Macular Diseases
Nikolas J.S. London
Mitchell S. Fineman
Macular Epiretinal Membrane
Macular epiretinal membrane is an acquired formation of a semitransparent fibrocellular membrane in the macula.
Epidemiology and Etiology
Macular epiretinal membranes may be either idiopathic or secondary to other intraocular abnormalities, including retinal breaks, retinal vascular disease, uveitis, blunt and penetrating trauma, and surgery. Idiopathic macular epiretinal membranes are most common after 50 years of age.
Males and females are affected equally.
Bilateral macular epiretinal membranes occur in approximately 20% of patients, with the severity of the membranes usually asymmetric.
Most patients have posterior vitreous detachment (PVD), and it is thought that this phenomenon may contribute to formation of the membrane. After PVD, a portion of posterior cortical vitreous may be left behind on the macula, or the internal limiting membrane may be disrupted, allowing glial cells to proliferate on the surface of the retina. These proliferating glial cells form the epiretinal membrane. After retinal break formation and trauma, retinal pigment epithelial cells may escape into the vitreous cavity and settle upon the macula. These cells may then undergo metaplasia to glial cells, allowing the formation of macular epiretinal membranes.
History
If the macular epiretinal membrane is thin and not producing distortion of the retina, then the patient is usually asymptomatic. With progressive thickening and contraction of the membrane, patients begin to notice decreased vision, metamorphopsia, and macropsia or micropsia.
Most patients maintain vision better than 20/50.
Most epiretinal membranes progress slowly and occasionally improve spontaneously.
Important Clinical Signs
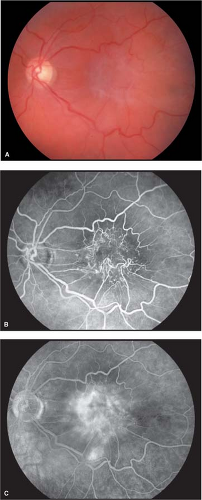
Macular epiretinal membranes initially appear as translucent, glistening membranes over, or adjacent to, the central macula (Fig. 2-1). There may be a “pseudohole” appearance over the fovea. As the membrane matures, it becomes more opaque (Fig. 2-2A).
There may be associated superficial hemorrhages or nerve fiber layer infarcts.
The membranes contract centrally and cause wrinkling and distortion of the underlying retina. This wrinkling and distortion gives rise to other names for this condition: cellophane maculopathy, surface wrinkling retinopathy, and macular pucker. If the distortion is severe, cystoid macular edema and even shallow tractional retinal detachment can occur.
Differential Diagnosis
Cystoid macular edema
Choroidal neovascularization (CNV)
Macular hole
Choroidal folds
Combined hamartoma of the retina
Retinal pigment epithelium
Diagnostic Evaluation
Usually measurement of visual acuity, Amsler grid testing (to assess for metamorphopsia, macropsia, or micropsia) and ophthalmoscopy suffice to make the diagnosis of macular epiretinal membrane. Slit-lamp biomicroscopy with either a noncontact or a contact lens greatly facilitates diagnosis.
Optical coherence tomography (OCT) has become standard in documenting the presence of an epiretinal membrane. OCT reveals a hyperreflective band on the surface of the retina, associated with retinal distortion, thickening, and/or cystoid edema in advanced cases.
Fluorescein angiography will clearly delineate retinal vascular distortion and leakage that may occur from cystoid macular edema (Fig. 2-2B, C).
Prognosis and Management
Most patients with uniocular macular epiretinal membrane and vision better than 20/50 are usually not significantly symptomatic and do not require intervention. Patients with vision of 20/60 or worse and those patients with intolerable distortion may benefit from pars plana vitrectomy with membrane peeling and removal (Fig. 2-3).
Rarely, the membrane may spontaneously release, leading to resolution of symptoms.
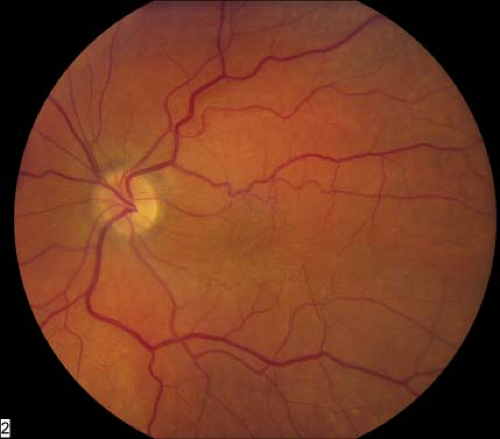 Figure 2-1. Macular epiretinal membrane. Glistening membrane over the macula causing distortion of central retina. |
Idiopathic Macular Hole
Idiopathic macular hole is an acquired full-thickness defect of retina in the central macula.
Epidemiology and Etiology
Idiopathic macular holes typically occur in the sixth through eighth decades of life with a 3:1 predominance in women.
The incidence of bilaterality is 5% to 10%.
Tangential vitreoretinal traction is the presumed cause of development of idiopathic macular hole.
The Gass classification of the stages of idiopathic macular hole development is helpful in understanding the progression of the disease and the biomicroscopic findings (Table 2-1).
Patients with a full-thickness macular hole in one eye and an impending macular hole with no PVD in the fellow eye are at substantial risk of progression to stage 2 macular hole in the second eye. Patients with a full-thickness macular hole in one eye and a normal retina with PVD in the fellow eye are at very little risk of progressing to macular hole in the second eye.
History
Patients usually report decreased visual acuity with central scotoma. There may be metamorphopsia.
Often patients notice decreased vision in the affected eye when the fellow eye is incidentally covered as for a routine eye examination.
Important Clinical Signs
Depending on the stage and severity of the macular hole, the visual acuity may be near normal or severely reduced to less than 20/400.
Amsler grid testing will often reveal metamorphopsia or a central scotoma. Ophthalmoscopy and slit-lamp biomicroscopy reveal findings consistent with the stage of the macular hole.
Table 2-1. Stages of Development of Idiopathic Macular Hole
Stage 1A
Early contraction of outer part of vitreous cortex with foveolar detachment (impending macular hole)
Stage 1B
Further vitreous contraction and condensation of the prefoveal vitreous cortex with foveal detachment (impending macular hole)
Stage 2
Small (<400 μm) perifoveal dehiscence
Stage 3
Larger (>400 μm) central full-thickness hole usually accompanied by a rim of retinal elevation; the posterior cortical vitreous remains attached; there may be a small operculum overlying the macular hole
Stage 4
Macular hole has an associated complete posterior vitreous detachment; these holes are usually large (>400 μm).
Based on Gass JD. Reappraisal of biomicroscopic classification of stages of development of a macular hole. Am J Opthalmol. 1995;119:752–759.
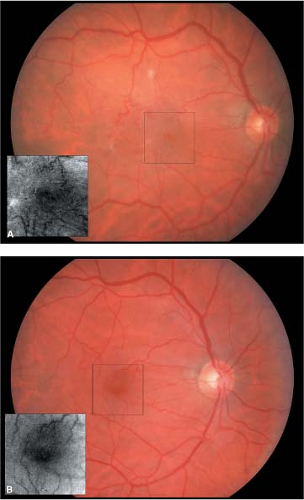
Stage 1 holes appear as a small yellow cyst or ring around the fovea with a loss of the foveal depression (Fig. 2-4).
Stage 2 holes appear as a small round or crescent-shaped defect in the fovea (Fig. 2-5).
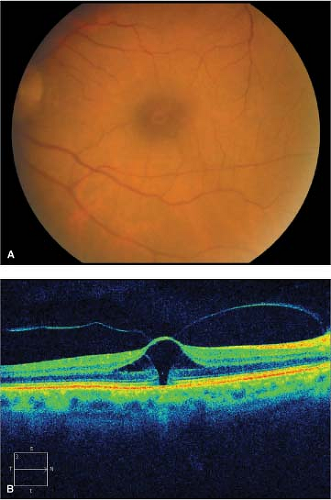
Stage 3 holes have a dark round defect in the fovea, often with a cuff of subretinal fluid accumulation (Fig. 2-6).
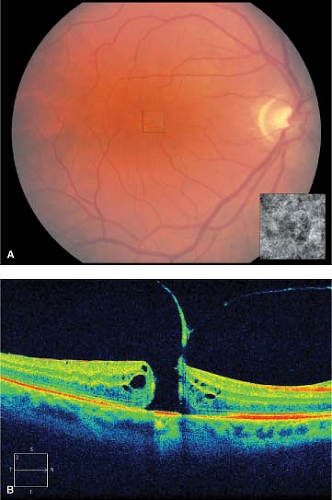
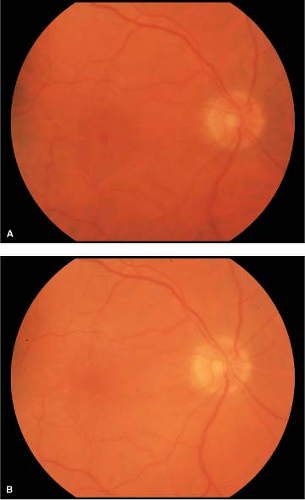
Stage 4 holes are often larger than stage 3 holes and are associated with a PVD (Fig. 2-7).
There are often small yellow dots in the center of the hole at the level of the retinal pigment epithelium (Fig. 2-8).
Associated Clinical Signs
The Watzke–Allen sign is the patient’s description of discontinuance in the center of a thin slit beam shone over the fovea.
Differential Diagnosis
Macular epiretinal membrane with pseudohole
Cystoid macular edema
Central serous retinopathy (CSR)
Choroidal neovascular membrane
Solar retinopathy
Adult vitelliform dystrophy
Diagnostic Evaluation
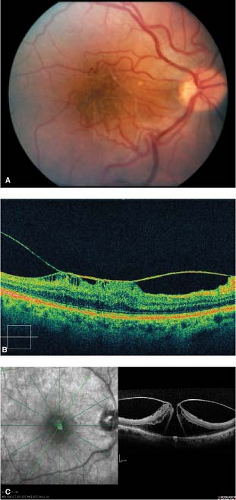
Clinical examination alone is often diagnostic. Fluorescein angiography in eyes with stage 2, 3, or 4 macular holes will reveal early central hyperfluorescence in the fovea corresponding to loss of xanthophyll pigment and retinal pigment epithelial depigmentation and atrophy at the base of the hole. OCT clearly distinguishes between partial- and full-thickness holes. (Fig. 2-9).
Prognosis and Management
No treatment is recommended for stage 1 macular holes because these resolve spontaneously in 50% of cases. Spontaneous resolution of more advanced stages of macular hole can occur, but it is rare.
Vitrectomy can be performed for more advanced stages of macular hole. The surgery consists of a standard pars plana vitrectomy, peeling of the posterior hyaloid, and injection of a long-acting gas such as perfluoropropane. Peeling of the internal membrane may also be done. Patients must then maintain face-down positioning for 1 to 2 weeks to allow the gas bubble to tamponade the hole. Recent evidence suggests that face-down positioning is not as critical as once believed, but this needs to be studied further. The success rate for macular hole surgery approaches 80% to 90% with closure of the hole and improvement in visual acuity (Fig. 2-10).
Negative prognostic indicators include a long duration of hole presence (i.e. greater than 1 year) and larger size of the hole.
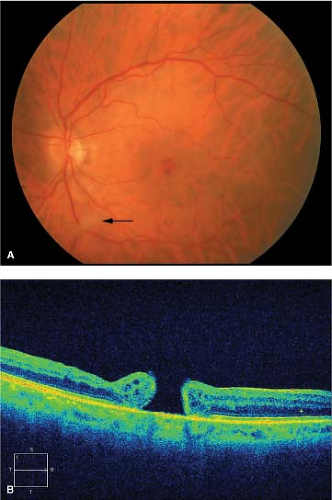
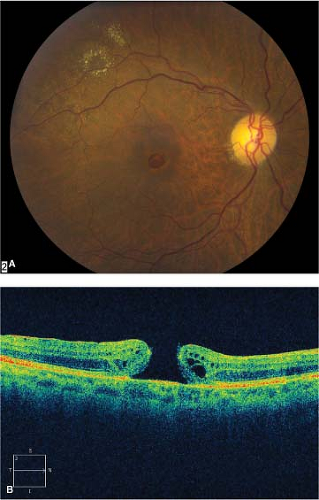 Figure 2-6. Idiopathic macular hole, stage 3. A. Stage 3 macular hole with cuff of subretinal fluid around the hole. B. Optical coherence tomography showing stage 3 macular hole. |
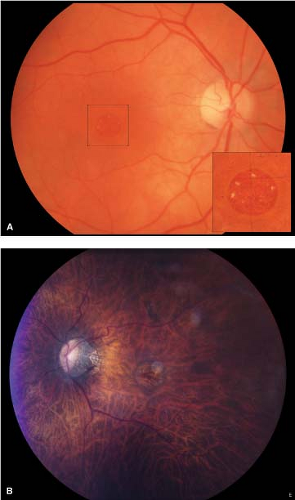 Figure 2-8. Idiopathic macular hole, chronic. A. Chronic stage 4 macular hole with subretinal precipitates (inset). B. Retinal pigment epithelial ring around macular hole indicates chronicity. |
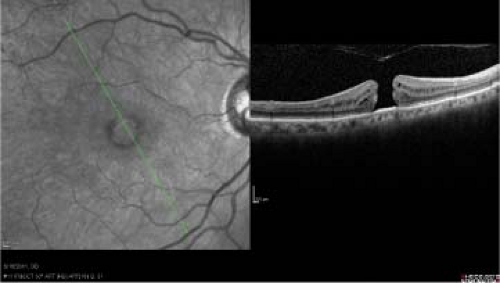 Figure 2-9. Idiopathic macular hole. Optical coherence tomography (OCT) of stage 4 macular hole showing complete defect in retina. |
Vitreomacular Traction Syndrome
Vitreomacular traction syndrome (VMTS) is an acquired condition in which there is partial separation of the posterior hyaloid with persistent attachment to the macula and, occasionally, the optic nerve head.
Epidemiology and Etiology
VMTS occurs in the same age group as those who develop PVD. PVD is uncommon before 50 years of age and is present in over 50% of people aged 70 years and older.
History
Patients who have VMTS experience progressive distortion and visual loss, which is often more severe than that occurring with macular epiretinal membrane.
Important Clinical Signs
The posterior hyaloid is visibly thickened. There is macular distortion, often with tractional retinal detachment in the macula. Retinal striae may be present. There may be traction in the peripapillary region. An epiretinal membrane may be seen clinically (Fig. 2-11).
Differential Diagnosis
Macular epiretinal membrane
Combined hamartoma of the retina and retinal pigment epithelium
Diagnostic Evaluation
Fluorescein angiography may reveal retinal vascular distortion and leakage. There may be cystoid macular edema and optic nerve edema.
Spectral-domain OCT helps determine the presence of VMTS as well as associated macular edema and tractional detachment.
Prognosis and Management
Occasionally patients may experience spontaneous improvement if PVD occurs.
Surgical intervention is indicated if visual acuity is reduced to 20/70 or worse. During vitrectomy surgery, the posterior hyaloid is removed, as are any epiretinal membranes in the macular region. The retinal architecture can be restored to a normal appearance. The vision can be improved but usually complete recovery does not occur due to residual macular edema.
Pharmacologic vitreolysis with microplasmin is currently under investigation for the treatment of VMTS, with promising initial results.
Cystoid Macular Edema
Cystoid macular edema (CME) is the result of accumulation of intraretinal fluid in the perifoveal region. Fluid accumulates in cystic spaces that may be visible clinically and on fluorescein angiography.
Epidemiology and Etiology
CME is most commonly seen after cataract surgery. Other types of ocular surgery, such as trabeculectomy, laser photocoagulation and cryoretinopexy, may also give rise to CME.
Less commonly, CME is seen in association with diabetic retinopathy, CNV, uveitis, retinal vein obstruction, perifoveal telangiectasis, retinitis pigmentosa, and other entities.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree