Chapter 120 Macular Translocation
Background and rationale
Preservation of the integrity of the foveal photoreceptors is vital to visual outcome in patients with macular disease, including diseases such as age-related macular degeneration (AMD), myopic degeneration, and pattern dystrophies. Many of these diseases initially affect subretinal layers, including the retinal pigment epithelium (RPE), Bruch’s membrane, and the choriocapillaris. These layers provide critical support for overlying outer retina, including the foveal photoreceptors.1 Ultimately, disruption of these tissues results in structural and functional deterioration of the photoreceptors with subsequent vision loss. Macular translocation surgery moves the fovea from over a severely diseased subretinal bed to a new location with healthier subretinal tissues to allow for improved function and ideally restoration of functional central vision.
Animal studies
Machemer and Steinhorst utilized a rabbit model to demonstrate the feasibility of purposeful retinal detachment with subretinal infusion via a transscleral approach.2 Electron microscopy confirmed preservation of photoreceptor nuclei and mitochondria in the inner segments with some disruption of the outer segments, suggesting this technique was reasonably atraumatic and may be well tolerated. In addition, successful retinal translocation around the axis of the optic nerve following 360° peripheral retinectomy was also demonstrated. Reattachment of the retina resulted in retinal folds due to the rigidity of the medullary wings. Additionally, maximal shaving of the vitreous base was found to be critical for creation of the retinectomy. Residual vitreous resulted in increased difficulty and less predictability during creation of the retinectomy.2
The rabbit model has also been used to demonstrate the utility of calcium- and magnesium-free infusate in reducing retinal adhesion without creating cellular toxicity.3,4 Collateral damage to photoreceptor outer segments was also reduced by using calcium- and magnesium-free solutions. Temporary electroretinogram (ERG) depression of b-wave amplitude was noted on postoperative day 1, but this was transient.3 Animal studies have also revealed that dark adaptation results in easier retinal detachment with reduced retinal damage. In model surgery, a red-free intraocular light source was used during the surgical procedure to prevent reversal of dark adaptation.4
Historical perspective and evolution of technique
In 1983, Lindsey first proposed translocation of the retina.5 Tiedeman et al. published their proposal for retinal translocation in 1985.6 Machemer published the first human surgical cases in 1993. All three subjects had AMD and were treated with 360° peripheral retinectomy and translocation (MTS360, MT360).7 The original MTS360 technique involved pars plana vitrectomy with transscleral injection of subretinal fluid with 360° retinectomy, removal of subretinal blood and choroidal neovascularization (CNV); partial fill with silicone oil; retinal translocation; complete silicone oil fill, and finally laser retinopexy. All surgeries were performed under general anesthesia. One of the original patients had significant improvement in central acuity (1/200–20/80).7
The original procedure has gone through multiple evolutionary iterations with major developments by Eckardt et al., Toth and Freedman, and Tano.8–10 Changes to technique have focused on facilitating retinal detachment, effectively translocating the macula, reducing complications (e.g., proliferative vitreoretinopathy, PVR), and decreasing the duration of surgery.8–18 Because retinal rotation results in significant cyclotorsion,8,9,12,14,17,19 extraocular muscle surgery to counter-rotate the globe is used routinely to manage the cyclotropia.8 Attempts at reducing the extent of the retinectomy (e.g., <360°) have not been widely adopted due to very high rates of PVR.10,11,20,21
Variations on MTS360 also included the development of limited macular translocation (LMT) which shifted the macula a shorter distance than with MTS360. De Juan developed a technique to shorten the sclera following detachment of the superotemporal retina across the macula. This resulted in redundant retina that allowed the foveal center to be relocated downward by gravity after surgery, by positioning the patient upright with a partially gas-filled eye. Because of the variable and limited distance of macular displacement, this procedure has decreased in use.22–24
Principles of foveal relocation
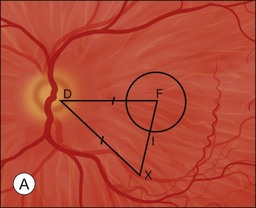
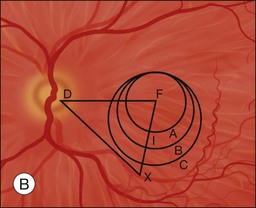
The primary goal of macular translocation surgery is to relocate the fovea to a new location of healthier subretinal tissues in order to preserve and maintain foveal function to maximize visual acuity. Thus the fovea should be relocated to an area outside that of the subfoveal lesion.1 The distance from the preoperative foveal center to the edge of the lesion is the minimum desired translocation. The distances of postoperative foveal displacement versus the minimum desired translocation have been found to be predictors of anatomical success following macular translocation.25
Characteristics of the underlying lesion (e.g., shape, size) determine the minimum desired translocation. Most surgeons move the retina further than the minimum distance, in order to have a reasonable margin between the lesion edge and the new foveal location. Lesion location is also critical to required translocation distance. Central larger lesions may require the same displacement distance as smaller eccentric lesions (Fig. 120.1). The postoperative foveal location in MTS360 is highly predictable, since the new foveal location is determined intraoperatively and the retina is reattached at the new location. In LMT, the postoperative foveal location is much less predictable because the fovea is not placed at a new position but rather settles to a new position after surgery. Because LMT results in a maximum translocation distance of 1500 µm, frequently with lesser displacement, and is generally only in a downward direction, it is frequently insufficient for treatment of subfoveal lesions that progress to surgical intervention.25–30 On the other hand, MTS360 is more versatile with an average foveal displacement of 3500 µm and the capacity to translocate the macula upward or downward.8–11
Preoperative considerations
Indications
Although clear guidelines for the indications for macular translocation are not established, there are several generally agreed upon principles. Anti-VEGF (vascular endothelial growth factor) therapy is clearly the first-line treatment for subfoveal neovascular disease.31,32 MTS360 surgery is only considered in patients with bilateral central vision loss due to the postoperative torsional diplopia. The surgical eye is typically the eye with better visual potential, more recent vision loss and greater preservation of retinal architecture. It may be considered in nonresponders to anti-VEGF treatment, eyes with extensive subretinal fibrosis, or some cases of massive subretinal hemorrhage, retinal pigment epithelial tears or submacular diseases that are not VEGF-driven.8,9,12,15,17,26,33–39 Although macular translocation has been reported in numerous diseases including neovascular AMD, non-neovascular AMD with geographic atrophy, myopic degeneration with CNV, ocular histoplasmosis with CNV, adult-onset foveomacular vitelliform dystrophy, punctate inner choroidopathy with subfoveal CNV, angioid streaks with CNV, North Carolina macular dystrophy, and central serous chorioretinopathy, there are cases where it has a lower likelihood of success.8,9,12,15,17,26,33,36–39 In cases of central geographic atrophy with AMD, the atrophy has often recurred in the new foveal location, and many would consider this a contraindication to surgery. Underlying ocular inflammatory diagnoses (e.g., punctate inner choroidopathy, ocular histoplasmosis) may also be associated with worse outcome.33 General inclusion and exclusion criteria for patients being assessed for MTS360 are outlined in Table 120.1.
Table 120.1 Inclusion and exclusion criteria for macular translocation with 360° peripheral retinectomy
* Duration of vision loss determined by patients’ reports of when they were unable to perform vision-related daily activities such as reading and driving.
Diagnostic testing
Preoperative evaluation for macular translocation may include fundus photography, fluorescein angiography, optical coherence tomography, fundus autofluorescence, fixation, and microperimetry. These tests may be useful to identify eyes that are poor candidates for MTS360 such as eyes with severe macular chorioretinal scarring, extensive neurosensory retinal atrophy, widespread RPE atrophy, CNV in the site of proposed translocation, retinal angiomatous lesions, chorioretinal anastomoses, or unexpected retinal vascular disease. Unfortunately, overall these tests, including fixation and microperimetry have been shown to be poor predictors of 1 year visual outcomes in patients who were considered eligible for MTS360.40
Optical coherence tomography (OCT) has revolutionized the diagnosis and management of numerous vitreoretinal diseases. The high-resolution view of the ultrastructure of the retina with spectral domain can be helpful in identifying outer retinal atrophy which may limit the utility of macular translocation. Histological evidence has shown significant photoreceptor atrophy overlying disciform scars.41 If outer retinal atrophy is present in the macula on OCT, patients are probably unlikely to realize improvement in visual function after macular translocation. Surprisingly, retinal thinning on time domain OCT before surgery was associated with good visual outcomes in one study.40 However, time domain OCT lacks the resolution of SDOCT imaging, therefore resolution was not sufficient to identify photoreceptor layer details. In some cases, perhaps the more compact retina was healthier than the edematous retina before MTS360. Additionally, OCT may be helpful in identifying other structural abnormalities that are important to preoperative planning (e.g., large intraretinal cysts, areas of adhesion).
Fluorescein and/or indocyanine green angiography are utilized to assess the extent, location, and activity of the CNV and/or submacular disease process. Additionally, preoperative fluorescein angiography can be helpful in identifying retinal angiomatous proliferation or chorioretinal anastomosis, which should be considered in surgical planning, as these can result in a retinal tear or significant intraoperative hemorrhage during the separation of the retina from the RPE and choroid.42,43 Additionally, the RPE integrity and perfusion status of the choroid and retina can be assessed. Fundus autofluorescence can also be useful for evaluation of the RPE health of the future subfoveal RPE bed. Hyper- or hypofluorescence in the area of the future foveal location should be considered before committing to surgery.
Surgical technique for macular translocation
MTS360
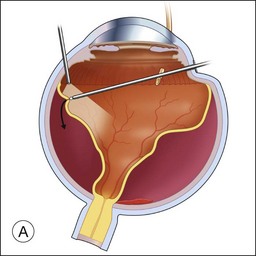
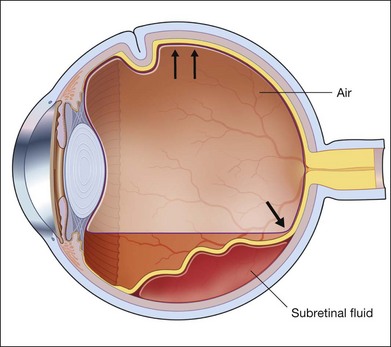
If a patient is phakic, cataract extraction and intraocular lens placement are performed simultaneously at the time of macular translocation. Complete pars plana vitrectomy with elevation of the posterior hyaloid, if attached, is the first step of MTS360. Careful, close shaving of the vitreous base with the vitreous cutter and 360° of scleral depression is performed. Retinal detachment is induced with subretinal fluid injection through a retinotomy that is typically located peripherally within the vitreous base. Posterior retinotomies have been associated with greater epiretinal membrane formation (Fig. 120.2A). A specialized cannula is utilized to infuse the subretinal fluid.8–11,14,15 A very small retinotomy is made inferonasally with the vitreous cutter at the ora serrata. The retinotomy is marked using endodiathermy. In order to limit back flow, a silicone-tipped cannula larger than the retinotomy is used to infuse subretinal fluid to detach the retina.9 If progression of retinal detachment does not occur, a fluid–air exchange is performed which facilitates subretinal fluid displacement from areas of detached retina to areas of attached retina. Although animal models have shown that calcium- and magnesium-free solutions may facilitate retinal detachment, prolonged use of these solutions in humans may result in higher postoperative keratopathy and the use of these solutions is not common.
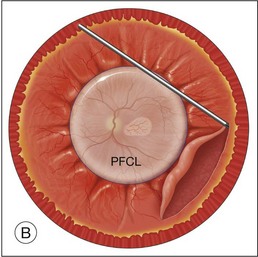
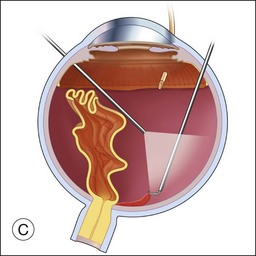
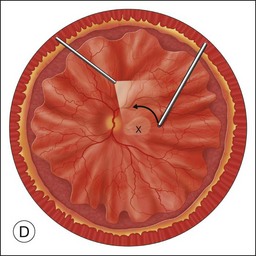
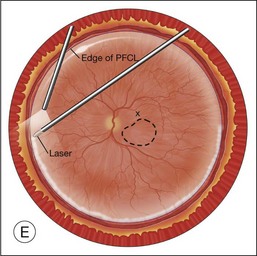
Once the retina is totally detached, the retina is cut at the ora serrata with the vitreous cutter (peripheral retinectomy)9–11 or retinal scissors (peripheral retinotomy) (Fig. 120.2B).8,10,11,15 Perfluorocarbon liquid (PFCL) is utilized over the posterior pole of the retina to stabilize the retina while performing the peripheral retinectomy. Once the retina is completely free from the ora serrata, the PFC is removed and the retina is reflected allowing removal of the subfoveal lesion, if present (Fig. 120.2C). The retina is then translocated into the new position. Most commonly, the retina is translocated superiorly (Fig. 120.2D). A small bubble of preretinal PFCL is utilized to stabilize the retina while the translocation is performed. Multiple instruments have been utilized for the retinal manipulation during translocation including retinal forceps8,10,11 and a silicone-tipped needle,14 although the diamond-dusted silicone-tipped needle is excellent for atraumatically snagging the retinal surface with very gentle pressure over an arcade, and then sliding the retina. The tip may be connected to a syringe containing perfluorocarbon liquid.9 Once appropriate displacement has been achieved (typically approximately 45° off the CNV bed, which equates to the center of the old CNV bed under the inferotemporal arcade), additional PFCL is added to reattach the retina in the manner of a giant retinal tear. Laser photocoagulation is then applied to the retinectomy margins under PFCL tamponade. A direct PFCL/silicone oil exchange is performed to avoid retinal slippage (Fig. 120.2E).
Limited macular translocation
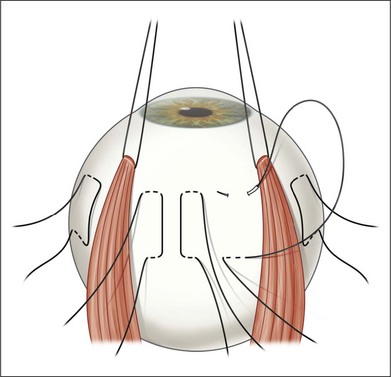
Rectus traction sutures are placed prior to vitrectomy under the lateral rectus and superior or inferior rectus muscles, depending on the planned location of translocation. Five to six horizontal mattress sutures, often 5–0 nylon, are preplaced before inducing a retinal detachment. The sutures are located along the equator, extending for 150–160°, centered in the superotemporal (or rarely inferotemporal quadrant as superior translocation is much more difficult to achieve) (Fig. 120.3). If inferior translocation is needed, the superotemporal quadrant is shortened by placing one suture nasal to the superior rectus, one suture inferior to the lateral rectus, and 3–4 sutures in the superotemporal quadrant. The sutures are left untied until after creation of the retinal detachment. Although this technique creates chorioscleral infolding, other techniques including chorioscleral outpouching have also been reported.44–46
Early postoperative management
Positioning
Positioning following MTS360 is based on the surgeon’s preferences. Approaches include face-down positioning or alternating side-to-side positioning thought to diminish pooling of factors for PVR in aqueous medium at the 6 o’clock position. With LMT, the patient is brought to an upright position (Fig. 120.4) for 24 hours to facilitate inferior translocation.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree