Chapter 28 Management of Bell’s Palsy and Ramsay Hunt Syndrome
BELL’S PALSY
Bell (1774-1842)1 first described a patient with a facial paralysis in 1818; subsequently, all patients with facial palsy of unknown etiology have come to bear his name. The etiology of this “idiopathic” disorder has become much clearer in recent years. Although first proposed in 1972 by McCormick,2 herpes simplex virus (HSV) has been identified as the disease vector only recently,3–5 and an animal model has been designed.6 Murakami and associates5 identified HSV type 1 (HSV-1) DNA fragments in perineural fluid in 11 of 14 patients undergoing facial nerve decompression. In this study, no control subjects had HSV-1 DNA in perineural fluid. Using polymerase chain reaction to analyze the saliva of patients with Bell’s palsy, Furuta and colleagues3 identified HSV-1 DNA in 50% of patients, which was significantly more often than controls, a finding confirmed by other groups.7 Polymerase chain reaction has also been used to isolate HSV-1 genomic DNA from the geniculate ganglion of a temporal bone in a patient dying during the acute phase of Bell’s palsy.4
Sugita and coworkers6 proposed an animal model of Bell’s palsy. Six days after inoculation of HSV-1 into either the auricle or the tongue of mice, a temporary ipsilateral facial paralysis was identified that recovered spontaneously within 3 to 7 days. Histopathologically, neural edema, vacuolar degeneration, and inflammatory cell infiltrate with associated demyelination or axonal degeneration were shown in the affected facial nerve and nucleus.8 HSV-1 antigens were identified within the facial nerve, geniculate ganglion, and facial nucleus 6 to 20 days after inoculation.6 Similar pathologic findings have been shown in rabbits after HSV-1 inoculation, but without the associated facial paralysis.9
The histopathologic changes seen in autopsy specimens of patients who died with acute idiopathic facial paralysis have provided some insight into the underlying cellular mechanisms in Bell’s palsy. Reddy and coworkers10 found degeneration of the myelin sheath and axons, perivascular inflammation, and a phagocytic cell infiltrate in 10% to 30% of nerve fibers in a patient 17 days after the onset of an acute idiopathic facial paralysis. Fowler11 found diffuse vascular engorgement throughout the intratemporal facial nerve and evidence of acute hemorrhage within the intracanalicular, labyrinthine, and geniculate portions of the facial nerve in a patient who died shortly after the onset of Bell’s palsy. In examining an autopsy specimen of a patient who died 13 days after the onset of Bell’s palsy, McKeever and colleagues12 found a diffuse lymphocytic infiltration of the intratemporal facial nerve with ongoing myelin phagocytosis. They later re-examined the same specimen and noted the most pronounced lymphocytic infiltration of the nerve at the labyrinthine segment, findings that these authors believed were most consistent with an ongoing compression-type injury to the facial nerve.
Multiple reports have found that there seems to be evidence of constriction of the nerve at the meatal foramen in cases of Bell’s palsy. In specimens examined during the acute phase of Bell’s palsy, lymphocytic infiltration, perineural edema, and myelin degeneration have been noted.13,14 In a nerve specimen examined 1 year after the diagnosis of Bell’s Palsy lymphocytic infiltration, perineural edema, and fibrotic changes were noted.15 Intraoperative biopsy specimens of the greater superficial petrosal nerve from patients undergoing nerve decompression procedures for Bell’s palsy have shown axonal demyelination and degeneration with lymphocytic infiltration.16 Together, the histopathologic findings in Bell’s palsy show demyelination and axonal loss with lymphocytic or phagocytic infiltration, findings that seem to be more pronounced at the labyrinthine segment of the facial nerve.
In considering this evidence, the pathogenesis of Bell’s palsy becomes more apparent: a virally induced, inflammatory response that produces edema within the nerve. Fisch and Felix16 first proposed that the facial nerve was entrapped at the meatal foramen as a result of neural edema. Intraoperative conduction studies have shown an electrophysiologic blockage at this site.17 The constriction imposed produces a conduction block at first; however, with prolonged or increased constriction, ischemia results. Subsequently, wallerian degeneration occurs, producing axonotmesis or neurotmesis or both. A spectrum of injury within the nerve from neurapraxia to neurotmesis may occur in Bell’s palsy.11,18 The proportion of each of these determines the amount of facial function that returns when the acute phase of the disease subsides.
Bell’s palsy accounts for nearly three quarters of all acute facial palsies.19 The incidence of Bell’s palsy is 20 to 30 cases per 100,000 per year.20 The median age is 40 years, but it can occur at any age.21 The incidence is highest in patients older than 70 years and lowest in children younger than 10 years. The left and right sides are affected equally. Men and women are equally affected, but the incidence of Bell’s palsy is higher in pregnant women (45 cases per 100,000).
The clinical presentation of Bell’s palsy is well known; however, the clinician must exclude other causes of facial paralysis based on history and physical examination findings. Patients describe an abrupt onset of unilateral paresis that occurs over 24 to 48 hours. The paresis can progress over 1 to 7 days to complete paralysis. Bilateral involvement, either simultaneously or consecutively, has been described.23 A history of progression of weakness over weeks to months, repeated episodes of paralysis, and twitching of the facial muscles should not be considered symptoms of Bell’s palsy. Other associated symptoms of hearing loss, vestibular symptoms, or other cranial nerve neuropathies also rule out the diagnosis of Bell’s palsy.
Audiometric evaluation should reveal symmetric function except for an absent ipsilateral acoustic reflex. If unilateral hearing loss or acoustic reflex decay is present, further evaluation for retrocochlear pathology is necessary. If vestibular complaints are present, an electronystagmogram is performed, and diagnosis of Bell’s palsy should be questioned. If the history and clinical presentation are highly suggestive of Bell’s palsy, magnetic resonance imaging (MRI) and computed tomography (CT) are not performed. High-resolution CT scans and MRI are obtained if patients have associated symptoms of otorrhea, vestibular complaints, and hearing loss. Planned surgical decompression and persistent dense paralysis after 6 months are also indications for imaging. Serologic tests for Lyme disease (IgG, IgM) are an important part of the work-up for unexplained facial paralysis in endemic areas.24 Serologic testing for antibodies to HSV or varicella zoster have provided some correlative evidence for a viral etiology in Bell’s palsy, although laboratory investigations in general have not been found to provide clinically relevant data.
Electrodiagnostic testing is an important element of the diagnostic evaluation of facial paralysis. Testing can determine the extent of facial nerve injury and provide useful prognostic information for the development of management strategies. The technique of electroneuronography (ENoG), developed by Esslen,25 can distinguish the nerve fibers that have undergone wallerian degeneration from fibers that are temporarily blocked (neurapraxia). ENoG testing is not performed until 3 to 4 days after the development of complete unilateral paralysis because wallerian degeneration does not become apparent until 48 to 72 hours after an acute injury to the nerve. Electrodiagnostic testing is not performed when the patient exhibits paresis only. If the paresis progresses to total paralysis, electrodiagnostic testing is performed 3 days after the onset of total paralysis. Presence of voluntary movement 4 to 5 days after the onset of paresis indicates only minor injury, and complete recovery should be anticipated.
ENoG is most accurate when it is performed within 3 weeks of the acute injury. The test is performed using standard electromyography (EMG) equipment, but requires the use of special surface stimulating and recording electrodes.26 The recording electrodes are in a hand-held carrier and are manipulated in the nasolabial fold with the Esslen technique. The recording electrodes are not taped to the skin, as has been described by others.27
An evoked electric stimulus generates synchronous facial muscle movement that can be recorded from the skin surface (termed the compound muscle action potential [CMAP]). The amplitude of the biphasic CMAP has been found to correlate with the number of blocked or neurapraxic nerve fibers. As the percentage of degenerated fibers within the nerve increases, the amplitude of the CMAP decreases compared with the normal side of the face. Fisch and Esslen28 determined that if 90% or more of the fibers within the facial nerve degenerate within the first 14 days of an acute paralysis, a severe injury has occurred, and the chances of complete recovery are less than 50%. Patients who do not reach the 90% degeneration level by 3 weeks have a very good prognosis and are likely to regain normal facial motion without synkinesis. The time course of degeneration is also important; the more rapid the degeneration, the more severe the injury.29 A patient showing greater than 90% degeneration at 5 days would have a worse prognosis than a patient with 90% degeneration at 14 days.
Topognostic testing is widely reported to be useful in determining the site of injury in acute facial paralysis; however, intraoperative studies have shown that the Schirmer test is not accurate in diagnosing Bell’s palsy.26 The Schirmer test may be used to determine the extent of lacrimation and the need for eye protection.
A review of the natural history of Bell’s palsy shows that approximately 85% of patients begin to display some return of facial movement within 3 weeks of the onset of paresis.30 The remaining 15% begin to improve 3 to 6 months after the onset of the disease. Most patients show a complete return of facial function, but 10% to 15% have residual unilateral weakness and develop secondary deformities of synkinesis, tearing, or contracture. Some motion returns in almost all individuals with Bell’s palsy by 6 months. If no movement returns, a vigorous search for another etiology should begin anew.
The management of patients with Bell’s palsy varies, depending on the type of specialist initially seeing the patient and on the training of the individual specialist. Figure 28-1 presents an overview of our management strategy. Patients presenting within the first week of facial weakness with paresis are placed on steroid therapy (prednisone, 60 to 80 mg/day for 7 days) and an antiviral agent (valacyclovir, 500 mg three times per day for 10 days). If patients are seen 7 to 10 days after onset and motor function is stable or improving, medical treatment is unnecessary. Patients are instructed to return in 1 week for re-evaluation to determine if neural degeneration has occurred. Electrodiagnostic testing is unnecessary as long as voluntary facial movement is present. If total paralysis occurs in the interim, the total paralysis protocol of Figure 28-1 is followed. Stable or improving patients are seen in 1 month.
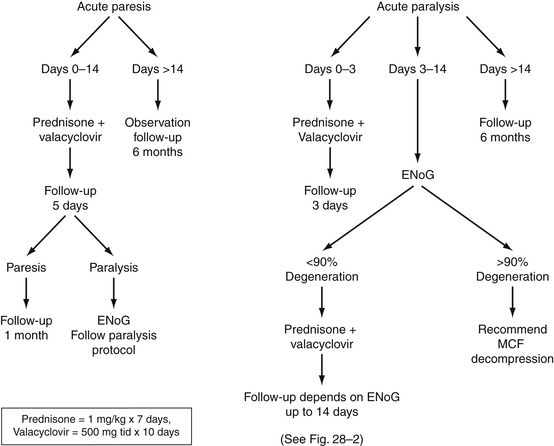
FIGURE 28-1 Algorithm for management of Bell’s palsy. ENoG, electroneuronography; MCF, middle cranial fossa.
The use of steroids in Bell’s palsy has been the subject of much debate. Numerous studies of varying designs in adults have shown better outcomes in patients treated with steroids,21,31,32 especially when initiated early in the course of the disease.33–35 Other randomized studies36 and meta-analyses,37 including many studies in children,38–40 have concluded that steroids did not affect the ultimate facial function outcomes in Bell’s palsy. Grogan and Gronseth,41 in a comprehensive, evidence-based review, concluded that there seemed to be a beneficial effect in the use of steroids in Bell’s palsy. Ramsey and coworkers42 performed a meta-analysis of 47 trials of steroid therapy for Bell’s palsy, and concluded that there seemed to be an improved odds of recovery in patients treated with steroids (49% to 97%) compared with untreated controls (23% to 64%). We use prednisone in a dose of 1 mg/kg daily for 7 days in all cases in which it is not medically contraindicated, in anticipation of speeding recovery, reducing the number of degenerating axons, and reducing the number of patients needing decompression.
The combination of steroids and antivirals may be superior to either one alone. A double-blind, randomized, controlled trial of acyclovir and prednisone versus prednisone alone in the treatment of Bell’s palsy showed better results with the combination therapy.43 This study documented poor facial function recovery in 23% of the prednisone-only group compared with 7% in the acyclovir-plus-prednisone group. Interpreting the results of this study, Grogan and Gronseth41 reported that patients who receive antiviral therapy in addition to steroids were 1.22 times as likely to have a good facial nerve outcome. A multicenter, randomized, placebo-controlled study comparing treatment of patients with Bell’s palsy with steroids and antivirals with treatment with steroids alone concluded that the addition of valacyclovir improved the recovery rate from 75% to 90% in cases of complete palsy, and from 89% to 96% in all cases of facial palsy.44 Many other reviews of the topic have made similar conclusions.45–48
Other studies have found no significant difference between this combination of drugs and the natural history of the disease.24,49
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


