Lucentis (Ranibizumab)
Joanna L. Goulah
D. Virgil Alfaro III
Carlos E. Ortiz
INTRODUCTION
Ranibizumab is marketed as LUCENTIS (ranibizumab injection) and was approved by the U.S. Food and Drug Administration (FDA) in June 2006 for the treatment of neovascular (wet) age-related macular degeneration (AMD).
Ranibizumab has also received regulatory approval for the treatment of neovascular AMD in more than 100 countries worldwide. Ranibizumab binds to the receptor binding site of active forms of vascular endothelium growth factor-A (VEGF-A), including the biologically active, cleaved form of this molecule, VEGF110.
VEGF is a secreted homodimeric protein that is a potent vascular endothelial cell mitogen (1). VEGF stimulates vascular endothelial cell growth, functions as a survival factor for newly formed vessels, and induces vascular permeability. VEGF expression is up-regulated by hypoxia as well as by a number of other stimuli. Although other angiogenic factors have been identified, VEGF is perhaps the most potent and specific, with a well-defined role in normal and pathologic angiogenesis. Inappropriate overexpression of VEGF has been hypothesized as playing a key role in the growth of solid tumors and in vascular retinopathies such as AMD (Fig. 17.1).
VEGF-A has been shown to cause neovascularization and leakage in models of ocular angiogenesis and is thought to contribute to the progression of AMD. Inhibition of inappropriate VEGF activity is a so-called antiangiogenic approach to the treatment of these diseases. The binding of ranibizumab to VEGF-A prevents the interaction of VEGF-A with its receptors (VEGFR1 and VEGFR2) on the surface of endothelial cells, reducing endothelial cell proliferation, vascular leakage, and new blood vessel formation. Ranibizumab, an anti-human VEGF, affinity-matured Fab, has been developed by Genentech as a therapeutic agent for treating ocular vascular disease by intravitreal (ITV) injection.
RANIBIZUMAB
Drug Biochemistry and Formulation
Affinity-matured ranibizumab is produced by standard recombinant technology methods in an Escherichia coli expression vector and bacterial fermentation. Ranibizumab is not glycosylated and has a molecular mass of approximately 48,000 Da.
The lyophilized form of the drug is produced from a freeze-drying process. The solid cake seen in each vial consists of protein and the excipient components that are stabilizing agents for the protein. The lyophilized form of ranibizumab requires reconstitution with Sterile Water for Injection (SWI), USP. After reconstitution with SWI, the highly concentrated protein solution is further diluted with vehicle prior to ITV administration. This presentation was useful in early clinical dose-ranging studies and was used in early phase I/phase II studies.
The liquid formulation is the commercial form. This presentation provides excellent stability and eliminates the need for reconstitution and further dilution. Liquid ranibizumab is formulated as a sterile solution aseptically filled in a sterile, 2-mL glass vial. Each vial is designed to deliver 0.05 mL of 10 mg/mL of ranibizumab aqueous solution with 10 mM histidine HCI, 10% α,α-trehalose dehydrate, and 0.01% polysorbate 20 (pH 5.5). The vial contains no preservative
and is suitable for single use only. Vials should be protected from light. The liquid formulation was assayed for purity and identity as described for the lyophilized form. The assay for potency is a biologically relevant, cell-based assay in which the ability to inhibit VEGF-dependent growth in vitro with a cell line requiring VEGF for growth is quantified.
and is suitable for single use only. Vials should be protected from light. The liquid formulation was assayed for purity and identity as described for the lyophilized form. The assay for potency is a biologically relevant, cell-based assay in which the ability to inhibit VEGF-dependent growth in vitro with a cell line requiring VEGF for growth is quantified.
VEGF THE TARGET
Human VEGF exists as at least six isoforms (VEGF121, VEGF145, VEGF165, VEGF183, VEGF189, and VEGF206) that evolve from alternative splicing of mRNA of a single gene (1). VEGF165, the most abundant isoform, is a basic, heparin binding, dimeric glycoprotein with a molecular mass of approximately 45,000 Da (1). Two VEGF receptor tyrosine kinases, VEGFR1 and VEGFR2, have been identified (2,3,4,5,6,7). VEGFR1 has the highest affinity for VEGF, with a Kd of approximately 10 to 20 pM (8), and VEGFR2 has a somewhat lower affinity for VEGF with a Kd of approximately 75 to 125 pM (9,10,11).
VEGF has numerous biologic functions, including regulation of VEGF gene expression under hypoxic conditions (1), mitogenic activity for micro- and macrovascular endothelial cells (4,5,6,7,12,13,14), and induction of expression of plasminogen activators and collagenase (15). Of particular significance to wet AMD are the angiogenic properties of VEGF, which have been demonstrated in a variety of in vivo models, including the chick chorioallantoic membrane (15,16), rabbit cornea (17), and rabbit bone (17). VEGF also functions as a survival factor for newly formed endothelial cells (16,18). Consistent with prosurvival activity, VEGF stimulates expression of the antiapoptotic proteins Bcl-2 and A1 in human endothelial cells (19). VEGF has been shown to induce vascular leakage in guinea pig skin (19). Dvorak et al. (20,21) suggested that an increase in microvascular permeability is a crucial step in angiogenesis associated with tumors and wound healing. According to this hypothesis, a core function of VEGF in the angiogenic process is the induction of plasma protein leakage. This outcome would result in the formation of an extravascular fibrin gel, which serves as a substrate for endothelial cells. This proposal may have substantial bearing for AMD as it is well known that permeability of the choroidal neovascularization (CNV) membrane results in the transduction of serum components beneath and into the retina, creating serous macular detachment, macular edema, and vision loss.
VEGF is expressed in an assortment of cells in the normal human retina. Colocalization of VEGF mRNA and protein is detected in the ganglion cell, inner nuclear and outer plexiform layers, the walls of the blood vessels, and photoreceptors (22). Retinal pigment epithelium (RPE), Müller cells, pericytes, vascular endothelium, and ganglion cells all manufacture VEGF (23,24).
Studies have documented the immunohistochemical localization of VEGF in surgically resected CNV membranes from AMD patients. Kvanta et al. (20) demonstrated the presence of VEGF mRNA and protein in RPE cells and fibroblast-like cells. Lopez et al. (19) noted that the RPE cells that were strongly immunoreactive for VEGF were present primarily in the highly vascularized regions of CNV membranes, whereas the RPE cells found in fibrotic regions of CNV membranes showed little VEGF reactivity. Kliffen et al. (25) also demonstrated increased VEGF expression in RPE cells and choroidal blood vessels in maculae from patients with wet AMD compared with controls.
An increase in VEGF expression has been noted in experimental models of CNV in rats and in nonhuman primates (26). In addition, transgenic mice with increased VEGF expression in photoreceptors (27) or RPE (28) developed neovascularization reminiscent of CNV seen in
humans with neovascular AMD. This further supports the participation of VEGF in ocular neovascularization. These lines of evidence suggest that VEGF is a practical target for therapeutic intervention in neovascular AMD.
humans with neovascular AMD. This further supports the participation of VEGF in ocular neovascularization. These lines of evidence suggest that VEGF is a practical target for therapeutic intervention in neovascular AMD.
Inhibition of VEGF Activity by Ranibizumab
In Vitro Evaluation of Ranibizumab
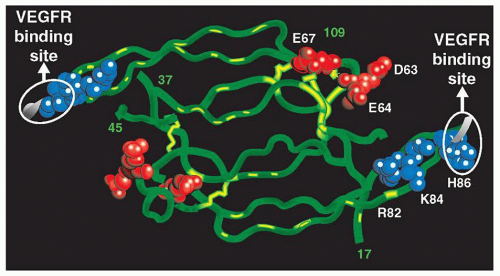
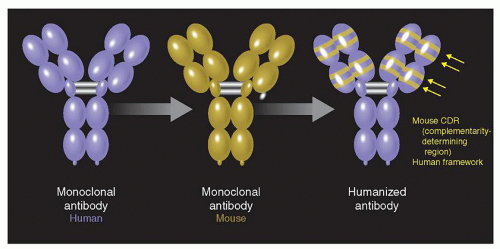
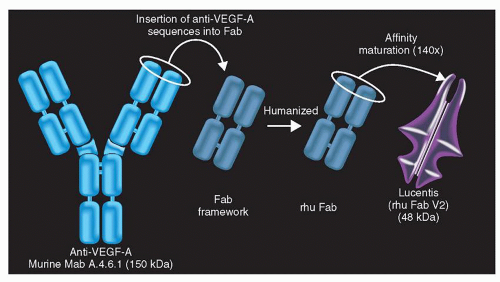
A number of antibodies that can bind VEGF or inhibit VEGF activity were considered in vitro and in vivo for molecule selection. muMAb VEGF A.4.6.1 is a full-length murine monoclonal antibody of the IgG1 isotype that continuously and potently defuses the biologic activities of VEGF, including the endothelial cell mitogenic activity, vascular permeability-enhancing activity, and angiogenic properties in the chick chorioallantoic membrane (29). This antibody also inhibits growth of various human tumor types in animal models1, recognizes all isoforms of VEGF, binds to VEGF with a Kd of approximately 8 × 10−10 M, and neglects to recognize other peptide growth factors. Bevacizumab (rhuMAb VEGF) is a humanized version of muMAb VEGF A.4.6.1 that was produced by site-directed mutagenesis (30) (Figs. 17.2 and 17.3).
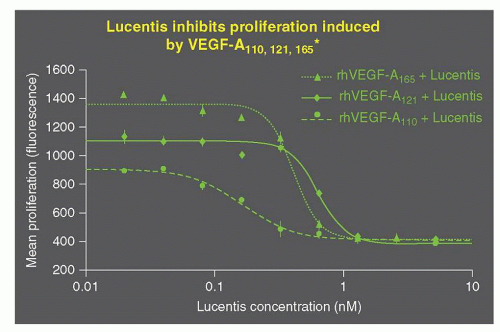
Ranibizumab is a recombinant humanized antibody Fab fragment that neutralizes VEGF. Ranibizumab shows high affinity for binding VEGF (Fig. 17.4). In addition, ranibizumab potently inhibits survival of human umbilical vein endothelial cells (HUVEC) stimulated with 5 ng/mL of recombinant human VEGF. Ranibizumab also inhibits VEGF-mediated vascular permeability in the Miles assay (Fig. 17.5).
Animal Models
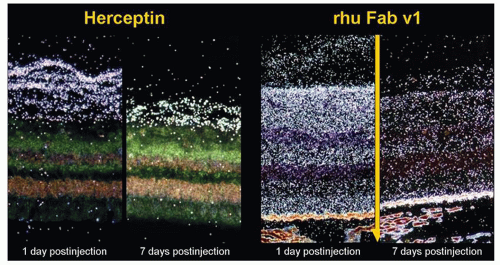
Nonclinical pharmacology studies to measure the safety of ITV administration of ranibizumab during and following laser-induced CNV in cynomolgus monkeys were conducted. Study 99-166-1757 evaluated single-agent ranibizumab treatment, while study 00-580-1757 assessed ranibizumab in combination with verteporfin photodynamic therapy (PDT). The findings of these studies point out that ITV administration of ranibizumab during laser induction of a choroidal neovascular membrane and during its subsequent formation do not exhibit any irregular toxicity. Moreover, treatment with ranibizumab in the cynomolgus monkey model prevents membrane formation and attenuates permeability from already-formed choroidal neovascular membranes (31) (Figs. 17.6 and 17.7).
Following ITV administration in normal rabbits and cynomolgus monkeys, ranibizumab was cleared from the vitreous humor with a half-life of approximately 3 days and was distributed to all layers of the retina and cells where VEGF is expressed. In rabbits, a fraction of ranibizumab was cleared from the vitreous humor and through the choroidal capillaries. Concentrations of aqueous humor declined in parallel with those in the vitreous




Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree