The incidence of cutaneous malignant melanoma (CMM) continues to increase at a remarkable rate, and mortality from this disease also continues to rise. Despite our best attempts to understand the molecular basis of CMM, only recently have some of the critical molecular determinants of this disease been uncovered and used as a basis for new medical treatments. Thus, successful treatment of this disease relies heavily upon the surgeon. In this chapter, we will review some of the recent developments in the staging, evaluation, and treatment of CMM and present guidelines for treatment and management of melanoma of the head and neck.
EPIDEMIOLOGY
Despite an overall modest decrease in cancer incidence and cancer-related deaths over the past 20 years (largely related to lung, colon, breast, and prostate cancers), melanoma incidence rates and deaths continue to rise yearly. In the United States alone for 2013, it is estimated that 76,690 new melanoma cases will be diagnosed, and 9,480 deaths will be attributed to melanoma (
1). While melanoma accounts for roughly 4% of all skin cancers, it is responsible for more than 77% of skin cancer deaths.
The major risk factors for development of cutaneous melanoma include fair complexion, tendency to sunburn easily, large congenital nevi (greater than 20 cm), presence of atypical/dysplastic nevi (
2,
3), a personal history of skin cancer (melanoma or otherwise) (
4,
5,
6,
7), genetic predisposition (
8), and immunosuppression (
9). Head and neck melanomas occur more commonly in men (2:1), with a median age at diagnosis of 55 years, and a range of 12 to 92 years (
10).
Epidemiologic studies demonstrate higher rates of melanoma in those living in geographic areas that are exposed to intense sunlight (i.e., Australia). This finding and investigations in the laboratory demonstrate that exposure to ultraviolet light plays a major role in the pathogenesis of melanoma, and UV-A and UV-B radiation have both been implicated. This is further supported by the lower rates of melanoma reported in people whose skin natively has more pigmentation, and the incidence of melanoma is approximately 10 times higher in whites than in blacks in the same geographic region (
11).
Although the combined head and neck region accounts for only 9% of body surface area, 15% to 30% of all melanomas arise within the head and neck (
12,
13). Multiple factors may contribute to this anatomic predilection for the skin of the head and neck, including higher levels of sun exposure and melanocyte content of the skin that is two- to threefold higher than other regions (
14). Of note, melanomas arising on the face and scalp are associated with higher rates of local recurrence and regional lymph node disease than melanomas arising in other parts of the body.
MELANOMA SUBTYPES
Melanoma is divided into four distinct clinicopathologic subtypes: lentigo maligna melanoma (LMM), superficial spreading, nodular, and acral lentiginous melanoma. These lesions demonstrate either a radial (intraepithelial) or vertical (intradermal) growth phase or a combination of the two. Radial growth is circumferential in nature and confined to the dermal-epidermal junction. The vertical growth phase (intradermal) demonstrates invasion through the dermal-epidermal junction. The radial growth phase may indicate a lesion’s capacity for growth and invasion into the papillary dermis. These lesions may lack metastatic potential. Conversely, cells in the vertical growth phase may represent a clonal change in cells with a growth advantage over neighboring cells, resulting in the clone’s ability to invade and metastasize.
Cutaneous melanomas may mimic a host of other pathologic cutaneous lesions.
Differential diagnosis includes seborrheic keratosis, benign nevi (including junctional, compound, and dermal), hemangioma, blue nevi, pyogenic granuloma, and pigmented basal cell carcinoma.
Lentigo maligna (LM), also known as melanoma
in situ, is a premalignant pigmented lesion that frequently develops in the head and neck region of elderly patients (
Fig. 114.1A). Known historically as Hutchinson melanotic freckle, these lesions are associated with solar skin damage and feature atypical melanocytes, which spread radially along the dermal-epidermal junction, exhibit focal nesting, and occasionally extend along skin appendages into the dermis. Up to 5% of LMs or melanomas in situ progress to invasive LMM. LMM lesions should be excised with 0.5- to 1.0-cm margins, although prediction of tumor margins can prove difficult. This is the least common type of cutaneous melanoma and accounts for between 6% and 10% of melanoma lesions. Its growth is characterized by a slow radial phase that may take up to 10 years to progress. At first, they are quite slow to invade deeply, and affected patients have a better prognosis when compared with those with other forms of melanoma.
Superficial spreading melanoma represents the most common type of melanoma and comprises between 65% and 75% of cases (
Fig. 114.1B). These lesions may demonstrate a wide variety of colors, including pink, blue-gray, brown, tan, and black. They also may demonstrate radial growth for 5 to 7 years and then become invasive, an event frequently heralded by ulceration and bleeding. High cure rates have been reported when these lesions are clinically detected in the radial growth phase. Spontaneous regression of superficial spreading melanomas also has been reported.
Acral lentiginous is the most common type of melanoma seen in the African American population. Because these lesions commonly occur on the soles of the feet, surfaces of the hand, and oral/anogenital mucosa, head and neck surgeons less commonly encounter them.
Nodular melanoma comprises between 10% and 15% of all melanomas and may affect areas of both exposed and non-sun-exposed areas of the skin (
Fig. 114.1C). They tend to develop in patients older than 50 years. Nodular melanoma is considered to be the most invasive of the cutaneous melanomas, and affected patients have the poorest prognosis.
Desmoplastic melanoma is a histologic variant of melanoma that accounts for fewer than 1% of melanoma cases overall (
Fig. 114.1D). However, as many as 75% of these tumors occur in the head and neck region. Clinically, desmoplastic melanomas may be amelanotic, a feature that impedes early recognition and often leads to significant delays in diagnosis and treatment. Another important characteristic feature of these lesions is their neurotropism.
This predisposes them to perineural invasion and spread that often accounts for local recurrence despite histologically “negative margins” with recurrence rates reported between 23% and 48% (
17,
18,
19,
20,
21,
22). Therefore, it is suggested that wider resection margins be taken around a desmoplastic tumor and that adjuvant radiation is often recommended after definitive excision. Multiple studies have demonstrated a lower rate of regional nodal spread in patients with desmoplastic melanoma as compared to other histologic subtypes of melanoma (
23,
24,
25,
26,
27). The lower rate of positive sentinel lymph nodes has made the role of SLNB in this population less widely recommended (
25). However, a distinction needs to be made between those lesions which are purely desmoplastic and are associated with a low rate of nodal metastasis and those that have a combination of desmoplastic and a more conventional melanoma histology which spread to lymph nodes at the higher frequency associated with the nondesmoplastic histology (
25,
26). Therefore, SLNB can be recommended in the combined desmoplastic cases, which meet other criteria for performing the biopsy such as depth of invasion, number of mitoses, and/or ulceration.
MUCOSAL MELANOMA
A relatively rare entity, mucosal melanoma deserves special mention. Primary melanoma may arise in the mucosa of the respiratory, alimentary, and genitourinary tracts, as they all contain melanocytes. Mucosal melanoma accounts for 1% to 2% of all melanomas, with the majority being found in the head and neck region (
28). More than 50% of reported cases arise within the nasal cavity. Other affected areas include the paranasal sinuses, nasopharynx, oral cavity, and oropharynx. Esophageal mucosa can also be affected. Nasal obstruction is the most common presenting symptom for sinonasal lesions. However, a significant number of mucosal melanomas may be asymptomatic until they have progressed to an advanced stage, which contributes to the poor prognosis of patients with this diagnosis. Stern and colleagues (
29) at the M.D. Anderson Cancer Center found an average of 9 months between the first onset of symptoms and physician intervention. In addition to delay in diagnosis, the poorer prognosis associated with mucosal melanomas may also be related to an inherently more aggressive behavior of mucosal melanomas, or earlier dissemination, because of the rich lymphatic and vascular supply of the mucosa.
The differential diagnosis includes vascular lesions, angiomas, and dental amalgam tattoos. It is not uncommon for the characteristic melanosis to be absent in mucosal melanoma lesions. Biopsy is usually confirmatory, and immunohistochemistry may be used to assist in diagnosis. Desmoplastic variants of mucosal melanoma have been described in the head and neck (
30). No studies have conclusively demonstrated that depth of invasion is of prognostic importance in mucosal melanoma.
The majority of these patients have a poor prognosis. These tumors have a high rate of local recurrence, which is typically a harbinger of disseminated disease, and most of these patients die from complications related to distant metastases.
Anatomic constraints frequently preclude the use of excisional biopsy techniques in most patients with mucosal melanoma. Although wide local excision is the mainstay of therapy, radiation therapy has been used as a primary modality and postoperative adjuvant therapy with unclear benefit (
31,
32,
33). It should especially be considered in situations when adequate resection margin is not possible, where it may help prevent local recurrence (
34).
Patients may experience multiple local recurrences and ultimately die of a combination of uncontrolled local and distant disease. Five-year survival rates for mucosal melanoma range between 10% and 45% (
35,
36,
37,
38,
39).
MELANOMA STAGING SYSTEM
The staging of malignant melanoma has evolved over the past half century in efforts to more reliably determine prognosis and to identify the most appropriate treatment schemes. Clark and Breslow made significant contributions to the microscopic grading of primary cutaneous melanoma. Clark (
40) described five levels of anatomic invasion through the layers of the skin. In the Breslow system (
41), the maximum thickness of melanomas is used as a prognostic indicator.
The current staging system for melanoma developed by the American Joint Committee on Cancer (AJCC) and adopted by the International Union for Cancer Control, relies upon assessment of the primary tumor (T), regional lymph nodes (N), and distant metastatic sites (M). The seventh, and most recent, edition of this staging system is based upon long-term analysis of over 38,900 patients with cutaneous melanoma (
42) (
Table 114.1).
Primary Tumor
Tumor thickness, presence or absence of ulceration, and mitotic rate are the histopathologic details factored into staging, as they have been demonstrated to correlate closely with prognosis. Clark level has clinical utility in the evaluation and treatment of patients with thin melanomas, and thus, is only included in the staging of melanomas less than or equal to 1 mm in thickness.
Regional Nodal Staging
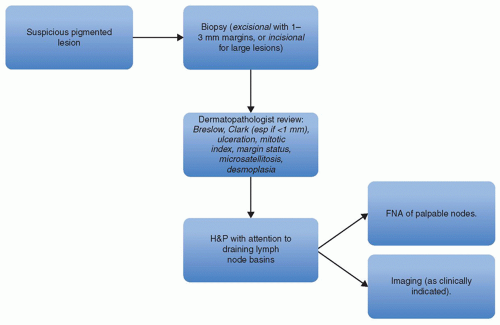
Regional nodal status is the most powerful predictor of survival for patients with melanoma (
43) and has led to the widespread adoption of SLNB in the evaluation of patients with primary cutaneous melanomas. For patients with clinically detectable lymph nodes on presentation, regional imaging (CT or MRI) as well as fine-needle aspiration
may be performed to confirm the presence of melanoma; if found, therapeutic lymph node dissection and systemic staging are recommended (
Fig. 114.2). The extent of dissection remains controversial and will be addressed later in this chapter.
The decision to perform SLNB is typically based upon characteristics of the primary tumor, as the risk of regional metastasis is known to correlate with tumor thickness and ulceration (
44,
45,
46,
47,
48); high mitotic rate and younger patient age have also been associated with SLN positivity rates (
49,
50,
51). With respect to SLN status, patients with T1 lesions have a 3% to 5% risk of SLN positivity; T2, 8% to 12%; T3, 23% to 27%; and T4, 24% to 44% (
46,
50,
52).
When reporting the pathologic nodal status after SLNB or a more comprehensive lymphadenectomy, it is important to include the number of nodes involved (
53), microversus macrometastases (
53), and the presence or absence of in-transit metastases. In-transit metastases are a unique manifestation of intralymphatic tumor dissemination, characterized by the presence of melanoma in either cutaneous or subcutaneous tissue situated between the primary tumor and the draining regional lymph node basin (
54). In early stage disease, these develop in 2% to 13% of patients (
55,
56), and based on poor patient outcomes associated with their presence, they are considered equivalent to the presence of significant nodal disease (
Table 114.1).
The prognosis for patients with microscopic metastases found at the time of SLNB (67% 5-year survival) is better than for patients with macroscopic metastases detected clinically (43% 5-year survival) (
57). For patients with microscopic metastases, the number of tumor containing nodes is the most important prognostic indicator followed by the mitotic rate (
57). For patients with macroscopic metastases, the number of nodes, primary ulceration, and patient age are the most important prognostic factors (
57). Buzaid et al. (
58) showed that the number of lymph nodes involved with regional metastases is more predictive of treatment outcomes than is the size of lymph nodes harboring metastatic disease.
In the head and neck, lateralized tumors may drain into the primary echelon nodal basins, including preauricular, parotid, postauricular, suboccipital, posterior cervical, anterior cervical (external or internal jugular), and supraclavicular nodal groups. Nodal drainage basins in the
head and neck are less predictable when compared with trunk and extremity sites. Although some areas on the scalp, face, and ears may have predictable lymphatic drainage, bilateral or contralateral metastases are not uncommon, and ambiguous drainage patterns can be seen in up to 55% of patients (
59). Preoperative and intraoperative lymphoscintigraphy helps to precisely define an individual’s pattern of lymphatic drainage in primary melanomas of the head and neck region (
60).
Metastatic Workup
The National Comprehensive Cancer Network (NCCN) guidelines for melanoma do not recommend routine use of imaging studies such as CT, MRI, or PET scans for the systemic staging of patients with thin melanomas (stage I and II) since the risk of distant metastasis is exceedingly low in this group of patients (
61). For patients with thin melanomas with adverse features such as positive deep margins, lymphovascular invasion, or mitotic rate greater than 1 per mm
2, imaging is recommended only to investigate specific signs or symptoms that may be attributable to a metastatic lesion.
For patients with stage III melanomas (metastatic disease in a lymph node, satellite, or in-transit metastases), the NCCN guidelines do not make specific recommendations and leave the workup to the discretion of the treating physician. Without any signs or symptoms, the likelihood of finding an occult metastatic lesion with screening CT or PET scan is low.
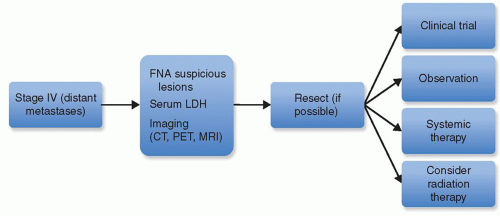
For stage IV (distant metastases), the NCCN guidelines recommend an LDH level plus chest x-ray and/or chest CT (
Fig. 114.3). A brain MRI or CT scan with contrast should be performed if the patients have even minimal suggestions of symptoms or physical findings of brain involvement or if the results would affect treatment decisions. LDH levels have been shown to correlate with melanoma-specific survival (
62). Nevertheless, in our practices, in order to help select and prioritize treatments for patients with Stage III or IV disease, we will often perform systemic imaging to include and brain MRI and either a PET/CT or CT of the head and neck, chest, abdomen, and pelvis. As an example, a patient presenting with regional nodal metastatic disease found to have distant metastatic disease on systemic imaging might be offered systemic treatment rather than surgery and radiation as initial treatment. Thus, we find imaging quite helpful in these instances to make informed treatment decisions.
DETECTION OF OCCULT LYMPH NODE DISEASE
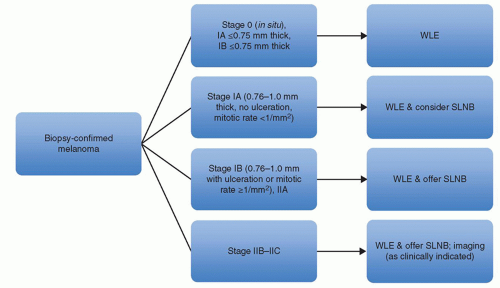
As stated previously, the most powerful predictor of survival is lymph node status. In patients with lesions less than 0.75 mm in depth, lymph node observation is appropriate. Patients with lesions between 0.76-1.0 mm in depth, without ulceration and less than 1 mitosis per mm
2 should consider lymph node evaluation. Patients with lesions between 0.76-1.0 mm in depth with ulceration or ≥1 mitosis per mm
2 or with lesions >1 mm with any characteristic should be offered lymph node evaluation. Of the two main ways of evaluating for occult lymph node metastases, SLNB has supplanted elective lymph node dissection (ELND) as the preferred method.
The rationale for determination of nodal status is to appropriately stage patients and to identify patients who may benefit from regional therapy with surgery, radiation, or both to improve locoregional control. The goal is to also identify patients at greater risk for systemic recurrence who could potentially benefit from systemic adjuvant therapy.
Elective Lymph Node Dissection
The utility of ELND in the initial management of patients with CMM of intermediate-thickness or thicker lesions is questionable in the current era of SLNB. In terms of using ELND to attempt surgical cure, four prospective randomized clinical trials failed to show a benefit from ELND in melanoma (
63,
64,
65,
66). However, data from the World Health Organization (WHO) Melanoma group Trial 14 and the Intergroup Melanoma Surgical Trial suggest that certain subgroups of patients may benefit from ELND (
44,
67,
68). In particular, patients in the WHO study with truncal melanoma greater than 1.5 mm thick and microscopic regional nodal disease had improved survival compared to those patients in whom lymph node dissection was delayed until they developed palpable lymphadenopathy (5-year survival 48.2% vs. 26.9%,
P = 0.04). In the intergroup study, patients younger than 60, with nonulcerated primary lesions 1 to 2 mm thick, had a survival benefit from ELND. While these studies demonstrate survival benefits in select groups of melanoma patients with clinically undetectable disease, the majority of patients (80% to 85%) will not have occult metastases.
The development of SLNB has enhanced the sensitivity and specificity of lymph node biopsy, and it has replaced ELND, a procedure with greater potential morbidity and disability, in the initial assessment of regional metastatic disease in patients with CMM of the head and neck region.
Sentinel Lymph Node Identification and Biopsy
Seminal work by Dr. Donald Morton (
14) and others has supported the hypothesis that, within each nodal basin, an orderly progression of lymphatic drainage is found from a first-echelon or sentinel node to nodes of lower echelons. This implies that if one can identify the sentinel lymph node in a basin at risk for spread from a melanoma and find it to be free of metastasis, then the remainder of nodes in that basin should also be free of metastatic tumor. This concept has been subsequently supported in a number of trials for non-head and neck CMM. Universal adoption of SLNB for CMM of the head and neck was somewhat delayed secondary to the belief that nodal drainage patterns in the head and neck region might be too complex to make SLNB feasible. Eicher et al. were among the first to dispel this notion (
69), demonstrating SLNs in 98% of patients in whom SLNB was performed in the context of a regional lymphadenectomy in patients with intermediate-thickness cutaneous melanomas in the head and neck region. No patients were found to have a positive non-SLN when the SLN was negative. Overall, studies reflect that the predictive value of a negative SLN approaches 99% and that the SLNB procedure has a low false-negative rate (less than 4%) (
70,
71); however, this rate may be slightly higher in head and neck melanomas as compared to the trunk or extremities (
72). Studies of SLNB for patients with head and neck primary melanomas report a range of regional recurrence from 2% to 8% in patients who had negative SLN (
67,
70,
72).
Patients with melanomas ≤0.75 mm in depth with or without adverse characteristics are not offered sentinel lymph node biopsy. Patients with melanomas 0.76-1.0 mm in depth without ulceration and <1 mitosis per mm
2 should consider SLNB. Patients with melanomas 0.76-1.0 mm in depth with ulceration or >1 mitosis per mm
2, or >1 mm in depth with any characteristic should be offered SLNB (
61) (
Fig. 114.4).