Lens-Induced Uveitis and Related Intraocular Inflammation
Narsing A. Rao
Daniel V. Vasconcelos-Santos
The diagnostic category of lens-induced uveitis encompasses three main clinical and pathologic entities, characterized by variable intraocular inflammation that is induced by lens protein.1 The first entity, phacoanaphylactic endophthalmitis, is characterized by severe granulomatous inflammation directed against the lens.2 The second entity, phacogenic nongranulomatous uveitis, previously referred to as phacotoxic uveitis, is a milder, nonspecific, chronic intraocular inflammatory related to lens protein.3 It is possible that these two conditions represent parts of the same disease spectrum.1 The third entity, phacolytic glaucoma, is considered a lens-induced uveitis by some authors but not by others. Patients with phacolytic glaucoma exhibit some features of intraocular inflammation, but histologically the process consists of a macrophage phagocytic response to liquefied proteins released from a cataractous lens.4 The clinical presentations of these three entities overlap to a certain extent: Patients characteristically have some degree of intraocular inflammation manifested as inflammatory cells in the anterior chamber and in the vitreous cavity. These three entities can be differentiated histopathologically, however, largely by the type of cellular infiltration and by the pattern of inflammation around the lens.1
PHACOANAPHYLACTIC ENDOPHTHALMITIS
Phacoanaphylactic endophthalmitis, also known as phacoanaphylactic uveitis, is a granulomatous inflammatory process associated with a disrupted lens.5 This entity was first reported as a toxic lens–induced inflammation by Straub in 1919. In 1922, Verhoeff and Lemoine, who referred to this entity as “endophthalmitis phacoanaphylactica,” proposed an autoimmune response to lens protein as the cause of this granulomatous inflammation.6 Some authors have stated that phacoallergic uveitis may be the preferred term for this entity,7 as the term “anaphylaxis” is usually employed when the immune reaction is mediated by immunoglobulin E.1 Nevertheless, the term phacoanaphylactic endophthalmitis and its distinctive histopathologic features have been consistently incorporated in the ophthalmic literature.
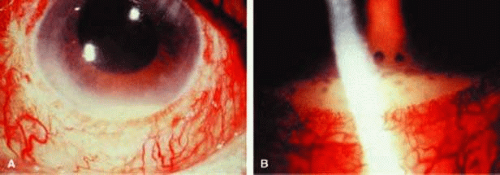
Clinically, phacoanaphylactic endophthalmitis usually follows traumatic or surgical disruption of the lens capsule, but it may also occur after spontaneous capsular rupture.8 The inflammation usually begins within 14 days of lens injury and characteristically presents as a unilateral, severe uveitis in the traumatized eye. Occasionally, this inflammation develops in combination with sympathetic ophthalmia when the lens capsule is disrupted, either by trauma or by intraocular inflammation itself.1 In most cases, the ocular inflammation is severe, with large numbers of cells in the anterior chamber and vitreous cavity. Large keratic precipitates are generally present, and extensive posterior synechiae formation often occurs. Fragments of lens cortex may be observed in the anterior chamber or in the vitreous cavity, and in most cases, the lens material exhibits cataractous changes. In some cases the inflammation is intense enough to cause hypopyon (Fig. 1). If left untreated, inflammation can lead to formation of a cyclitic membrane, tractional retinal detachment, and, eventually, phthisis bulbi.5,9
A milder form of phacoanaphylactic uveitis occurs following extracapsular cataract extraction, with or without posterior chamber intraocular lens implantation.10,11 In these cases the granulomatous inflammation develops between 1 week and 6 months after the surgical procedure. This form of phacoanaphylaxis is believed to reflect limited immune response to a small amount of lens protein remaining in the ocular cavity.10,11 In some patients, a sympathetic response may occur in the opposite eye.
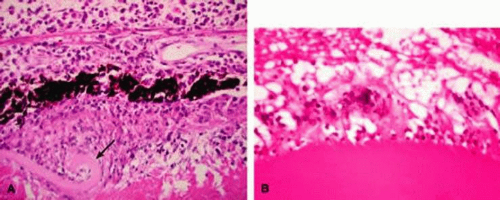
Histopathologic evaluation of enucleated eyes with severe phacoanaphylactic endophthalmitis reveals a zonal type of granulomatous inflammation concentrated around the disrupted lens capsule and the exposed lens cortex or nucleus1 (Fig. 2). Typically, a zone of polymorphonuclear leukocytes is present around the lens cortex and the cortical fibers show degenerative changes in the form of fragmentation and disintegration. Surrounding the polymorphonuclear leukocytes is a zone of mononuclear cells, which consists primarily of macrophages and epithelioid cells but is admixed with a few scattered multinucleated giant cells. Some of these giant cells show features of foreign body type cells, whereas others exhibit features typical of Langhans cells (Fig. 2B). A third zone, made up of lymphocytes, surrounds these epithelioid cells. This lymphocytic zone may also contain fibroblasts or granulation tissue. The surrounding iris, ciliary body, and anterior choroid are usually infiltrated by lymphocytes and plasma cells. This lymphoplasmacytic infiltration is marked in the anterior uvea and tends to be milder in the posterior uvea. The retina may show some perivascular lymphocytic infiltration. In many cases, extensive iridolenticular adhesions are present (Fig. 2A), often accompanied by chronic inflammatory cells in the anterior chamber and in angle structures. The endothelial surface of the cornea may contain collections of mononuclear cells forming keratic precipitates.1,8
The classic zonal pattern may be modified and limited by the size of the capsular rupture and adhesions of iris and ciliary body to the lens.1 Eyes enucleated during more chronic stages of phacoanaphylactic endophthalmitis may show only a few polymorphonuclear leukocytes around the disrupted lens. However, a zone of granulomatous inflammation can be seen, made up of epithelioid cells admixed with many multinucleated giant cells, as can a surrounding zone of lymphocytes and fibroblasts. These eyes may also exhibit fibrosis that encloses the granulomatous inflammatory mass. Such eyes usually show a cyclitic membrane, tractional retinal detachment, osseous metaplasia of the retinal pigment epithelium, and phthisis.
Etiology
Although the precise cause of the initial events that evoke intraocular inflammation in phacoanaphylactic endophthalmitis is unknown, it is believed to be initiated by an autoimmune process directed at the lens protein. Past reports have suggested that lens protein is organ specific and, under normal conditions, is sequestered from the immune system by the lens capsule.12 However, more recent studies have shown that lens crystallin proteins are expressed in the retina, heart, and other sites.13 It also has been documented that most healthy people, with no demonstrable ocular disease, have anti-lens antibodies,14 and when they do undergo extracapsular cataract surgery, they rarely develop phacoanaphylaxis. Based on these observations, it is less likely that phacoanaphylaxis is an immune reaction to the so-called sequestered lens antigen that is released by traumatic or surgical disruption of the lens capsule. Moreover, experimental animal studies have shown that lens proteins are not sequestered solely in the lens capsule and that these proteins are not organ specific. Based on his experimental animal studies, Marak15 proposed a hypothesis for the pathogenesis of phacoanaphylaxis. According to his theory, the development of phacoanaphylactic endophthalmitis is dependent on abrogation of normal tolerance to lens protein.15
Granulomatous inflammation can be experimentally induced around surgically disrupted lens in various strains of rats that have been sensitized to lens protein.15 The enucleated eyes of these animals show histopathologic features very similar to those observed in human phacoanaphylaxis. Immunologic investigations of such animals demonstrated that the lens-induced uveitis is an antibody-mediated autoimmune disease that develops when normal immune tolerance (T-cell tolerance) is terminated by stimulation of B cells with gram-negative bacterial lipopolysaccharide.15 Based on such experimental studies, it appears that phacoanaphylactic endophthalmitis in humans could be an anti-lens protein antibody-mediated localized immune complex disease triggered by traumatic release of lens protein and stimulation of B cells.
Differential Diagnosis
The intraocular inflammatory disorders that should be differentiated from phacoanaphylactic endophthalmitis include infectious (bacterial or fungal) endophthalmitis, phacogenic nongranulomatous uveitis, phacolytic glaucoma, sympathetic ophthalmia, and toxic anterior segment syndrome.5,16 Exogenous acute bacterial endophthalmitis following a penetrating wound to the globe or following cataract extraction may present with signs of severe intraocular inflammation and hypopyon. However, unlike in phacoanaphylactic endophthalmitis, it is unusual to observe large keratic precipitates in acute bacterial endophthalmitis. Some chronic cases of bacterial or fungal endophthalmitis may be difficult to differentiate from phacoanaphylaxis. In such cases, a definitive diagnosis can be established by histologic examination and by culture studies of the intraocular inflammatory exudate. Localized endophthalmitis by Propionibacterium acnes is of special concern and will be addressed later in this chapter.
Exogenous fungal endophthalmitis may present with several clinical features typical of low-grade phacoanaphylactic endophthalmitis. These include delayed onset of ocular inflammation with hypopyon, inflammatory exudate in the anterior chamber and vitreous cavity, and puffball vitreous opacities. Furthermore, some of these patients may exhibit keratic precipitates similar to those noted in phacoanaphylactic endophthalmitis; but cultures and histologic studies of the intraocular exudate should point to the proper diagnosis. Phacoanaphylactic endophthalmitis may also prove difficult to differentiate from phacogenic nongranulomatous uveitis and phacolytic glaucoma, entities that are discussed in detail subsequently in this text.
Phacoanaphylactic endophthalmitis occurs typically as a uniocular inflammatory process involving the traumatized eye but can present occasionally as bilateral intraocular inflammation.5 Such bilateral, lens-induced intraocular inflammation must be differentiated from sympathetic ophthalmia, which is invariably a bilateral disease. Patients with sympathetic ophthalmia rarely develop hypopyon. Moreover, sympathetic ophthalmia typically results in angiographic findings of hyperfluorescence and focal leakage at the level of the retinal pigment epithelium during the early arteriovenous phase, with pooling of dye in the subretinal space during late stages of angiography. Echographic evaluation may also be useful because phacoanaphylactic endophthalmitis causes inflammatory thickening of the anterior uvea, whereas sympathetic ophthalmia induces marked thickening of the posterior uvea. It should be noted that in some cases of penetrating injury to the globe, phacoanaphylactic endophthalmitis and sympathetic ophthalmia may coexist in one eye, and the opposite eye may show only features of sympathetic ophthalmia.1,5
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree