o_id) { scroll_to_id(event,’R6-16′); return false; }” xpath=”/CT{06b9ee1beed59419df930f3684536bce0fc4006bb10f91b1796b0d3a4d76c942a73fc532448567c6174d50603f427fd6}/ID(R6-16)” title=”6″ onmouseover=”window.status=this.title; return true;” onmouseout=”window.status=”; return true;”>6,7). An aptamer is a synthetic short strand of RNA oligonucleotide that adopts a specific three-dimensional conformation (8), joining to its target molecule (Fig. 16.1). Because aptamers have high affinity for their target molecules, the aptamer-target interaction is very similar to that of an antibody and antigen.
Aptamers are created using a process knowndow.status=”; return true;”>2).
Before the appearance of pegaptanib sodium, there was no FDA-approved treatment for the majority of patients with neovascular AMD. PDT was only approved for patients with subfoveal membranes with the predominantly classic subtype of CNV (3,4). Later, this indication was extended to cases with minimally classic or occult subtype of CNV, but only if they were less than four disk areas in size and were associated with progression of the disease. Extra- and juxta-foveal CNV had traditionally been treated with thermal laser photocoagulation, and prior to the advent of PDT, subfoveal lesions had been treated with laser as well (5). These traditional treatment approaches to neovascular AMD (i.e., PDT and thermal laser photocoagulation) were destructive therapies that led to retinal tissue scarring. Pegaptanib sodium emerged as a nondestructive therapy and showed efficacy for the treatment of most subtypes of neovascular AMD.
MECHANISM OF ACTION
Pegaptanib sodium is a pegylated aptamer with high affinity for VEGF (12). In the latter work, three stable, high-affinity candidate anti-VEGF aptamers were characterized, one of which was selected for development as pegaptanib. In a process called pegylation, the addition of a 40-kDa polyethylene glycol (PEG) moiety to the 5′ end of the aptamer improved bioavailability (7). An aptamer is a synthetic short strand of RNA oligonucleotide that adopts a specific three-dimensional conformation (8), joining to its target molecule (Fig. 16.1). Because aptamers have high affinity for their target molecules, the aptamer-target interaction is very similar to that of an antibody and antigen.
Aptamers are created using a process known as the systematic evolution of ligands of exponential (SELEX) enrichment process (9). Through this process, 1014 aptamers are produced in a test tube and then screened for their ability to bind or interfere with various target molecules (9). Using VEGF165 as the target, SELEX methodology was performed in three separate approaches at NeXstar Pharmaceuticals, ultimately producing the aptamer that became pegaptanib. Aptamers that blocked the actions of VEGF in vitro were first described in 1994 (10) followed by the use of amino-substituted nucleotides to improve the resistance of anti-VEGF aptamers to nuclease attack in 1995 (11) and, subsequently, the use of additional substitutions to further improve stability and affinity in 1998 (15); the interaction between pegaptanib and this domain is shown in Figure 16.1B (16).
CHEMICAL STRUCTURE
The molecular formula for pegaptanib sodium is C294H342 F13N107Na28O188P28[C2H4O]n (where n is approximately 900), and the molecular weight is 49 kDa (17). The chemical structure of pegaptanib sodium is depicted in Figure 16.2.
RATIONALE FOR CLINICAL USE
The VEGF family is a group of secreted proteins that include VEGF-A, -B, -C, -D, and placental growth factor (PlGF). These factoice/CT{06b9ee1beed59419df930f3684536bce0fc4006bb10f91b1796b0d3a4d76c942a73fc532448567c6174d50603f427fd6}/C16-FF1.gif?geom=500×500″>
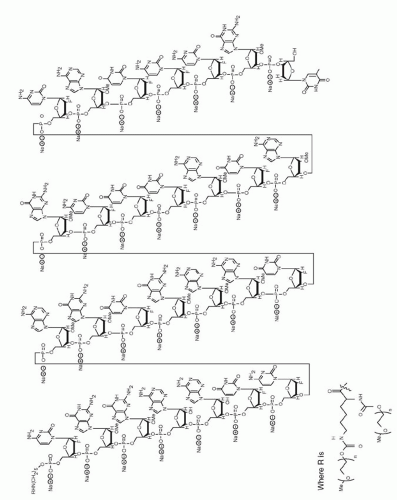
Figure 16.1 ▪ Pegaptanib structure and target binding. A. Sequence and predicted secondary structure of pegaptanib. 2′-O-methylated purines are shown in red, 2′-fluorine-modified pyrimidines are shown in blue, and unmodified ribonucleotides are shown in black. The site of attachment of a 40-kDa PEG moiety is shown. B. Interaction between the 55-amino-acid heparin-binding domain of VEGF165 and pegaptanib. (From Ng EW, Shima DT, Calias P, et al. Pegaptanib, a targeted anti-VEGF aptamer for ocular vascular disease. Nat Rev Drug Discov. 2006;5(2):123-132.)
The selectivity of pegaptanib derives from its interaction with cysteine-137, an amino acid that is contained within the 55 amino acid heparin-binding domain of VEGF, which is not present in VEGF121 (15); the interaction between pegaptanib and this domain is shown in Figure 16.1B (16).
CHEMICAL STRUCTURE
The molecular formula for pegaptanib sodium is C294H342 F13N107Na28O188P28[C2H4O]n (where n is approximately 900), and the molecular weight is 49 kDa (17). The chemical structure of pegaptanib sodium is depicted in Figure 16.2.
RATIONALE FOR CLINICAL USE
The VEGF family is a group of secreted proteins that include VEGF-A, -B, -C, -D, and placental growth factor (PlGF). These factors selectively bind and activate receptors located primarily on the surface of vascular endothelial cells. Among these, it is well known that VEGF-A acts as a key regulator of pathologic vessel growth and vascular permeability in ocular neovascular disease such as AMD (18,19). VEGF is produced by many cells types in the retina (20,21)gic vasculogenesis and vascular homeostasis. The studies mentioned above showed that VEGF164 was selectively increased in pathologic neovascularisation, and blocking it results in inhibition of pathologic angiogenesis and vascular permeability without damage to normal vasculature (26,27).
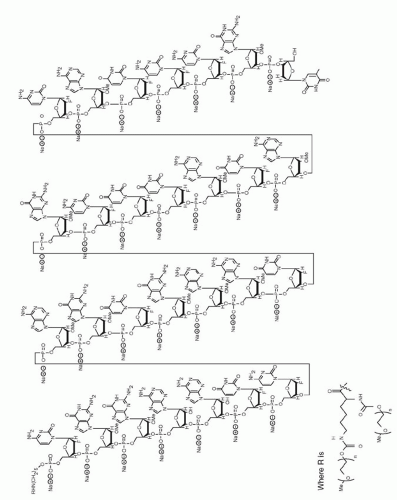 Figure 16.2 ▪ Schematic depicting chemical structure of pegaptanib sodium. (From Eyetech/Pfizer: Pegaptanib sodium injection in the treatment of neovascular AMD. Briefing Document for the FDA Dermatologic and Ophthalmic Drugs Advisory Committee. Rockville, MD, 2004.) | 25). Of the other isoforms, VEGF189 and VEGF206 are highly basic, are heparin binding, and exist primarily as matrix-bound forms. VEGF121, lacking the heparin-binding domain, is freely secreted (22). Pegaptanib sodium was developed to selectively inhibit the isoform VEGF165. In this way, the choice of aptamer was based on the characteristic of having high affinity and specificity for VEGF165. In fact, preclinical studies in mouse models of pathologic ocular neovascularisation demonstrate the effectiveness of pegaptanib sodium at inhibiting the activity of VEGF164 (the murine homologue to the human isoform VEGF165) (26,27). However, one of the main concerns during drug development was whether or not the inhibition of
The rationale behind this approach is that eliminating the stimulus for the formation of abnormal blood vessels, one can eliminate the unwanted effects of the abnormal blood vessels (i.e., intraretinal, subretinal, and sub-retinal pigment epithelium [RPE] fluid accumulation and hemorrhage) without the “collateral” damage induced by thermal laser photocoagulation or PDT. One of the main advantages of anti-VEGF treatment over thermal laser or PDT is that normal retina, RPE, and choroid are spared from damage (28).
CLINICAL PHARMACOLOGY
Pharmacokinetics
Absorption
In animals, pegaptanib is slowly absorbed into the systemic circulation from the eye after intravitreous administration. The rate of absorption from the eye is the rate-limiting step in the disposition of pegaptanib in animals and is likely to be the rate-limiting step in humans, too (17). Vitreous levels appear to follow an apparent first-order elimination process. Preclinical studies determined the terminal half-life in the vitreous to be 83 hours in a rabbit model (17).
Pharmacodynamics
Preclinical studies established that pegaptanib inhibited VEGF binding and VEGF-mediated cell signaling in cultured endothelial cells and that pretreatment of cells with pegaptanib inhibited proliferative responses only to VEGF165, but not to VEGF121 (<A onclick="if (window.scroll_to_id) { scroll_to_after a 3-mg monocular dose (10 times the recommended dose) (17). At doses less than 0.3 mg per eye, pegaptanib sodium plasma concentrations are not likely to exceed 10 ng/mL, which is more than 100 times less than the concentration observed in the nonclinical toxicology studies at the “no observed adverse effect level” doses (17). Circulating levels of pegaptanib sodium seen 4 to 6 weeks after an intravitreous 0.3-mg dose were below the lower limits of quantification (8 ng/mL) of the assay (17).
Excretion
After intravitreous and intravenous administration of radiolabeled pegaptanib to rabbits, the highest concentrations of radioactivity were obtained in the kidney. In rabbits, the component nucleotide 2′-fluorouridine is found in plasma and urine after single radiolabeled pegaptanib intravenous and intravitreous doses (17




Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree