Management of Astigmatism in Conjunction with Lens Surgery
Louis D. Nichamin
When assessing recent changes in modern cataract and intraocular implant (IOL) surgery, arguably, the single most pressing challenge facing today’s phacoemulsification (phaco) surgeon is the need to achieve predictable and accurate refractive outcomes. Surgeons and patients alike have come to largely measure the success of their surgery by the refractive outcome, and one of the leading causes for litigation in this field is the “refractive surprise.”1 In addition, refractive lens exchange has become an important component of the refractive surgeon’s armamentarium.2 As such, the fields of cataract and refractive surgery have merged to form an amalgam without distinct borders. Improved refractive results have come about by way of improvements in both surgical technique as well as advances in technology. Spherical results, for example, have become more predictable because of increased attention to biometry technique, as well as breakthrough technology such as partial coherence interferometry.3
No less important is the astigmatic component of the refractive equation. At one time during the evolution of small-incision surgery, it was the surgeon’s goal to not induce astigmatism.4 Today, to fully embrace the concept of “refractive cataract surgery,” one must be able to address and reduce significant pre-existing cylinder.
PATIENT SELECTION AND CONSIDERATIONS
Estimates of the incidence of significant, naturally occurring astigmatism vary widely from 7.5% to 75%.5 It has been reported that 3% to 15% of eyes may have 2 or more diopters (D) of astigmatism.6 In our clinical experience, 10% of patients presenting for surgery have greater than 2.0 diopters, 20% have between 1 and 2 diopters, and 70% have less than one diopter of pre-existing cylinder. In light of recent experience gained in the field of refractive surgery, many surgeons would agree that astigmatism of greater than 0.5 D will lead to symptoms of ghosting and shadows. Although the older cataract patient may be more tolerant of cylinder, the ambitious refractive cataract surgeon should likely approach an implant patient with the same high goals that he or she might with a younger keratorefractive candidate. Indeed, successful cataract practices are now aiming for both spherical and astigmatic outcomes of ±0.5 D.7
When considering astigmatism correction, one must take into account the location of the cylinder, age of the patient, and status of the fellow eye. Since most patients will drift against-the-rule (ATR) over their lifetime for example, toward plus cylinder at 180 degrees some surgeons advocate a less aggressive approach to the reduction of with-the-rule (WTR) cylinder. Authors have also suggested that residual WTR astigmatism may favor better uncorrected distance acuity given that most visual stimuli are of a vertical nature.8 Similarly, it has been contended that ATR cylinder may improve uncorrected near vision.9 The tenet that residual (myopic) WTR astigmatism is a desirable goal to enlarge the conoid of Sturm and hence optimize depth perception has, however, recently been called into question.10 Currently, with recent refinements in surgical technique, a spherical goal may be most desirable for most patients undergoing implant surgery.
TREATMENT OPTIONS
The first decision faced by the surgeon is whether to address pre-existing astigmatism at the time of cataract and IOL surgery, or to defer and treat the cylinder separately. One could reasonably argue that for optimal accuracy, sufficient time for wound healing should be allotted and a stable refraction ought to be documented prior to astigmatic correction. This consideration had been more germane with the use of rigid implants and larger incisions. Currently, most surgeons are using foldable IOLs and studies have well documented the nearly neutral astigmatic effect that these incisions bear when kept at or near 3.0 mm, as well as their early refractive stability.11,12,13 As such, many surgeons feel that concomitant treatment of pre-existing astigmatism is a more efficient approach and is favored since it will likely save the patient from having to undergo a second procedure.
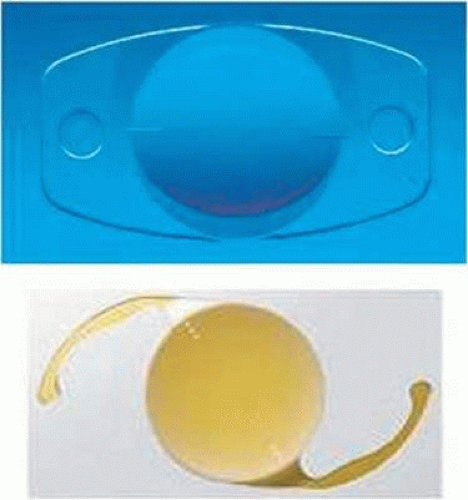
The next major decision is whether to treat the astigmatism through a lenticular approach, that is, to use a toric IOL, or to use a keratorefractive technique. From a theoretical perspective, it is hard to argue against the use of a toric implant, and their effectiveness has been widely reported.14 This option has the potential to avoid induced irregular astigmatism from corneal manipulation and provides the option of reversibility. In the United States, the first toric IOL to have received FDA approval was the single-piece plate haptic design manufactured by Staar Surgical (Fig. 12-1A). This silicone implant is available in two toric powers of 2.0 and 3.5 D. Propitious outcomes have been obtained with this device even with minimal experience by community-based surgeons.15 For surgeons using this particular toric implant, lens rotation is a recognized problem; Sun et al14 reported a need to return to the operating room for repositioning in 9.2% of cases. Ruhswurm et al16 further reported axis rotation of at least 25 degrees in 18.9% in their series. According to Euclidean geometry, an axis deviation of 5, 10, or 15 degrees will result in 17%, 33%, and 50% reduction, respectively, in effect.5 Optimal timing of the IOL repositioning would appear to be between 1 and 2 weeks following implantation as capsular fibrosis is underway, and may serve to permanently fixate the toric device in the proper meridian. Some surgeons have avoided this particular implant because of its plate haptic design and for the first-generation silicone elastomer from which it is composed. More recently, the FDA has approved the single-piece AcrySof toric IOL (Fig. 12-1B). This hydrophobic acrylic device is available in three toric powers of 1.5, 2.25, and 3.0 diopters, which are capable of correcting 1.03, 1.55, and 2.06 diopters, respectively, at the corneal plane. These newer designs offer better rotational stability and explain, in large part, their increased use and popularity.17,18,19,20 The economic value of reducing spectacle dependence through the use of a toric IOL, as compared to a standard monofocal implant, has been demonstrated by a short- and long-term analytic model.21
The notion of reducing astigmatism at the time of cataract surgery by way of adjunctive keratorefractive surgery, specifically astigmatic keratotomy, was first proposed by Robert Osher and dates back to the mid-1980s.22,23,24 Our personal experience with this technique began in 1988 and was based on the principles of astigmatic keratotomy as taught by Dr. Spencer Thornton.25 A transition to (intra)limbal relaxing incisions (LRIs) took place in 1994, spurred by the experience of Dr. Dave Dillman and his improved results using the peripheral limbal technique of Dr. Stephen Hollis, an early advocate and innovator in the use of LRIs. With refinement of the Hollis nomogram, we found this more peripheral approach to astigmatic keratotomy to be considerably more forgiving than conventional astigmatic keratotomy performed at a smaller (i.e.,7.0 mm) optical zone.26,27,28
Another viable option to decrease astigmatism is to manipulate the cataract incision by first placing it on the steep corneal meridian, and then by varying its size and design, affect a desired amount of wound flattening, and hence a decrease in cylinder.29 Specifically, one can increase or decrease the amount of wound flattening by increasing or decreasing the size of the incision. Similarly, wound flattening may be enhanced by moving closer to the visual axis, or by creating a more circum-parallel incision to the limbus. Also, a perpendicular component, or groove, may be added to the incision to further increase wound flattening and “against-the-wound” astigmatic drift.25 This approach, however, presents logistic challenges including movement around the surgical table, often producing awkward hand positions. In addition, varying surgical instrumentation may be needed along with a dynamic mindset, and this approach is effective only for relatively small degrees of pre-existing cylinder. For these reasons, this technique has largely been supplanted by the use of a consistent and essentially neutral phaco incision, typically located temporally for astigmatic stability, and then adding supplemental relaxing incisions (LRIs). A recent study by Kaufmann et al30 concluded that LRIs in combination with a temporal clear corneal incision provided superior astigmatic outcomes to that of on-axis surgery.
Several other options to reduce astigmatism deserve mention. Lever and Dahan31 have suggested the novel use of opposing clear corneal incisions to address pre-existing cylinder. In this technique, a second opposite penetrating clear corneal incision is placed over the steep meridian 180 degrees away from the main incision. This approach is technically simple and requires no additional instrumentation; however, a second substantial penetrating incision is now present, possibly increasing the risk of wound leak or even infection. In addition, single-plane beveled incisions are known not to be as effective, for a given arc length, at flattening the cornea as are more conventional perpendicular relaxing incisions.25,32
Yet another important and increasingly popular alternative is that of bioptics, a laser technique originally described to address residual refractive error following implantation of myopic phakic IOLs, but one that is just as useful in the setting of pseudophakic lens surgery.33,34,35 In this approach, one exploits the advanced technology and exquisite accuracy of the excimer laser. In a staged manner, one may treat both residual spherical as well as astigmatic error following implant surgery. In Zaldivar’s original description, a LASIK flap was created prior to the phakic implant procedure, and then as necessary, the flap was lifted and residual refractive error was corrected with the laser. Today, most surgeons prefer to perform both the flap and laser ablation concurrently following cataract surgery, as needed, thus reducing the number of unnecessary flaps that would otherwise be created. It has been our experience that LASIK may be performed safely following IOL surgery at 6 weeks, perhaps earlier. Of course, wound stability and healing must be confirmed, along with a stable refractive error. It might further be argued that photorefractive keratectomy (PRK) is particularly well suited for this older pseudophakic population that typically has only modest amounts of residual refractive error, and within whom a higher incidence of dry eye and corneal surface problems exist. Custom wavefront-guided ablation may also be considered since the pseudophakic eye no longer has a dynamic lens component, though obtaining a reliable wavefront aberrometry reading may be difficult with some lens designs, i.e., refractive multifocal optics.36 For most refractive cataract surgeons, bioptics has become an integral part of the preoperative discussion with the patient, and in our experience its use is required in approximately 10% of cases, depending on the magnitude of the patient’s preop refractive error. Although of limited popularity, conductive keratoplasty used in an off-label fashion has also been described as a means by which residual hyperopia and hyperopic astigmatism may be effectively reduced following cataract surgery.37
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree