 21
21 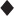
Management of Glaucoma in Pregnancy
Brian R. Sullivan
Introduction
This topic deals with the diagnostic and treatment concerns evolving from the occurrence of primary or secondary glaucoma in women of reproductive age, especially during the natal and postnatal periods.
Definition
Why Is Pregnancy an Issue of Concern in Glaucoma?
Therapeutic dilemmas may arise during the management of glaucoma in women of childbearing age due to the risks of impaired fertility, maternal and fetal toxicity, teratogenicity, and harmful effects on the nursing infant. Treatment of glaucoma in pregnant and nursing women should be approached with caution. This problem may become more common in the future as more women are choosing to defer childbearing until later in life.
Epidemiology and Importance
How Common Is Glaucoma in Pregnant Women?
The coexistence of glaucoma and pregnancy is an unusual clinical problem, and glaucoma is rarely initially diagnosed during pregnancy. Although the fertility period extends from the second to the fifth decades of life, the prevalence of glaucoma in this population of women is undefined. The clinical variability of glaucomatous disorders adds to the difficulty in determining confusion over prevalence data in this group of young females. The term glaucoma refers not to one entity, but to a large group of diverse disorders with the common features of ocular hypertension, visual field loss, and optic neuropathy. In addition,there is increasing evidence that primary open-angle glaucoma (POAG), the most common glaucoma diagnosis, may be a genetically heterogeneous collection of clinically similar or even indistinguishable diseases. The general demographic and socioeconomic characteristics of POAG and other forms of glaucoma are discussed elsewhere in this text, but such data should be interpreted cautiously in addressing glaucoma issues in women of reproductive age. Unfortunately, there is remarkably little pregnancy-specific epidemiologic data available regarding the various glaucomas, but some conclusions can be drawn from existing knowledge of glaucoma and pregnancy demographics.
Because POAG is associated with aging and the elderly, the disease is generally considered uncommon in pregnancy. The prevalence of POAG increases with age,1 and it is very uncommon under age 40. Of one series of young glaucoma patients between ages 10 and 35, only 25% had the diagnosis of POAG.2 However, POAG occurring in younger patients has been observed to be associated with high initial intraocular pressure (IOP) and may represent a more severe form of the disease.3 Therefore, POAG that is coincident to the condition of pregnancy, although rare, may require relatively aggressive management.
Has the Incidence of Glaucoma in Pregnancy Increased in Recent Years?
Currently there are no data available supporting an increasing coincidence of pregnancy and glaucoma. It might be suggested that the clinical scenario of POAG during pregnancy could become more common in association with increasing childbearing in middle adulthood. There is a national trend of increase of births in women who have delayed starting their families until later in their careers,4 but the incidence of glaucoma has not yet been studied in this population of mothers.
What Type of Glaucoma Occurs During Pregnancy?
In the general population, POAG is the most common form of glaucoma, but, for the reasons discussed above, this observation cannot be extrapolated when considering the population of young women. It has been suggested that juvenile open-angle glaucoma is the predominant type of glaucoma occurring among women of childbearing potential.5 Glaucoma that occurs during adolescence and young adulthood often is also the result of underlying ocular or systemic disease, and in many cases abnormalities of the anterior chamber angle can be identified. Therefore, when referring to women of childbearing age, the developmental and secondary glaucomas are of increasing significance, particularly those associated with inflammatory ocular diseases, chronic steroid use, prior intraocular surgery, and/or trauma. It should be noted that there are no reported cases of pregnancy-induced open-angle glaucoma, and there are no forms of secondary glaucoma unique to pregnancy. Narrow-angle glaucoma may worsen with advancing pregnancy complicated by preeclampsia,6 but this relationship has not been clearly established by published findings. Acute-angle-closure glaucoma has been reported in one case to be precipitated by labor, perhaps triggered by emotional and physical stress at delivery.7 However, acute angle closure more typically occurs past the fifth and sixth decades of life, and this problem is rare in young females.
Is There a Relationship Between Glaucoma and Pregnancy-Induced Hypertension in Preeclampsia or Eclampsia?
The positive correlation between ocular hypertension and systemic hypertension8–10 is an issue worthy of consideration in addressing glaucoma in pregnancy because the incidence of pregnancy-induced or -aggravated hypertension has been reported to be as high as 5 to 10% of a general population of pregnant women, and over 20% in nulliparous women.4 It has been observed by Qureshi et al11 that ocular tensions of third trimester hypertensive women were significantly higher than tensions of third trimester nonhypertensives. In this report, a total of 200 women were studied, including 40 nonpregnant controls. A difference of mean IOP of 0.6 mm Hg, measured by Goldmann applanation, was found between late pregnancy normotensives and hypertensives, and, although small, this difference was found to be statistically significant. However, this study contradicted earlier work by Phillips and Gore12 demonstrating no significant difference of mean IOP between third trimester hypertensive and nonhypertensive women. In their report, hand-held Perkins applanation tensions were obtained in a total of 97 women including 25 nonpregnant normotensive controls. Mean late pregnancy IOP was 12.1 and 12.4 mm Hg among normotensives and hypertensives, respectively, and the small difference was not found to be statistically significant. A finding of pregnancy-induced lowering of IOP in the third trimester was observed in both hypertensive and nonhypertensive groups in both of the above studies. In both reports the drop was found to be statistically significant, and the mean decrease in IOP ranged between 2.0 and 2.7 mm Hg (13–18%). This interesting phenomenon is further discussed below.
Does Pregnancy-Induced Diabetes Mellitus Affect IOP?
An increased prevalence of POAG and ocular hypertension in general populations of diabetics has also been suggested,8,13–15 but this relationship has not been demonstrated in pregnancy-induced diabetes mellitus.
Could Additional Epidemiologic Data Improve Patient Care?
The currently available information is of insufficient quality to estimate incidences of various types of glaucoma in women during reproductive years. It can only be stated that glaucoma during pregnancy is considered uncommon. As childbearing at older maternal age becomes more frequent, it is not known if the coincidence of glaucoma and pregnancy is increasing accordingly. Advanced age has not yet been identified as a risk factor for glaucoma during the condition of pregnancy, and the significance of other glaucoma risk factors in pregnancy is unclear. IOP normally decreases during pregnancy, and it is unlikely that systemic hypertension during pregnancy affects IOP in a clinically significant manner. Further epidemiologic study could enable better glaucoma management decisions for the patient who is pregnant or at risk of pregnancy. Determination of pregnancy-specific prevalences, risks, and prognostic factors would be useful for improved glaucoma assessment, and possibly in avoiding unnecessary and potentially hazardous treatment.
Diagnosis and Differential Diagnosis
How Are Various Glaucomas Diagnosed in the Pregnant Patient?
Assessment of glaucoma in the pregnant patient involves application of the same clinical principles as in any patient with suspected glaucoma. Such evaluation should include appropriate classification and diagnosis of the type of glaucoma, and determination of the stage of the disease by optic nerve examination and perimetric analysis. Potential risk factors must be identified including IOP, refractive error, other ocular disease, age, race, family history, and systemic disease. In the young female patient, a particular effort should be made to elicit histories of prior glaucoma or ocular hypertension, nonglaucomatous ocular disease, and eye trauma. A review of records from previous eye examinations is important to characterize the duration and progression of the disorder. Medical history and review of systems are also pertinent, particularly in cases of secondary glaucoma.
Features of the glaucoma exam are discussed in more detail elsewhere in this text, and, although younger and healthier, the pregnant woman should be evaluated equally as thoroughly as the more typical elderly glaucoma patient. Careful examination of pupils, external features, anterior segment, and posterior segment should be performed with particular attention to intraocular tension, gonioscopy, and optic nerve appearance. The optic nerve exam should be detailed to include characteristics of cupping, color, contour, vascular changes, and disc hemorrhages. Any asymmetric findings should be carefully noted.
Classification of glaucoma in any patient is based on the underlying mechanisms leading to the common pathways of optic neuropathy and visual field loss. Discrimination should be made between open- and closed-angle glaucoma with attention to identifying any underlying ocular, systemic, or genetic disorders. In dealing with glaucoma in younger ages associated with pregnancy, one must be mindful that the relative occurrences of developmental glaucomas and secondary glaucomas are likely to be higher than that of POAG in women of childbearing years.2 In cases of suspected or known secondary glaucoma, it is important to document all orbital and ocular abnormalities, especially inflammatory, posttraumatic, and neovascular findings. Pregnant or potentially pregnant patients with uveitis of unknown etiology should always undergo extensive systemic evaluation using laboratory and other ancillary studies to rule out identifiable underlying conditions that could threaten the welfare of mother, fetus, or nursing infant.
In Addition to Physical Examination, What Other Studies Are Useful?
Because progressive loss of visual field is a common potential outcome for all forms of glaucoma, visual field testing is the most important adjunctive diagnostic tool in detecting and following the disease. In managing the pregnant glaucoma patient, perimetry remains equally invaluable because it is a safe, sensitive, and noninvasive test. Because various forms of glaucoma do not characteristically worsen at an accelerated rate during pregnancy, it is not usually necessary to obtain serial perimetry data in greater frequency than in the nonpregnant glaucoma patient. Visual field screening during pregnancy may also yield results atypical for glaucoma, and the reported findings have been well discussed in a review of the effects of pregnancy on the eye.16
Nonglaucomatous visual field loss in pregnancy has been described in older literature as early as 1923,17–19 but contemporary reports are surprisingly lacking. The reported abnormalities include the findings of bitemporal or concentric field loss, usually detected near term. Other studies failed to verify such findings.19 Such changes, when discovered, are usually asymptomatic and usually resolve shortly after delivery.16 Initial investigators suggested that the field defects might be attributed to physiologic enlargement of the pituitary gland during normal pregnancy. However, in pregnancy, the degree of enlargement of an otherwise normal pituitary gland is insufficient to affect the chiasm, and visual field abnormalities are not explained by this mechanism.19 It is recommended, therefore, that any woman who demonstrates unexplained visual field abnormalities during pregnancy should undergo further evaluation for underlying disease affecting the visual sensory system, particularly central nervous system lesions.
Other ancillary tests, which may also be helpful in evaluating glaucoma disorders, include optic nerve photography, and computerized disc and nerve fiber layer analysis.
Does Pregnancy Have Any Effect on IOP?
The interpretation of IOP during pregnancy warrants additional discussion. It has been demonstrated that in all trimesters of pregnancy, intraocular tensions are lower than in nonpregnant controls. The ophthalmology literature consistently describes a trend of decreasing IOP with advancing pregnancy, especially in the second and third trimesters.12,16,20,21 Using applanation tonometry, Phillips and Gore12 and Qureshi et al11 recorded IOP data in totals of 97 and 200 women, respectively. Both reported third trimester drops in IOP ranging between 13% and 18% compared to nonpregnant controls, with a lesser degree of decrease of mean IOP in hypertensive patients.12 Additionally, the finding of decreased IOP has been observed to persist for several months after delivery.22 Wilke23 observed reduced episcleral venous pressure in pregnancy by measuring repeated applanation tensions in 20 pregnant women in the third trimester and comparing to those of 20 nonpregnant controls. It was suggested that decreased episcleral venous pressure contributes to the ocular hypotensive effect of pregnancy, as a result of the observed systemic phenomenon of lowering peripheral vascular resistance.23,24 Patterson and Miller25 performed repeated tonography throughout pregnancy on seven healthy primigravid women, and found an initial steep increase of outflow facility with peaks at approximately 20 and 26 weeks. The observed percentage increase of the coefficient of outflow ranged between 9% and 133%. The facility of outflow values declined near parturation, with further decreases in the postpartum period to near-normal values.
There is substantial evidence of a hormonal mechanism20 for pregnancy-induced lowering of IOP. Systemic progesterone, which gradually rises endogenously throughout pregnancy, has been noted to decrease IOP in glaucoma patients.26,27 Relaxin, another hormone that increases during pregnancy, has been shown to reduce IOP and increase outflow facility in glaucoma subjects.20 A third hormone of pregnancy, β-human chorionic gonadotropin (β-hCG), reduces IOP by suppressing aqueous production.28
How Does Pregnancy Affect the Glaucoma Patient?
The above observations have several important implications for the pregnant glaucoma patient. First, there has been a general impression that preexisting glaucoma improves during pregnancy.16,20,29 Women with the existing diagnosis of glaucoma may require fewer or no glaucoma medications for adequate IOP control while pregnant. Second, the IOP-reducing effect of pregnancy may cause normal or near-normal tension measurements when the pregnant woman with glaucoma is initially evaluated, potentially masking the disorder. This phenomenon could result in missed diagnosis or underestimation of the severity of the glaucoma, especially in those cases in which earlier clinical information is unavailable. Third, because the IOP-lowering benefit diminishes slowly after delivery, women with glaucoma need careful monitoring of IOP control in the postpartum months. Glaucoma suspects should be followed similarly because ocular hypertension may appear or worsen.
Treatment and Management
Is She Pregnant? Does She Think She Might be Pregnant?
Whenever considering any treatment of a woman of childbearing age, it is important to be mindful of the possibilities that she could be pregnant or become pregnant during the treatment period. This issue is especially significant in treatment of chronic diseases such as POAG. Failure by any physician to address these possibilities can result in inappropriate management and medicolegal concerns. Specific history taking prior to initiating management is therefore crucial. Female patients do not typically volunteer the possibility of pregnancy during ophthalmic consultation. Questions should specifically determine the woman’s estimated likelihood that she may be pregnant or contemplating future childbearing. The date of the most recent menstrual period should be documented to note any delay or cessation of menses. Current specific method of contraception should also be noted if considering the use of a known or suspected teratogenic agent. The diagnosis of pregnancy may also be inexpensively determined with fairly high accuracy via laboratory pregnancy testing by detection of urine or blood β-hCG. Referral to the patient’s primary care physician or obstetrician should be considered if the diagnosis of pregnancy is in question.
Ideally, in treating glaucoma in young women, patients should be counseled prior to conception regarding any known or estimated risks of drug therapy. However, in practice, medications taken chronically are usually not discontinued prior to the time pregnancy is diagnosed, and exposure occurs well beyond the period of conception and critical organogenesis. Therefore, the prescribing ophthalmologist should be vigilant of any teratogenic or toxic potential of all medications used in managing glaucoma in women of reproductive age, regardless of known or planned pregnancy. Particular caution should be taken in prescribing any medication during a recognized pregnancy, because litigation can result from an adverse outcome despite nonsupporting or contrary scientific evidence for toxicity and teratogenicity. Hence the term “litogen.”30
How Can It be Determined if Glaucoma Medications Cause Harm During Pregnancy?
Any glaucoma drug that is absorbed systemically should be considered to have the potential to affect the fetus or to be excreted in breast milk. In theory, the biochemical properties of most glaucoma drugs should allow transfer across the placenta without impediment.31 However, the use of any topical ophthalmic medication is uncommon during pregnancy, and data regarding teratogenic or toxic effects are generally inadequate. Early evidence of human teratogenic effects of a pharmacologic agent arises from clinical case reports, but high-quality epidemiologic studies are necessary to determine meaningful and statistically significant associations between drug exposure and offspring abnormalities.32 Because the coincidence of glaucoma and pregnancy is uncommon, studies of large human populations are not possible to assess the toxicities of even common glaucoma medications to the fetus or nursing infant. Conversely, agents that are inappropriately “accused” of being human teratogens may also be associated with sparse scientific literature, and the proposed cause-effect relationship should be viewed critically. Those drugs unlikely to pose a human hazard are often not exonerated by scientific support due to insufficient data.32 It is important for the patient and practitioner to be mindful that pregnancy has inherent risks, and the majority of congenital malformations occur in the absence of drug or chemical exposure.33
In counseling patients regarding drugs with insufficient human scientific data involving risks in pregnancy, it may be of some benefit to examine the results of animal tests. Teratogenic studies in laboratory animals are available regarding many glaucoma drugs, but the results cannot always be extrapolated to infer risk in humans.34 This problem is partly due to the usually high doses used in most animal experiments, resulting in systemic levels incomparably higher than those resulting from typical dosages of medications used in ophthalmology. Similarly, comparison of data from human experience is difficult because an ophthalmic drug administered topically is generally associated with systemic levels fractional to levels achieved by oral or other routes used for nonophthalmic indications. Much of the useful information regarding adverse reactions in humans arises from case reports and epidemiologic studies generated after a drug has come into general use, but because the coincidence of glaucoma and pregnancy is uncommon, specific data remain sparse despite even widespread use of a glaucoma agent. Unfortunately, premarketing evaluation of drug effects on reproductive function and pregnancy, whether in vitro, in animals, or in humans, is usually inadequate to properly estimate risks of teratogenicity and other toxic effects.35 Drug testing is intentionally avoided in the specific population of pregnant women.36
If there is no human or animal evidence to evaluate potential pregnancy-related hazards of a drug, it is difficult to assess risk at any exposure levels. Patients should be advised that the risk is not known. Sometimes, more knowledge may exist regarding safety of other drugs in the same class of compounds. If the drug has a physical structure or mechanism of action similar to that of a known toxicant or teratogen, then it is reasonable to suspect increased potential for adverse effects, despite the absence of formal evidence. Because patients seek rational guidance from their eye care providers, it is important to disclose the amount and quality of scientific information supporting the advice provided, especially when hazard data is incomplete.
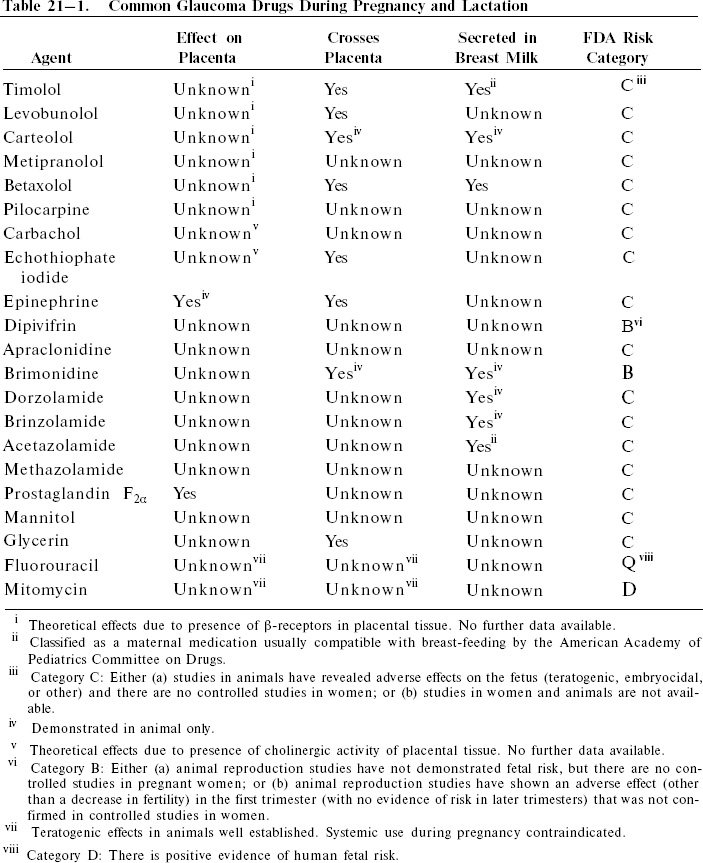
A summary of bioavailability and Food and Drug Administration (FDA) category data of common drugs used in glaucoma treatment are listed in Table 21–1.
What Glaucoma Management Issues Should be Considered for Women During the Reproductive Years?
Recommendations for ophthalmic drug use near or during pregnancy, therefore, evolve from limited available data, with the assumption that eye drops are absorbed systemically via the nasolacrimal duct system and nasal mucosa.37 The patient and her ophthalmologist must attempt to resolve the dilemma of benefits versus risks on an individual basis. For a young woman with no near-term plans for childbearing, the potential of a drug for impairment of future reproductive capacity must be considered, although usually this risk is unknown. When a woman becomes pregnant or plans pregnancy, the glaucoma management plan should be reassessed. Any nonessential ophthalmic drug should be avoided. Particular attention should be paid to ensure that drugs with known contraindications are not used, with cautious and reserved use of any others. Judgment of the benefit to risk ratio involves weighing the often poorly defined teratogenicity and toxicity risks against the possible visual sequelae resulting from deferral of glaucoma treatment. Women in earlier stages of glaucoma may be able to tolerate short-term moderate elevations of ocular tensions for up to 9 months if glaucoma drugs are temporarily discontinued.
Because of the intrinsic IOP lowering effect of pregnancy itself, the IOP control may be found to be surprisingly stable after withholding medications. If drug therapy is deemed necessary, the safest possible choice should be made, and the medication should be administered in the lowest dose estimated to be effective. Dosage and duration of use should be minimized as much as is consistent with acceptable glaucoma care.38 Women should be clearly instructed in nasolacrimal occlusion after instillation of eye drops to minimize systemic absorption. It may be necessary to advise against breast-feeding because of theoretical harm from drug distribution into milk, especially if the uses of β-antagonists or carbonic anhydrase inhibitors cannot be withdrawn. Pregnancy also presents an opportunity to consider early use of laser trabeculoplasty, iridotomy, and other nonpharmacologic approaches to glaucoma management. As in all cases of glaucoma, those women with late-stage optic nerve cupping or poor IOP control, with risk factors for progression, should be treated more aggressively. Filtration surgery, which could decrease or eliminate dependency on pharmacologic agents, particularly carbonic anhydrase inhibitors, should be considered as an early alternative to medical management in women contemplating pregnancy. Surgical intervention should be strongly considered in advanced glaucoma cases and optimally should be performed well before the onset of pregnancy.39 However, surgical management, even when considered successful, often does not eliminate dependency on drug therapy for glaucoma.
In summary, the ophthalmologist must consider reproductive issues in decision making and patient counseling for all young women with glaucoma requiring medical treatment. Contemporary methods of glaucoma management should enable good treatment before and during pregnancy with satisfactory perinatal maternal and fetal outcomes, even in advanced cases of glaucoma.
What Is Known About β-Antagonist Use in the Pregnant Glaucoma Patient?
Despite limited maternal-fetal toxicity data, some classes of glaucoma drugs such as beta-blockers are generally thought to be acceptable for use with caution during pregnancy. Concerns over maternal adverse effects of β-antagonists are the same as those in the nonpregnant patient, including the potentials for precipitation or aggravation of obstructive lung disease or heart failure, bradycardia, drowsiness, and interference with diabetic control.40,41 Such maternal effects are thought to be dose dependent, with the potential for compromise of fetal well-being. There are data showing that atenolol (Tenormin), an oral cardioselective beta-blocker, is associated with a significant increase in intrauterine growth retardation when prescribed from the first trimester,42 and it has been suggested that other beta-blockers are likely to have the same effect.41
Several investigators have shown an abundance of β-adrenergic receptors in human placental tissue,43–46 even in early gestation.47 In addition, several of the commonly used ophthalmic β-antagonists are known or suspected to traverse the placenta.31,48 In an animal model, timolol was observed to prevent the rebound tachycardia normally seen after hypoxia in the fetus.49 However, the effects, if any, of topical ophthalmic β-antagonists on the placenta or fetus have not been specifically observed. The reported clinical experience with ophthalmic β-antagonists in pregnancy is, overall, somewhat limited despite their widespread use, especially in younger glaucoma patients. Beta-blockers have been anecdotally reported to be relatively safe for use during pregnancy,50–53 but contraindicated after delivery in nursing mothers.16,38,54,55 Maternal use of oral β-antagonists near delivery has been associated with persistent and potentially hazardous beta-blockade in the newborn infant.56–59 Timolol (Timoptic) has been demonstrated to be secreted in breast milk, with milk levels up to six times that of serum after topical ophthalmic use.54,60 Betaxolol (Betoptic) was also found to be concentrated in breast milk, with a milk/plasma ratio of 3.0.52 Although the observed milk levels in these cases remained well below cardioeffective dosages, topical β-antagonists should be prescribed very cautiously during nursing due to the serious and possible lethal side effects of beta-blockade in infants, particularly apnea.61,62 Though not contraindicated, the use of β-antagonists near delivery should be avoided when possible, and infants exposed in utero should be monitored closely during the first 24 to 48 hours after birth for apnea, bradycardia, and other symptoms. Despite the above concerns, the American Academy of Pediatrics Committee on Drugs has classified timolol as a maternal medication usually compatible with breast-feeding.63
Are Miotics Prescribed in Pregnancy?
Ophthalmic use of parasympathomimetic agents has not been demonstrated to be teratogenic in humans, but, although not contraindicated, this class of glaucoma medications should not be used indiscriminately during pregnancy. Consideration could be made in withdrawing miotic use when the woman is near term, due to possible transient effects on the newborn infant. Pilocarpine has been associated with signs mimicking neonatal meningitis, and transient newborn muscular weakness has been observed in association with cholin-esterase inhibitor use during pregnancies of mothers treated for myasthenia gravis.55 Case evidence has been published demonstrating that echothiophate iodide crosses the human placental barrier with apparent inhibition of fetal pseudocholinesterase activity.64 No clinical complications of mother or infant were noted, but caution was advised by the authors due to unknown fetal hazards. There is also well-documented cholinergic activity of the human placenta, but the functions of these cholinergic components are not yet well understood.65 Among other suggested roles, the placental cholinergic system has been linked to myometrial and placental prostaglandin release during parturation, release of placental hormones, and regulation of blood flow in placental vessels.66 It is not known whether ophthalmic cholinergic agonists have any influence on the placenta function and hormonal milieu. Carbachol and pilocarpine have been associated with developmental abnormalities of animal embryos,67–69 but to date these drugs have not been implicated as human teratogens. Case reports of safe use of miotics during pregnancy and lactation have been described in the ophthalmic literature,54,70 but caution is warranted due to unknown, perhaps unlikely, placental and fetal effects.
What Information Exists Regarding Pregnancy and the Use of Adrenergic Agents?
Topical drugs in a third class of topical glaucoma medications, the sympathomimetics, are also considered relatively acceptable for use in pregnancy, but reported experience is sparse. First trimester use of ophthalmic epinephrine has been described with no adverse outcome.54 Epinephrine readily crosses the placenta,71 but because epinephrine occurs endogenously, it is difficult to distinguish effects of the administered drug from naturally occurring epinephrine. One concern is that epinephrine administration during pregnancy might decrease uterine perfusion. In a pregnant animal model, a greater than 34% reduction of uterine blood flow was observed after administration of a low dose of epinephrine that did not affect blood pressure.72 Studies of possible impairment by epinephrine of fetal gas exchange have been conflicting.73,74 Animal studies have failed to demonstrate harmful fetal effects of dipivefrin (Propine), but human experience is limited, with no controlled studies in pregnant women.75 Nursing women should be treated with caution because it is not known if topical ophthalmic epinephrine or dipivefrin is significantly secreted into milk.
Experience with α2-adrenergic agonists is similarly limited. Animal studies have shown no impairment of fertility or fetal harm of brimonidine tartrate (Alphagan), but the drug, in animal models, is known to cross the placenta and is excreted in breast milk.76 Unlike β-receptors, α-adrenergic receptors are not found on the maternal or fetal sides of the human placenta.77 There is evidence of an α-adrenergic role in the mechanism and regulation of prolactin secretion,78 but it is not known if ophthalmic brimonidine use has any significant influence on plasma prolactin levels in animals or humans. Reproductive studies of apraclonidine (Iopidine) in animals have shown no impairment of fertility, but embryocidal effects and maternal toxicity have been observed with high doses during pregnancy.79 There are no adequate human studies involving either drug in pregnant women. It is recommended that α-adrenergic agonists be used with caution during pregnancy and nursing, weighing unknown potential fetal risks with the value of treatment of the mother.
Have Carbonic Anhydrase Inhibitors Been Used in Pregnancy?
Oral carbonic anhydrase inhibitors are relatively contraindicated during pregnancy and lactation. Acetazolamide (Diamox) and methazolamide (Neptazane) have well-documented teratogenic potential in animals,80–84 and a case has been reported of an infant with sacrococcygeal teratoma, born to a mother treated with acetazolamide, among other medications, during the first trimester.85 However, the proposed carcinogenic effect of acetazolamide on the fetus has not been substantiated by other reports, and retrospective studies have suggested no increased human fetal risks.86,87 Use of acetazolamide after the first trimester has been described in a number of cases without adverse effects.64,87–89 Two cases of neonatal dehydration and metabolic disturbances have been reported, involving mothers who had been treated near term with acetazolamide.89,90 Acetazolamide has been shown to be secreted into human breast milk to levels of approximately one-third of maternal plasma concentrations,91 signifying the potential for infant exposure to the drug. In glaucoma treatment, oral carbonic anhydrase inhibitors should be avoided in nursing women whenever possible, especially in cases of impaired neonatal respiratory, hepatic, or renal function. It should be noted, however, that the American Academy of Pediatrics Committee on Drugs has classified acetazolamide as a maternal medication usually compatible with breast-feeding.63
The relative hazards of topical carbonic anhydrase inhibitors are even less clear because systemic drug levels are expected to be somewhat below those of oral medications. Brinzolamide (Azopt) has been found in milk of lactating rats at concentrations below those of plasma, but it is not known if brinzolamide is excreted into human milk during maternal use.92 Animal studies showed no adverse developmental or mutagenic potential of oral dorzolamide at 12 to 13 times the human ophthalmic dose, but treatment-related malformations were reported at much higher doses.93 The limited information suggests some degree of safety of dorzolamide (Trusopt) in the low doses used in ophthalmology. Again, caution must be taken in interpreting data from animal studies and in extrapolating the findings to humans. There are no adequate human studies evaluating adverse effects of topical carbonic anhydrase inhibitors on reproductive capacity or fetal development, and abnormal reactions of nursing infants are not known. Given the limited evidence for safety, it is recommended that topical carbonic anhydrase inhibitors be considered for glaucoma therapy with marked caution during pregnancy, with use limited only to the second and third trimesters. After delivery, options should include decisions to discontinue breast-feeding or to discontinue the drug in the nursing mother. In summary, alternative treatment to oral or topical carbonic anhydrase inhibitor use should generally be sought in managing glaucoma in childbearing women.
Can Pregnant Women be Treated with Prostaglandins for Glaucoma?
Chronic use of prostaglandin agents in the management of glaucoma, particularly POAG, has become increasingly popular in recent years. Human experience with the use of topical prostaglandin analogues in pregnancy is not described. Endogenous prostaglandins appear to have important roles in the reproductive cycle, including both normal and pathologic states, but mechanisms of action remain poorly understood. It is suspected that the vascular abnormalities in preeclampsia may be mediated by prostaglandin effects, and increasing data indicate that prostaglandins play a major role in parturation. Blood and amniotic fluid levels of PGF2α and other prostaglandins are elevated near term and during labor. Therapeutic uses of PGE2, PGF2α, and prostaglandin analogues have been described extensively for the induction of labor and treatment of pregnancy complications.94 Several such agents have also been used as abortifacients. Obstetrical use of prostaglandins remains limited by the associated side effects and complications, as well as by persistent confusion over their complex mechanisms, despite a large body of clinical data.94
Ophthalmic uses of prostaglandin analogues in fertile or pregnant women should be approached cautiously. Latanoprost (Xalatan), a PGF2α analogue, has been observed to have an embryocidal effect in animals at 15 to 80 times the maximal human dose, but no human pregnancy data are available.95 In mice and rats, PGF2α caused malformations and increased abortion,96,97 but was not teratogenic in rabbits or hamsters.98,99 PGF2α is a known potent stimulator of uterine contraction,100 and it has also been demonstrated to be a vasoconstrictor of the fetoplacental vascular bed,94 the ductus arteriosus,101,102 and the fetal pulmonary vessels.103 Thus, the issue of systemic absorption of prostaglandin analogues raises well-founded concern over increasing risks of pregnancy complications. It is not known if latanoprost or other prostaglandin analogues reach circulating levels of any clinical significance when administered in topical ophthalmic doses. The biologically active form of latanoprost is rapidly eliminated from human plasma (t1/2 = 17 minutes),95 suggesting perhaps minimal potential effect on the reproductive cycle. However, because of the known bioactivity of prostaglandin drugs in pregnancy, further study is needed to assess the safety of ophthalmic use of this class of drugs. It is this author’s recommendation that, until further data are available, latanoprost and other prostaglandin agents be avoided during pregnancy. If an ophthalmic prostaglandin is considered for use due to unacceptable alternatives, careful obstetrical comanagement of the patient is recommended to monitor for maternal-fetal side effects.
Is There a Role for Hyperosmotics in Glaucoma Management During Pregnancy?
Hyperosmotic agents have been used in ophthalmology usually for emergency or short-term treatment of glaucoma. Such drugs are particularly effective in acute angle-closure glaucoma. Despite well-known systemic effects and complications, discussions of potential problems with the use of these agents during pregnancy are nearly absent in the ophthalmic literature. Mannitol induces abortion when instilled in the amniotic cavity, but may result in only diuresis if given intravenously in pregnancy.104 In rats, intravenous mannitol has been observed to cause hemorrhagic abnormalities of fetal limbs, but malformations were not seen.105 Administration of hypertonic agents, including mannitol, to pregnant sheep has been observed to cause fluid shifts involving fetal compartments.106,107 Additional evidence does not suggest any specific teratogenic effects of mannitol. Human pregnancy experience with use of oral hyperosmotic agents such as glycerol (Osmoglyn) and isosorbide (Ismotic) is unavailable. It is known that glycerol is transferred across the placenta in small amounts,108 but reports of adverse fetal effects cannot be found. Systemic complications of hyperosmotic agents are more likely to occur with intravenous use, and short-term use may be hazardous to the mother and/or fetus. Potential metabolic, renal, and cardiovascular abnormalities may result in direct insult to the fetus or compromise of the fetoplacental circulation. Hyperosmotic agents are thus relatively contraindicated in pregnancy, and any seemingly unavoidable use should be preceded by extensive obstetrics consultation and patient counseling of risks.
What Other Glaucoma Treatment Options Could be Offered to Childbearing Women?
Nonmedical therapies for glaucoma should be considered in women of reproductive age as alternatives to the potential risks associated with glaucoma agents during pregnancy and nursing. The prospect of surgical management can be considered, especially in younger women who are dependent on oral carbonic anhydrase inhibitors for acceptable IOP control. Ideally, such intervention should occur well prior to the onset of pregnancy,39 and while the visual prognosis is still good. When medical control becomes inadequate, the ophthalmologist should avoid procrastination of surgery. Delay of glaucoma management due to unexpected pregnancy could perhaps result in further field reduction. It has been suggested that cases of late-stage glaucoma and advanced fixation that threaten visual field loss should receive aggressive management in the antenatal period due to the risk of progression precipitated by the bearing down effect during labor and delivery.109 In such situations, vaginal operative delivery or cesarean section could be considered in an effort to avoid further late-stage visual field loss.
Laser trabeculoplasty can be an excellent alternative to medical therapy in childbearing women, and the procedure may be safely performed during pregnancy or the postpartem period. Further, the option of argon laser trabeculoplasty (ALT) could be considered early in the management of POAG for women during the reproductive years, with the goal of decreasing or eliminating dependency on medications. Several reports have also described favorable results in support of early filtration surgery in the general population of patients with open-angle glaucoma.110–113 Young women with glaucoma may also benefit from early surgery, and the issue should be discussed if a patient is planning pregnancy. Intraocular surgery, however, should generally be avoided when possible during an existing pregnancy, because of anesthetic risks and potential difficulties in managing complications. The hazards of antimetabolite use as adjunctive treatment in filtering surgery during pregnancy are also unknown, although fluorouracil and mitomycin C have known teratogenic potential.5 Trabeculectomy and other forms of filtration surgery, if indicated, usually can be deferred until after delivery, unless, in the practitioner’s judgment, severe maternal vision loss is imminent despite maximally tolerated medical therapy. In the rare event of unavoidable surgery, it is this author’s recommendation that antimetabolites should not be used during pregnancy and lactation unless further data supporting such use become available (Fig. 21–1).
Future Considerations
Pregnancy after the age of 35 is becoming increasingly common in our society, often because of career issues. Successful pregnancies in cases of advanced maternal age have been enabled by improved fertility and obstetrical care, but the coincidence of glaucoma and pregnancy may be higher during later childbearing years. It is hoped that further research will eventually refine knowledge of glaucoma management in the subpopulation of women during reproductive age and pregnancy More epidemiologic information would be useful in assessing risks of glaucoma in pregnancy, particularly in identifying those risks of medical treatment. Practitioners have a responsibility for reporting adverse drug reactions, and cumulative case experiences may be helpful in modifying recommendations concerning indicated drug use during pregnancy. The issues of human teratogenicity and maternal toxicity are complex and difficult to address experimentally, and treatment of glaucoma in pregnancy continues to be challenging due to poorly defined risks. However, this uncommon clinical dilemma appears to attract little attention among glaucoma investigators, and it is anticipated that progress in this area will remain slow.
Figure 21–1. Suggested algorithm for management of POAG in pregnancy.
References
6. Kooner KS: Personal communication/unpublished observations.
16. Sunness JS: The pregnant woman’s eye. Surv Ophthalmol 1988;32:219–238.
18. Finlay CE: Bitemporal contraction of visual fields in pregnancy. Arch Ophthalmol 1923;52:50–55.
20. Kass MA, Sears ML: Hormonal regulation of intraocular pressure. Surv Ophthalmol 1977;22:153–176.
23. Wilke K: Episcleral venous pressure and pregnancy. Acta Ophthalmol 1975;125:40–45.
30. Brent RL: Editorial comment on “Teratogen update: Bendectin.” Teratology 1985;31:429–431.
32. Shepard TH: Human teratogenicity. Adv Pediatr 1986;33:225–268.
34. Flach AJ: Glaucoma treatment and pregnancy (correspondence). Arch Ophthalmol 1991;109:463.
35. Mitchell AA: Drugs and birth defects. Clin Toxicol Rev 1982;4(10):1–2.
36. Miller RK: Drugs during pregnancy: a therapeutic dilemma. Rational Drug Ther 1981;15(7):1–9.
39. Spaeth GL: Glaucoma treatment and pregnancy. Arch Ophthalmol 1990;108:1536.
45. Schocken DD: Adrenergic receptors of the placenta. Trends Pharmacol Sci 1982;3:215–217.
50. Blaul G: [Local beta blockaders in pregnancy]. Klin Monatsbl Augenheilk 1985;187:57–59.
51. Fishman WH, Chesner M: Beta-adrenergic blockers in pregnancy. Am Heart J 1988;115:147.
56. Woods DL, Morrell DF: Atenolol: side effects in a newborn infant. Br Med J 1982;285:691–692.
61. Williams T, Ginther WH: Hazard of ophthalmic timolol. N Engl J Med 1982;306:1485–1486.
66. Sastry BV: Human placental cholinergic system. Biochem Pharmacol 1997;53(11):1577–1586.
75. Product Information. Propine. Allergan, 1999. Irvine, CA.
76. Product Information. Alphagan. Allergan, 1999. Irvine, CA.
79. Product Information. Iopidine. Alcon Laboratories, 1999. Fort Worth, TX.
86. McBride WG: The teratogenic action of drugs. Med J Aust 1963;2:689–693.
90. Crane CH: Effect on fetus of mother taking a diuretic. JAMA 1957;165:1517.
92. Product Information. Azopt. Alcon Laboratories, 1999. Fort Worth, TX.
93. Product Information. Trusopt. Merck, 1999. West Point, PA.
95. Product Information. Xalatan. Pharmacia & Upjohn, 1999. Kalamazoo, ML
104. Craft IL, Musa BD: Hypertonic solutions to induce abortion. Br Med J 1971;2(752):49.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree