Chapter 117 Macular Hole
![]() For additional online content visit http://www.expertconsult.com
For additional online content visit http://www.expertconsult.com
Introduction
Macular hole (MH) is a round full-thickness opening in the foveal center. In most cases it is idiopathic, i.e., due to abnormal vitreofoveal traction. The role of the vitreous cortex in the pathogenesis of macular hole became better understood with the biomicroscopic observations of JD Gass.1 The advent of optical coherence tomography (OCT), which showed the partially detached posterior hyaloid, clarified the understanding of MH formation. Spectral domain OCT (SD-OCT) subsequently provided more detailed views of the initial foveal changes induced by vitreofoveal traction. Macular holes have been known about since the 19th century. However, they only aroused renewed interest after Kelly and Wendel2 had shown that pars plana vitrectomy (PPV) combined with vitreous cortex detachment and fluid–gas exchange could close MH in a significant proportion of cases, although it was assumed that the retina would be unable to heal. The success rate of MH surgery gradually increased progressively and this surgery is now one of the most successful vitreoretinal surgeries.
History
Macular hole was first described in 1869 by Knapp,3 of a traumatic case. Noyes4 (1871) gave a detailed ophthalmoscopic description of a traumatic case, and Ogilvie (1900) was the first to use the term “hole at the macula.”5 Early in the 20th century, Kuhnt6 (1900) and Coats7 (1907) suggested that the origin of MH was degenerative, although Zeeman8 (1912) and Lister9 (1924) attributed it to a vitreoretinal tractional mechanism. The modern history of MH started with J.D. Gass who proposed a staging system ranging from impending to full-thickness MH, on the basis of his biomicroscopic observations.10–12 Kelly and Wendel2 performed the first successful surgery of MH, and Hee and Puliafito13 were the first to describe the stages of MH on OCT scans.14,15
Epidemiology and risk factors for idiopathic full-thickness macular holes
Prevalence
The prevalence of MH reported in the literature varies greatly. It was 3.3 per 1000 in the Baltimore Eye Study;16 0.2 per 1000 in the Blue Mountains Study;17 0.9 per 1000 in the Beijing study,18 and 1.7 per 1000 in a study in Southern India.19 In the Beaver Dam study, the prevalence was 2.9 per 1000 as cited by McCannel et al.20 The incidence of MH was studied in a county in Minnesota (USA) and was found to occur in 7.8 persons per 100 000 per year, with a female-to-male ratio of 3.3 : 1. MH was bilateral in 11.7% of patients.20
Incidence in the fellow eye
The data on the incidence of bilateral MH vary considerably from 5% to 16%. In a retrospective study of 84 cases followed-up for 39 months on average, Akiba and colleagues21 (1992) found that 16.6% of patients had an MH in the fellow eye, and that the proportion rose to 36.8% when that eye presented with a foveal cyst or a central yellow spot. However, no MH occurred in the fellow eye when the vitreous was detached.21 In a retrospective study by Lewis et al.,22 the incidence was 13% in 4 years; it was 15.6% at 5 years in a prospective study of 144 patients, by Ezra and colleagues;23 and 7.6% at 6 years in another prospective study of 122 patients.24 In 2004, Chan et al.25 proposed the concept of Stage zero (0) MH to designate fellow eyes with a normal macular profile on OCT but a posterior hyaloid still attached to the foveal center. In their prospective study of 94 fellow eyes, MH only occurred in 4.5% of cases without visible vitreofoveal adhesion and in 42% of cases with a Stage 0 MH. However Chan et al.’s study was limited by the use of OCT1, which at this time did not allowed sufficiently accurate assessment of vitreofoveal adhesion. In 2011, Takahashi et al.26 selected 42 fellow eyes of 176 patients with MH, examined with OCT3, and presented with persistent vitreofoveal adhesion and early Stage 1 intrafoveal lesions. MH occurred in 11.9% of these eyes during the 5-year follow-up.
Pathogenesis, from posterior vitreous detachment to impending macular hole
History of theories on the pathogenesis of macular hole
Vitreomacular traction
Vitreomacular traction has long been suspected to be a cause of MH formation, but until the advent of OCT it was difficult to visualize the vitreofoveal interface routinely. However various indications of the role of the vitreous had been given in the literature. Thus, in 1952 Grignolo provided histologic evidence of strong vitreomacular adherence to the fovea.30 In particular cases, biomicroscopic observations strengthened the hypothesis that anteroposterior traction of vitreous fibers on the fovea played a role in the formation of MH.31–36 The biomicroscopic observation of opercula also strengthened the possibility that anteroposterior traction is the cause of MH.35,37
Foveal cyst
The existence of a foveal cyst in the fellow eye of an eye with MH was shown histologically by Kornzweig in 195038 and by Frangieh et al. in 1981.14 Also in 1981, Bronstein et al.,39 and in 1982, McDonnell et al.40 analyzed a series including fellow eyes and eyes with MH. They concluded that idiopathic macular cysts and holes were part of the same disorder and described the role of the vitreous body in their formation. On the basis of scanning laser ophthalmoscope observations, Kishi et al.41 deduced, in 1995, that foveal cyst formation due to vitreous traction was the first step in MH formation. In 1998, Folk et al.,42 who used laser slit-lamp photography, were able to obtain a direct image of the foveal cysts which they considered to be a pre-hole condition. This was confirmed in the same year by Hee and Puliafito13 using optical coherence tomography (OCT), which showed, for the first time, the vitreous cortex attached on the roof of a foveal cyst. Foveal cyst appeared then to be the initial change in the foveal structure that predisposes to the risk of progression to MH.43,44
Contraction of the premacular vitreous cortex
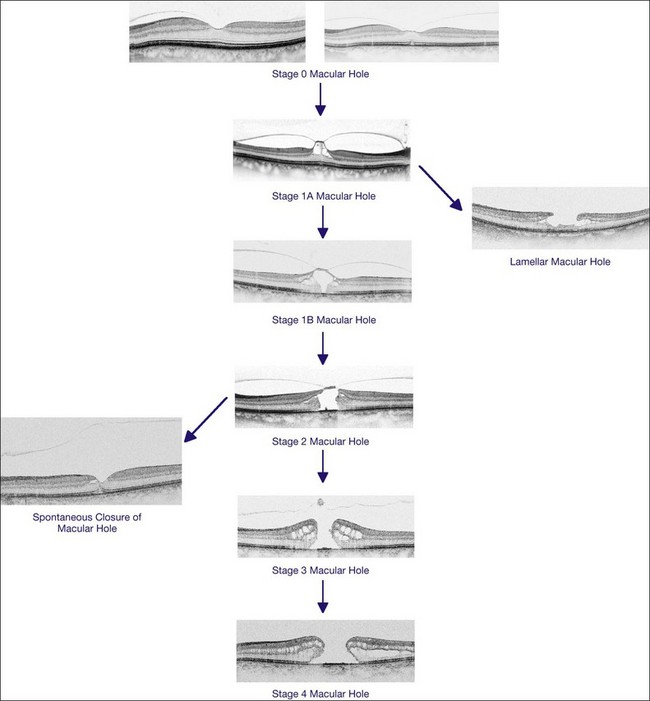
In 1988, Gass revised the biomicroscopic description of macular holes and proposed a new interpretation of the role of the vitreous in the pathogenesis of MH. He also proposed a system of MH staging, from impending to full-thickness MH. In his initial description in 1988, Gass postulated that the tangential contraction of the prefoveal “posterior hyaloid membrane” resulted in the detachment of the central photoreceptors and then in the opening of the fovea. The process of MH formation was divided into four stages which are still used today, despite the changes in their interpretation due to OCT findings. Finally, Gass proposed the use of a surgical approach design to prevent impending MH from evolving into full-thickness MH, by peeling off or dissecting the posterior hyaloid.1
Update on the pathogenesis of macular hole based on SD-OCT
For the first time, OCT has shown routinely the status of the posterior hyaloid or vitreous cortex, making it possible to combine in a single explanation, anteroposterior vitreous traction, the role of the vitreous cortex, and the formation of a foveal cyst as pre-hole condition. SD-OCT has now refined the description of the initial stages of impending MH, by showing discrete changes in the foveal tissue before the occurrence of a foveal cyst.26,45–47 These changes substantiate the concept of Stage 0 MH proposed by Chan et al. in 2004.25
Early stages of posterior vitreous detachment
How does PVD start in normal individuals?
The process of posterior vitreous detachment (PVD) is not yet clear, but its understanding has progressed thanks to the images obtained with OCT of the posterior hyaloid in normal eyes, and in fellow eyes of eyes with MH. It was usually thought that PVD occurred suddenly after a long period of vitreous gel liquefaction, intravitreal lacunae formation, and posterior hyaloid erosion by one of these lacunae, thus suddenly giving the liquefied vitreous access to the preretinal space.48–51 However, contrary to this belief, both OCT studies52 and ultrasonographic observations53 have established that in normal individuals, the process of PVD starts gradually at the posterior pole, around the fovea, and occurs relatively early in life, long before detachment of the Weiss ring.54
The attachments of the posterior hyaloid to the foveal center and optic disc are the last to be released. The reason for posterior hyaloid detachment from the retina remains unclear. It might be due to the action of anteroposterior forces that follow the direction of the collagen fibers of the vitreous body. An alternative explanation, which takes into account the presence of the precortical vitreous pocket,55–57 might be the passage of liquefied vitreous, behind the thinned posterior hyaloid which, combined with ocular movements, would extend its detachment. Both mechanisms might lead to the creation of oblique tractional forces on the foveal floor.
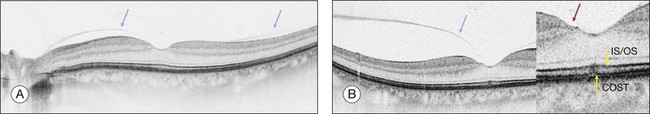
Early changes in foveal tissue (Stage 0 macular hole)
On the basis of the observation that in patients with MH, OCT of the fellow eye often showed partial vitreous detachment around the fovea,13,43,44 Chan et al.,25 who studied the risk of the fellow eye developing MH, proposed to call cases with partial perifoveal vitreous detachment but no foveal cyst, “Stage 0 MH” (Fig. 117.1 and see Fig. 117.2, available online). Today, in such cases, SD-OCT shows, even in asymptomatic eyes, slight changes in the foveolar structure long before the occurrence of the early foveal cyst that characterizes Stage 1 MH or impending hole. When this happens, it is possible to detect slight focal elevation of the inner curvature of the foveal center due to the traction exerted by the posterior hyaloid.46 Minor changes in the cone outer segment tips (COST, or Verhoeff membrane58), line at the foveal center,47 or subtle changes in the reflectivity of the center of the foveola, along an anteroposterior axis extending from the internal limiting membrane (ILM) to the inner segment/outer segment (IS/OS) junction line, may also be detected by high-definition SD-OCT scans. At this stage, complete normalization of the fovea may occur after vitreofoveal separation.
Impending macular hole
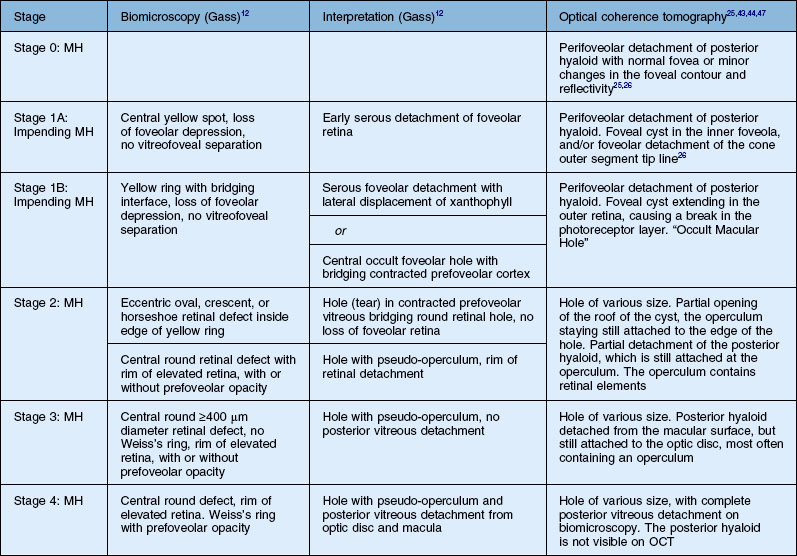
Stage 1A
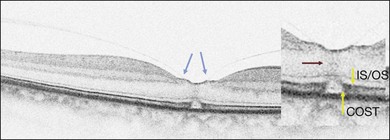
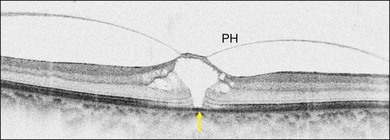
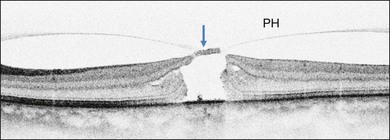
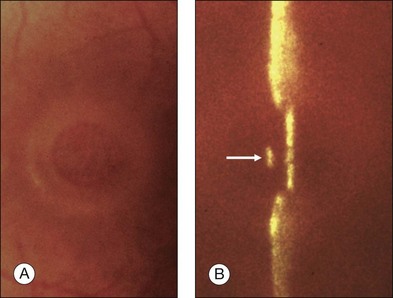
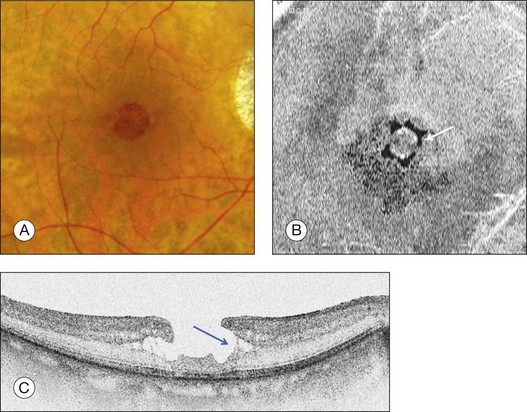
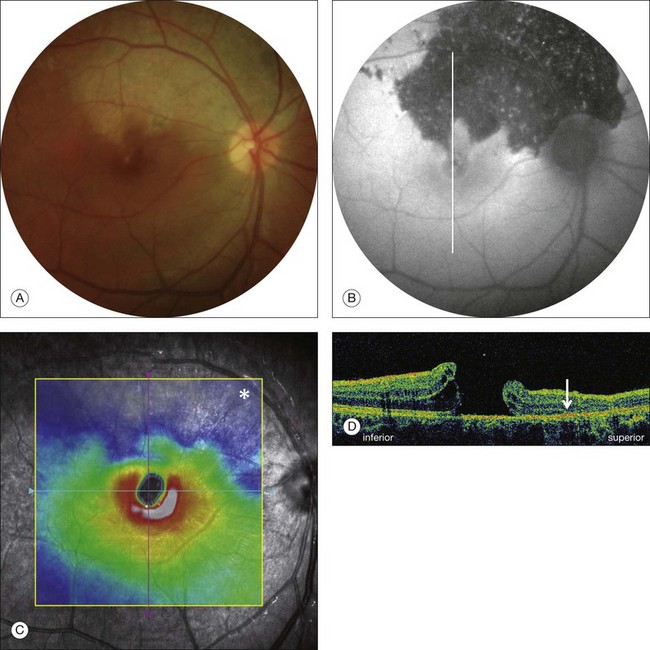
Stage 1A (Fig. 117.3 and see Fig. 117.4, available online) was defined by Gass as a central yellow spot and loss of the foveal depression associated with no vitreofoveal separation. The yellow spot was thought to be due to early serous detachment of the foveolar retina.12
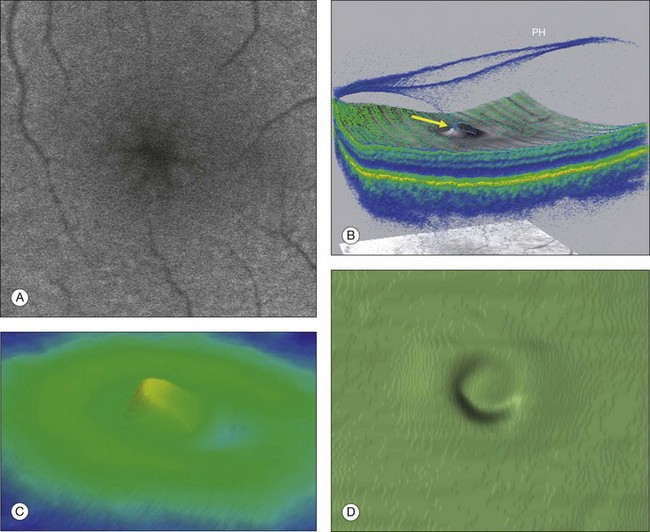
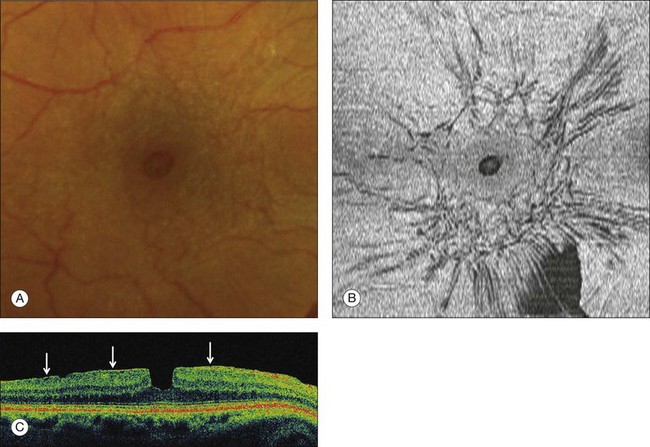
Fig. 117.4 Stage 1A impending macular hole (same case as in Fig. 117.3). (A) Autofluorescence photograph of the macula showing that the clear spaces of the cystic spaces encroach on the xanthophyll. (B) Reconstruction of the volume of the vitreoretinal interface showing the cone of the posterior hyaloid (PH) adherent to the top of the foveal pit (arrow). (C,D) Three-dimensional views of the foveal surface showing the elevation of the roof of the foveal cyst into the foveal pit.
OCT subsequently showed that Stage 1A was in fact characterized by an inner foveal cyst. The foveal floor is elevated by the traction exerted by the posterior hyaloid which is detached around the foveola but still attached at its center.43,44 Some dissociated Henle fibers are often visible inside the cyst but the retinal outer layers may be intact.26,44 However, SD-OCT has shown that in some cases the photoreceptor IS/OS layer may be elevated or even disrupted at the foveal center. Moreover, Takahashi et al.47 showed that the yellow spot might be due to the foveolar detachment of the cone outer segment tip line, even before the development of inner splits or cysts.
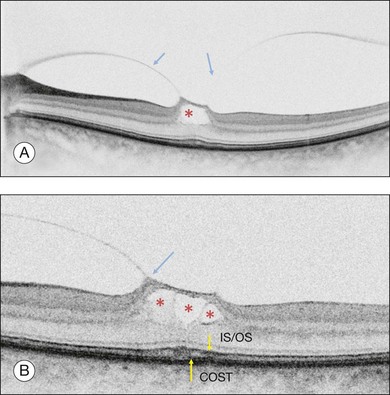
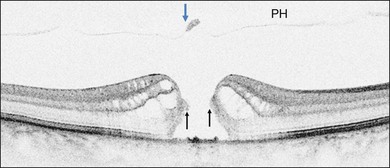
Stage 1B
Stage 1B (Fig. 117.5) was also called an occult hole by Gass,12 who characterized this stage as the combination of a yellow ring in the fovea, the absence of vitreofoveal separation and the loss of the foveal depression. These features were interpreted as centrifugal displacement of the foveolar retina, with the contracted pre-foveolar cortex continuing to bridge the hole. OCT has shown that the posterior hyaloid is detached from the macular surface except at the foveal center, and that the inner foveal cyst characterizing Stage 1A is completed by disruption of the outer retina up to the retinal pigment epithelium (RPE).26,43,44,47,59 In that sense, Stage 1B impending MH is a true occult MH. The yellow ring is due to the edematous border of the disrupted outer retina.44
Clinical and imaging features of full-thickness macular hole
Stage 2 macular hole
Stage 2 MH (Fig. 117.6) was described as an eccentric oval, crescent or horseshoe-shaped retinal defect inside the edge of the yellow ring.12,60 This was interpreted as a tear in the contracted pre-foveolar vitreous tissue bridging the round retinal hole, with no loss of foveolar retina.12 OCT however, has shown that the incompletely detached operculum is pulled in an oblique direction by the incompletely detached posterior hyaloid.13,43 Moreover, recent observations by Takahashi and colleagues showed that neuronal elements formed a constitutive part of the operculum at least in some cases.45,47 This observation corroborates the histologic findings of Ezra, which showed the presence of cones constituents in two-thirds of the opercula he examined.61
Stage 3 macular hole
According to Gass’s definition, the Stage 3 MH (Fig. 117.7 and see Fig. 117.8, available online) is a central round retinal defect more than 400 µm in diameter, with a rim of elevated retina, with or without pre-foveolar pseudo-operculum and without a Weiss’s ring. In fact, OCT has shown that whereas the vitreous is not detached from the optic disc in these cases, it is completely detached from the retinal surface over the posterior pole and is not connected to the hole edge. Moreover, precise measurement of the hole diameter on OCT scans has shown that the size of Stage 3 MH varies greatly. Although the diameter of Stage 2 holes generally tends to be smaller than that of Stage 3 holes, large Stage 2 and small Stage 3 holes are in fact not uncommon.47,62
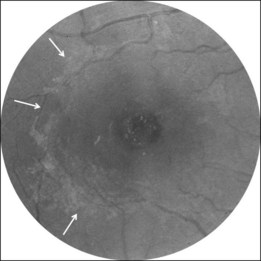
Macular hole and epiretinal membrane
A noncontractile epiretinal membrane (ERM) (Fig. 117.9) may cover the macular surface around the hole. It is especially well visualized on blue reflectance photographs.63 The frequency of the formation of an ERM increases with the stage, duration, and size of the hole.63–65 However, this does not affect the surgical prognosis once the membrane has been peeled off during surgery.
OCT classification of macular hole
OCT has now become an essential complement to biomicroscopy for the diagnosis of macular diseases and especially macular holes, and the Gass’s classification now needs to be revised according to the OCT features (Fig. 117.10). However, the classification in four stages is still useful, although the interpretation of each stage should be updated. Diagnosis of Stage 1, i.e., impending MH, is helpful in detecting eyes at risk of full-thickness MH. It should also be useful to add a Stage 0 to the present classification. At this stage, the posterior hyaloid is still attached to the foveal center, sometimes resulting in minor changes in the contour of the foveal pit. The special feature of Stage 2 is that the posterior hyaloid is still attached to the hole edge and might constitute the target of vitreolysis drugs as a first-line nonsurgical treatment. Stages 2 and 3 have in common the fact that the vitreous is not detached from the optic disc, and in both stages, detachment of the vitreous is one of the goals of surgery in these cases. By Stage 4, the vitreous is already detached from the retina and optic disc pit and surgery is easier. The diameter of the hole, which is widely accepted as a prognostic factor of the surgical success rate, is weakly correlated with the anatomic stage of the hole. Lastly, the present classification does not take into account the possible presence of an epiretinal membrane around the hole, which is more frequent in Stages 3 and 4 (Table 117.1).
Differential diagnosis
Lamellar macular hole
The term “lamellar macular hole” (LMH) was coined by Gass in 1975, to characterize a macular lesion resulting from the opening of the central cyst of a cystoid macular edema. The term “lamellar hole” was used to describe both the end stage of a cystoid macular edema14 and the aborted process of formation of a macular hole.44,66 With the advent of high resolution or spectral domain OCT, the thinning of the foveal center due to the contraction of an epiretinal membrane was also referred to as a lamellar macular hole.67
Histology of lamellar MH
Frangieh et al.14 described the histology of 17 cases of LMH of various etiologies. The cases were characterized by thinning of the foveal tissue leaving the RPE and photoreceptor layers intact, but causing partial loss of the inner nuclear layer. A cleft between the inner and outer retina was present at the edges of the lamellar hole, as well as cystic changes. In some cases, remnants of vitreous bands adhered to the lamellar hole edge. An ERM was found in several cases around the lamellar hole edge14,68 and was thought to be the cause of certain lamellar holes.68,69
Biomicroscopy
On biomicroscopy, LMHs differ from FTMHs because they are rarely round but rather bi- or tri-lobulated. Their center is reddish like that of FTMH, but their edge is thin whereas the FTMH edge is thick and elevated. The Watzke test, performed with a thin slit, is negative, but in small FTMH, the slit may appear as continuous.70 However, when the aiming beam of a laser is reduced to its minimum intensity and projected onto the center of an LMH, it is usually perceived by the subject, unlike that which occurs with a FTMH.71
Optical coherence tomography
Lamellar MH, i.e., defects in the inner fovea due to the avulsion of the roof of a foveal cyst (either tractional or due to cystoid macular edema), is characterized on OCT (Fig. 117.11) by irregular thinning of the foveal floor, a cleavage between the inner and outer retina at the lamellar hole edge, and the absence of a contractile epiretinal membrane.66 Using high-resolution OCT, Witkin et al.67 also described cases combining foveal thickening due to ERM contraction, with stretching of the foveal edge resulting in the thinning of the foveal floor. These cases may in fact represent a type of macular pseudohole induced by both centripetal and centrifugal contraction of the ERM between several eccentric epicenters.
Macular pseudoholes
The term “macular pseudohole” was coined by Allen and Gass72 in 1976, to designate a roundish centrofoveal image seen on biomicroscopy, which was due to the centripetal contraction of an epiretinal membrane. This contraction induces the verticalization of the edge of the foveal pit. Vision may remain relatively good and there are no microscotomas.
Other round foveolar images
Microholes
The term “microhole” was coined by Cairns73 in 1988, long before the use of OCT, for dark-reddish lesions in the center of the fovea ranging from 50 to 150 µm in diameter. None of these microholes turned into idiopathic FTMH. Retrospectively, it is today difficult to define the type of case, which at the time could have been described as a microhole, and what its OCT image would have been. Another series of cases presenting with a reddish foveal spot was examined with OCT1 by Douglas et al.74 who concluded that neither a hole nor an intraretinal break was disclosed by OCT. A better indication was given by Zambarakji et al.75 who studied cases very similar to those described by Cairns, and found, with OCT3, a break in the photoreceptor layer at the foveal center. What have been called “microholes” might therefore not be FTMH but in some cases, spontaneously closed FTMH.62 However, other diseases may also cause similar breaks in the centrofoveal inner segment/outer segment (IS/OS) line. Disruption of this line may be transient, as in multiple evanescent white dot syndrome,76 or either persist as a sequelae in solar retinopathy,77 or after exposition to drugs such as “poppers”.78
Non-idiopathic (secondary) MH
Orbital trauma and high myopia
These MH (Fig. 117.13, available online) usually occur in children and young male adults, due to accidents at work or in the home, or during ball games.79,80 They are due to sudden axial compression of the eye resulting in equatorial expansion and retinal rupture of the fovea. The hole may be combined with other fundus lesions such as choroidal or Bruch’s membrane disruption, commotio retinae, sclopetaria, or peripheral breaks. The visual prognosis depends not only on the closure of the hole but also on the topography of the other lesions. If a choroidal disruption passes through the fovea, or if the post-contusive RPE atrophy includes the foveal center, the visual prognosis will be poor. Unlike that which occurs in idiopathic MH, the vitreous is not detached at all from the posterior pole. Surgery is usually successful, but a traumatic MH may also sometimes close spontaneously within the first weeks of its occurrence.81–83
High myopia
MHs are one of the complications of high myopia, together with posterior staphyloma and choroidal atrophy. Their pathogenesis may be different from that of non-myopic eyes because the vitreous cortex often remains adherent to the retinal surface. Some of these MHs occur after a progressive decrease in vision due to the worsening of foveoschisis.84 Others may be asymptomatic and may only be disclosed by OCT examination of the fundus.85 Despite progress in vitreous cortex and ILM removal, the anatomic and visual postoperative prognoses for MH in high myopia remains lower than for idiopathic MH (see Chapter 113, High myopia and the vitreoretinal complications).86,87
Other rare causes
Many other rare causes of secondary MH have been described, the most significant of which are listed in Table 117.2.
Table 117.2 Rare causes of secondary macular holes