Macular Hole
Jeremy D. Wolfe
Allen C. Ho
A macular hole is a full-thickness defect or tear of retinal tissue involving the anatomic fovea of the eye. It was first described by Knapp1 in 1869, in a patient with a history of ocular trauma. Early case descriptions of macular holes focused on young traumatized eyes, but it is now known that “idiopathic” age-related macular holes in older adults comprise the majority of these lesions.
Therapeutic treatment of macular holes has become possible only relatively recently, as a result of advances and refinements of vitreoretinal surgical techniques. The optimal management and treatment of macular holes continues to be a source of active research.
Epidemiology
Macular holes are uncommon, affecting approximately 3.3 per 1,000 people.2 The cause of macular hole varies, but the most common origin is idiopathic. In a series from 1982, 83% of macular holes were classified as idiopathic and only 15% were secondary to trauma (usually blunt trauma).3 Full-thickness macular holes rarely have been associated with other conditions, including proliferative diabetic retinopathy, optic disc coloboma, high myopia, choroidal neovascularization, Best disease, adult vitelliform macular degeneration, retinal arteriovenous communication, scleral buckling for retinal reattachment, pneumatic retinopexy for retinal reattachment, perforating retrobulbar injections, and topical pilocarpine.
The Eye Disease Case-Control Study Group4 attempted to identify possible risk factors for idiopathic macular holes. Of 198 study patients who were identified with macular holes, 143 patients (72%) were women. The only other statistically significant risk factors for macular holes identified by this study were high plasma fibrinogen levels (>2.95 g/L) and a history of glaucoma. Estrogen use was negatively associated with risk of macular hole. The explanations for these findings are speculative. The Eye Disease Case-Control Study Group did not examine two ocular characteristics that have previously been associated with macular hole formation: macular retinal pigment epithelial changes (involutional macular thinning) and macular vitreous attachment.5
Pathogenesis
The pathogenesis of macular holes has been the subject of much debate and controversy. Historically, three basic theories on the origin of macular holes have been proposed: the traumatic theory, the cystoid degeneration and vascular theory, and the vitreous theory.6
Knapp’s1 original description of a macular hole described a patient with ocular trauma. Two years later Noyes7 provided the first accurate and detailed ophthalmoscopic description of a macular hole that was secondary to blunt trauma in a 13-year-old girl (Figs. 21.1, 21.2). These and other early case studies led most observers to attribute macular holes to ocular trauma. Presumably, blunt ocular trauma could cause immediate macular hole formation from mechanical energy created by vitreous fluid waves and contrecoup macular necrosis or laceration.
 Figure 21.1. Traumatic macular hole. Note the irregularly shaped hole, the retinal pigmented epithelial (RPE) changes and the choroidal rupture signifying prior trauma to the eye. |
The first histopathologic descriptions of full-thickness macular holes were provided by Fuchs (1901)8 and Coats (1907)9 and supported a cystic degeneration theory of macular hole formation. Coats noted cystic intraretinal changes adjacent to the macular hole and surmised that it was the coalescence of these intraretinal cysts that led to the formation of the macular hole. He further recognized that this macular cystoid degeneration did not occur exclusively in patients with a history of ocular trauma.
The recognition of atraumatic macular cystoid degeneration and atraumatic macular holes led to the theory that cystoid degeneration was the final common pathway toward macular hole formation. Both Coats and Kuhnt10 believed that age-related changes in the retinal vasculature led to cystoid degeneration and were responsible for the atraumatic macular hole. This vascular theory of idiopathic macular hole was sometimes characterized as ocular angiospasm.11 The vitreous theory of macular hole formation was initially proposed by Lister12 in 1912. Lister theorized that anteroposterior fibrous vitreous traction bands were the cause of macular holes. Beginning in the 1960s, clinical and histopathologic studies began to highlight the relationship of the vitreous and macula in eyes with macular holes. Current theories of macular hole pathogenesis continue to be altered and refined, but nearly all continue to recognize the predominant role of the vitreous in macular hole formation.
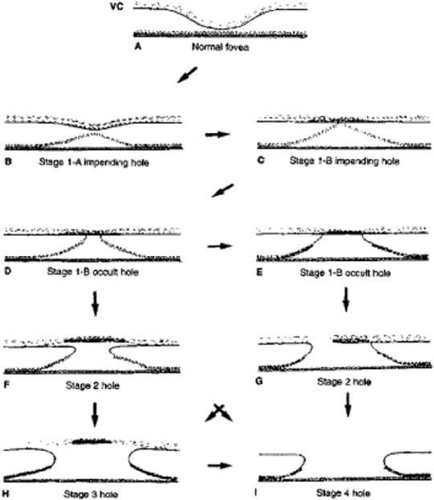
In 1988, Gass13 proposed a classification system for idiopathic macular holes, as well as a new hypothesis concerning their pathogenesis, suggesting that tangential vitreoretinal traction is responsible for hole formation. In 1995, Gass14 provided an updated biomicroscopic classification and anatomic interpretation of macular hole formation (Fig. 21.3).
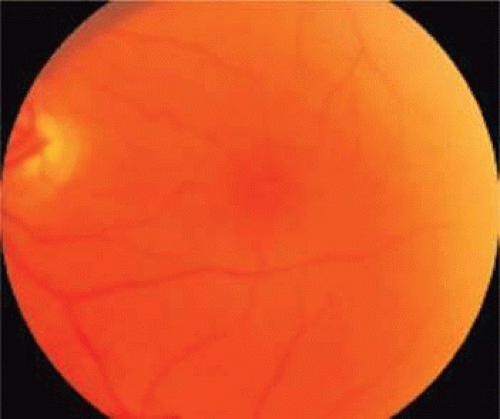
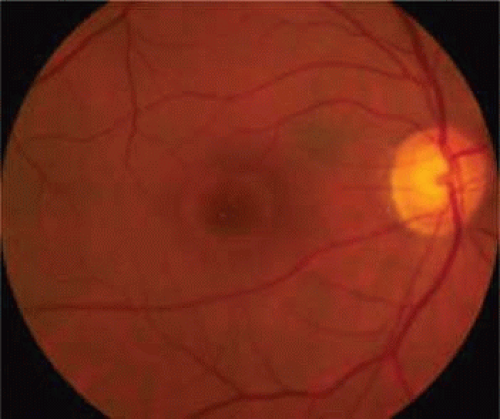
Spontaneous tangential contraction of prefoveolar cortical vitreous detaches the foveolar retina, creating a yellow spot approximately 100 to 200 μm in diameter (stage 1A: impending hole). The yellow color is felt to represent foveal intraretinal xanthophyll pigment (Fig. 21.4). As the foveal retina is elevated to the level of the surrounding perifoveal retina, the retinal receptor layer is stretched, and the yellow spot changes into a small donut-shaped yellow ring (stage 1B impending hole). Eventually, this thinned foveolar retina breaks at the umbo, and the surrounding retinal receptors, nerve fibers, and xanthophyll retract centrifugally, enlarging the ring (stage 1B occult hole, Fig. 21.5).
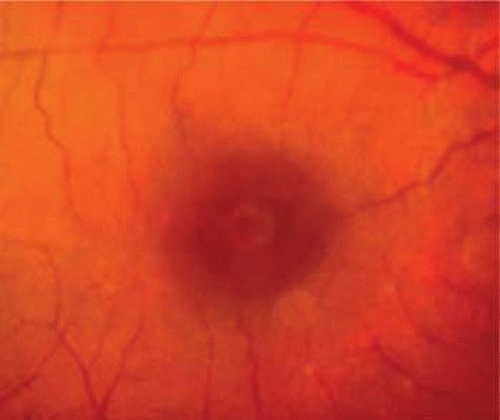 Figure 21.5. Stage 1B impending hole/occult hole. Note the small donut-shaped yellow ring, which develops as the retinal receptors retract centrifugally. |
Soon after, spontaneous vitreofoveal separation may occur. The contracted prefoveolar vitreous cortex (now detached from the underlying retina) may become visible as a semitranslucent prehole opacity (pseudo-operculum) lying anterior to a small foveolar hole. This condition is classified as a stage 2 full-thickness macular hole (Fig. 21.6).
Centrifugal retraction of foveolar retinal receptors continues to enlarge the diameter of the hole; Gass suggested that any hole 400 μm or larger should be classified as a stage 3 macular hole (Fig. 21.7).
After separation of the vitreous from the entire macula and optic disc, the hole is classified as stage 4, regardless of its diameter.
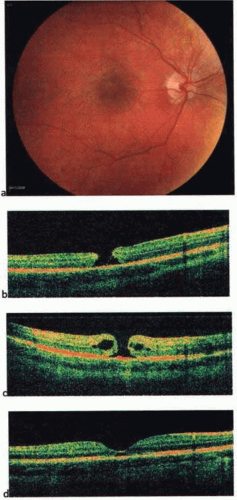
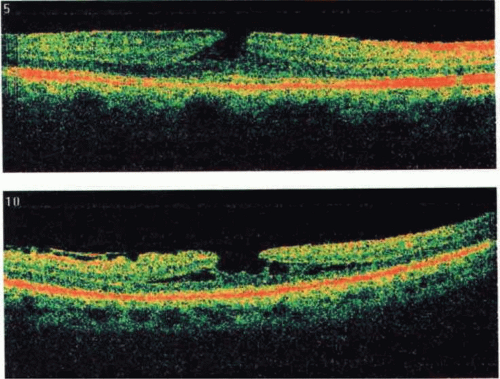
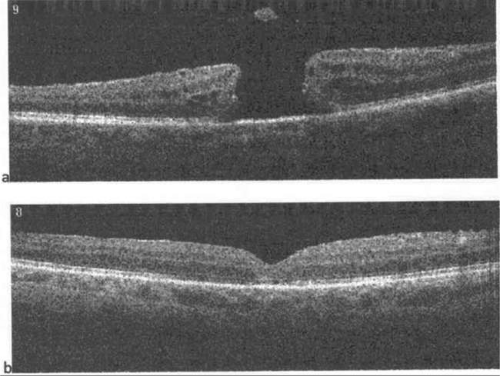
Recently, the introduction of optical coherence tomography (OCT) has allowed researchers to study the exact relationship between the vitreous and fovea in the development of macular holes. OCT is analogous to ultrasound but measures optical, rather than acoustic, reflectivity. The higher frequency of light waves compared with acoustic waves allows for a much higher longitudinal resolution (5 μm vs. 150 μm). Development of OCT technology over the last 15 years and its application in studying macular holes support the theory of vitreous traction in the pathogenesis of idiopathic macular holes. OCT evidence suggests that in most macular holes, the first step is actually the formation of a foveal pseudocyst (splitting of the retina at the fovea). It is postulated that anterior tractional forces from the prefoveal vitreous cortex cause this pseudocyst to form. The anterior wall of this cyst serves as a flap in stage 2 holes and as an operculum in stage 3 holes15,16,17 (Figs. 21.8, 21.9, 21.10 and 21.11).
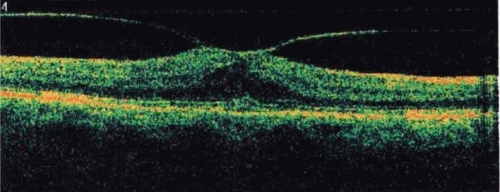 Figure 21.8. Spectral-domain optical coherence tomography (OCT) depicting vitreomacular traction with early foveal cyst. |
Histopathology
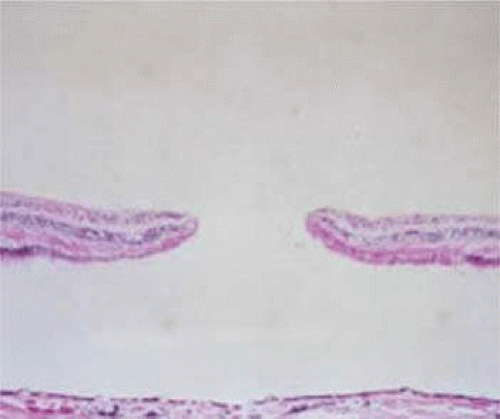
Two large histopathologic series have been reported on macular holes.18,19 Histopathologic examination of full-thickness macular holes demonstrates round or oval retinal defects surrounded by rounded retinal edges and a cuff of detached neurosensory retina with subretinal fluid (Fig. 21.12). In the most recent series, 79% demonstrated cystoid macular edema and 68% had epiretinal membranes. Photoreceptor atrophy was variable (200 to 750 μm, mean 480 μm, from the edge of the retinal margin) in this group of globes, some of which had long-standing macular holes. In some of the eyes, a thin, tapered layer of cortical vitreous was noted that may have caused traction on the edges of the macular hole.
The histopathology of lamellar macular holes has also been described.20 Lamellar holes are characterized by a partial loss of neurosensory retina, which looks like a sharply circumscribed round or petal-shaped red depression in the inner retinal surface. In half of these eyes, there was evidence of a thin layer of epicortical vitreous membrane causing tangential traction.
Diagnosis
Many macular lesions simulate macular holes or macular hole precursor lesions (stage 1A and 1B). In differentiating between the different entities, the most valuable clinical tool remains a careful contact lens biomicroscopic evaluation of the lesion, in conjunction with OCT.21 Clinical diagnostic tests for discriminating full-thickness macular holes from other lesions include the Watzke-Allen slit beam test and the laser aiming-beam test.
The Watzke-Allen test is performed at the slit-lamp biomicroscope by using a macular lens and placing a narrow vertical slit beam through the fovea. A positive test is noted when the patient detects a break in the bar of light. Most patients with a full-thickness macular hole report a positive Watzke-Allen test.22
The laser aiming beam test is performed similarly using a macular lens and placing a 50-μm laser photocoagulator aiming beam within the presumed macular hole. A positive test is observed when the patient cannot detect the aiming beam within the lesion but is able to detect it in surrounding intact tissue. This test is possibly more sensitive and more specific for full-thickness macular holes.23
Optical coherence tomography is valuable in the diagnosis of macular disease, including macular hole.24,25 The advertised axial resolution of the Stratus OCT and the spectral-domain OCT are 10 μm and 4 to 6 μm respectively. The high resolution obtained with current OCT technology make OCT (in conjunction with contact lens biomicroscopy) extremely sensitive in detecting macular pathology.26,27 OCT delineates partial-thickness and full-thickness macular holes, some of which can be challenging to differentiate by biomicroscopy by even a skilled observer.
Other diagnostic tests are generally less useful in detecting macular holes or distinguishing between macular holes and other simulating lesions. Amsler grid abnormalities are sensitive to macular lesions, but are not specific to macular hole lesions. Macular holes do not cause absolute scotomas detectable by Amsler grid testing.28 Fluorescein angiography may demonstrate early central hyperfluorescence in both true holes (79%)29 and pseudomacular holes (63%),30 and therefore is generally not a useful adjunct in distinguishing these lesions. Likewise, B-scan ultrasonography is generally not sufficiently sensitive to distinguish macular holes from masquerading lesions.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree