posteriorly, and successive layers of hemorrhagic and fibrinous vitreous are removed until the anticipated plane of the posterior hyaloid is approached. A constant surveillance is maintained for a gray membrane containing radially oriented vessels (undiagnosed detached retina). A small opening is made in the detached posterior hyaloid, through which unclotted blood is aspirated by active suction from a softtipped cannula or vitreous cutter. Once the retina has been visualized, it is best to remove as much retrohyaloid blood as possible to prevent dispersion into the vitreous cavity with consequent loss of visual control.
If the posterior vitreous cortex is not detached, it can be separated from the retina by gentle suction with a softtipped cannula or vitreous cutter at the edge of the optic disk. In cases where the hyaloid is difficult to detach from the retina, such as in children, a small amount of preservative-free triamcinolone acetonide can be introduced into the vitreous. We prefer to have the infusion off during the instillation of the drug, to prevent diffuse spread. The vitrector can then be placed in the eye with the infusion on, and the free triamcinolone can be removed. The residual triamcinolone may adhere to the hyaloid, highlighting its anatomy and facilitating the creation of a posterior vitreous separation.
After establishing the plane between the hyaloid and the retina, the surgeon attempts to remove the entire cortical vitreous except for the firmly attached portion at the anterior vitreous base. Cortex that does not separate with gentle manipulation is isolated from surrounding vitreous to eliminate traction on the retina. It is important to remove the cortical vitreous from areas on and adjacent to retinal breaks. Failure to do so may result in subsequent tangential traction and retinal detachment. When possible, retinopexy is applied to the posterior edge of the tear, and the flap of the tear is amputated. As discussed below, a scleral buckle should be considered if retinal breaks cannot be freed from surrounding vitreous cortex, and in some cases it may be considered when there is a greater risk for the development of future anterior peripheral base contraction.
The placement of sclerotomies close to the 3- and 9-o’clock positions facilitates maximal removal of the hemorrhagic anterior vitreous skirt, which thereby improves visualization of the peripheral retina and pars plana. Aided by coaxial illumination and scleral depression, the peripheral vitreous on the temporal side of the globe is trimmed with the cutter placed through the temporal sclerotomy to reach both the superior and inferior quadrants, after which it is transferred to the nasal sclerotomy, and the process is repeated. The fiberoptic endoilluminator may damage the lens if used internally to illuminate the peripheral vitreous on the opposite side of the globe. However, the cone of light from the probe may be directed externally through the cornea to augment or replace the internal coaxial light source. Alternatively, xenon-powered chandelier lights or lighted infusion cannulas can be utilized to increase the amount of illumination and provide diffuse, wide-field light to allow the surgeon to perform simultaneous vitrectomy and scleral depression without the aid of an assistant. Hemorrhagic retrolenticular vitreous can be stripped from the posterior lens capsule with gentle aspiration into the cutting port followed by withdrawal of the probe and simultaneous activation of the cutting mode. This technique may be dangerous when used in young children because the retrolenticular vitreous is adherent to the lens, which is sufficiently pliable to be aspirated into the port with consequent cataract formation. When the lens is clear, the process of removing peripheral and retrolenticular vitreous is less important than preserving lens integrity in most cases.
It is important to expose the peripheral retina and vitreous base as most retinal breaks caused by ocular contusion are located in this area.
5 The use of wide-angle contact and noncontact viewing systems such as the BIOM (Binocular Indirect Ophthalmo Microscope, Insight Instruments Inc., Stuart, Florida) can greatly facilitate visualization of the peripheral fundus.
6 All retinal breaks should be treated. Endolaser is used for posterior breaks, whereas peripheral breaks are treated with either a curved endolaser probe, indirect laser assisted by scleral depression, or transscleral cryoretinopexy. The xenon-illuminated endolaser probes are needed to treat retina pathology when the view is poor from anterior segment opacification either from catararact formation, corneal edema, or stromal scarring. Cryotherapy is preferred when residual opaque vitreous partially obscures the targeted break.
Encircling scleral buckles are not necessary after vitrectomy for nonclearing vitreous hemorrhage caused by ocular contusion when a clear view of the fundus periphery reveals no peripheral retinal tears or signs of traction. Similarly, the support of a buckle is usually not needed for treated retinal breaks without retinal detachment. A segmental scleral buckle element may be used to support areas with residual traction on a peripheral break. An encircling scleral buckle should support the peripheral retina when traction on breaks in the oral zone persists or the periphery is poorly visualized because of residual opaque vitreous.
Retinal Breaks
Retinal breaks are created at the time of nonpenetrating blunt injuries in 10% to 20% of eyes.
7,
8,
9 and
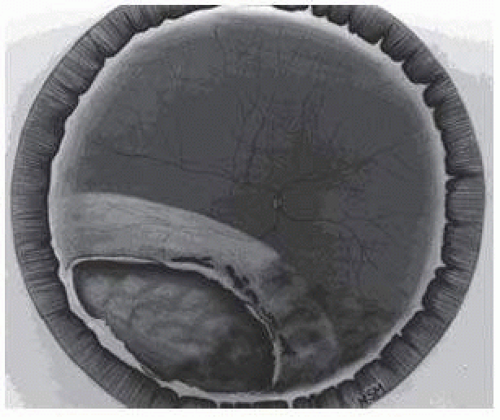
10 Retinal dialyses are most frequent, seen mostly in the lower temporal (
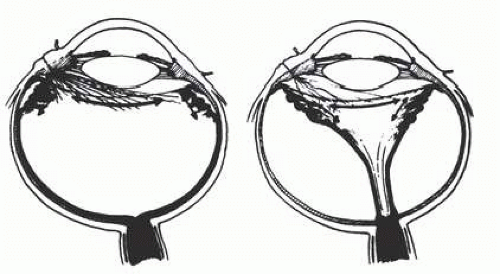
Fig. 66.2) and upper nasal periphery (
Fig. 66.3).
5,
11 Large irregular breaks at the point of impact of blunt trauma are less common but equally characteristic of nonpenetrating injuries (
Fig. 66.3).
5,
12 Horseshoe and operculated tears of the equatorial retina (
Fig. 66.3) are characteristically seen in 25% of eyes.
5 Small round holes in atrophic retina at the point of traumatic impact and macular holes (
Fig. 66.3) are infrequently observed after ocular contusion.
5,
12Prophylactic treatment is indicated for most traumatic retinal breaks. Breaks at the point of impact are one exception because they are frequently self-sealing. The surrounding necrotic retina and choroid often unite in a common scar without prophylaxis. It is wise, however, to treat these large tears when scleral depression reveals a slight elevation and movement of their edges and the surrounding retina.
Retinal Detachment
Retinal detachment may be seen in the setting of closed globe eye injury. Trauma may cause a rapid shortening of anteroposterior axis of the globe that is followed by equatorial elongation, leading to peripheral retinal tears and dialyses. Traumatic injuries are more commonly observed in a younger patient population. The vitreous gel is normally dense in young patients and may tamponade the retinal break. Later liquefaction may lead to retinal detachment months following the initial injury.
5 The detachment is typically shallow and slowly progressive because the large volume of formed vitreous gel in younger eyes inhibits the bullous retinal elevation that commonly occurs in older patients with nontraumatic retinal detachments.
Closed globe injury may also lead to traumatic retinal dialyses that are classically seen in the inferotemporal or superonasal quadrants.
11 Careful preoperative and
intraoperative indirect ophthalmoscopy with scleral depression is important to identify the etiologic rhegmatogenous sites for the successful treatment of detachments caused by traumatic retinal dialyses. Small dialyses at the vitreous base borders may be difficult to identify, particularly in the superonasal quadrant, and breaks of the pars plana epithelium, at the anterior vitreous base border, are less apparent than retinal tears at the posterior edge of the vitreous base. During scleral depression, small dialyses may be closed, and thus they may be more easily seen on the lateral slopes of the scleral indentation than on its crest. Transscleral cryotherapy is diagnostically helpful. It is not unusual to discover breaks at the vitreous base border for the first time when the edges of the tear are whitened by freezing a suspicious area.
It is prudent to treat the entire zone of vitreous base pathology with cryotherapy under direct visualization (
Fig. 66.3). In this way, treatment of all retinal breaks is assured. The anterior, posterior, and lateral limits of the treated zone are carefully localized and then supported by a broad scleral buckle to relieve traction to the entire area. The posterior edge of the dialysis should fall on the crest of the buckle, which must be sufficiently broad to support the anterior edge as well to prevent a recurrent detachment from anterior tracking of subretinal fluid. Segments of grooved solid silicone tires and an encircling band are positioned on the great circle of the globe to minimize anterior or posterior migration of the buckle. The band is anchored by a nonabsorbable mattress suture or scleral belt loop in each of the quadrants not occupied by the tire segment. A high encircling buckle promotes posterior gaping or “fish-mouthing” of the dialysis and is thus avoided.
The retinal detachment is often shallow. To avoid retinal perforation or incarceration, subretinal fluid is released through a sclerotomy in an area of sufficient retinal elevation, as determined by intraoperative indirect ophthalmoscopy with scleral depression. Viewed in profile, the scleral indentation helps gauge the distance between retina and retinal pigment epithelium. To avoid retinal incarceration and blowout, the drainage sclerotomy should be left unsutured when made in the bed of the buckle, and securely sutured if made posterior to the buckle, particularly if additional manipulation of the buckle or an intravitreal gas injection is anticipated. Nondrainage scleral buckling procedures are also acceptable in cases without retinal incarceration.
Retinal dialyses in the lower temporal quadrant are often large, with gaping posterior edges located well behind the equator (
Fig. 66.2). They are caused by injuries to the inferotemporal globe that result in the dissolution and disappearance of retinal tissue.
12 In contrast to nontraumatic giant retinal tears with rolled-over retinal edges, they respond favorably to scleral buckling without vitrectomy. A scleral buckle is indicated for smaller dialyses that can be closed with an explant of reasonable size. Very large or posterior breaks, as illustrated in
Figure 66.2, are best treated with vitrectomy, gas or silicone oil tamponade, and laser, as recommended for nontraumatic giant retinal tears, rather than with a very large scleral buckle.
Vitrectomy is often used to repair traumatic rhegmatogenous retinal detachments, particularly when there is an associated vitreous hemorrhage, giant retinal tear, or proliferative vitreoretinopathy. Intraocular gases, silicone oil, and perfluorocarbon liquids may all aid in the repair.
Traumatic Macular Hole
First described by Herman Knapp
13 in 1869, traumatic macular hole is a well-recognized complication of closed globe ocular trauma. It is thought to result from tangential traction to the posterior pole occurring with globe deformation from the blunt trauma.
14 Outward expansion of the equator is followed by flattening and subsequent posterior displacement of the posterior pole. The trampoline-like movement of the posterior pole creates tangential tractional forces on the retinal surface that lead to the formation of the macular hole. Spontaneous closure of the hole is not uncommon; therefore, a period of observation for several weeks following injury is appropriate prior to surgical repair.
15 Optical coherence tomography is a useful diagnostic tool to follow the progress of traumatic macular holes and diagnose vitreomacular traction.
16,
17 The presence of vitreomacular traction and/or the failure of the macular hole to close after a sufficient period of observation are indications for surgical repair.
Small gauge 23G or 25G transconjunctival vitrectomy may be used to repair the traumatic macular hole. A core vitrectomy, followed by removal of the posterior hyaloid is performed. The vitrectomy may then be carried anteriorly as per standard macular hole surgery technique. While the importance of routinely removing the internal limiting membrane (ILM) during the repair of idiopathic macular holes is controversial, we prefer to remove the ILM during the repair of traumatic macular holes.
18,
19 We typically utilize 0.2 cm
3 of 0.125% indocyanine green (ICG) dye to stain the ILM. We instill the dye with the infusion off, and allow it to sit on the surface of the retina for several seconds before removing the dye with aspiration. The ICG may highlight residual vitreous unintentionally left behind but will generally stain the ILM. The ILM is then pinched down upon using an end-grasping or ILM forceps, and removed from the entire macula in a
rhexis maneuver. Medium- to long-term gas tamponade, combined with facedown positioning, is performed in each case. More recent studies demonstrate a high rate of macular hole closure with facedown positioning of 3 days’ duration.
20 In pediatric patients, 0.4 IU of intravitreally injected plasmin enzyme may facilitate the induction of a posterior vitreous detachment during surgery.
21