Fig. 4.1
AS-OCT demonstrating a subluxed crystalline lens causing angle closure
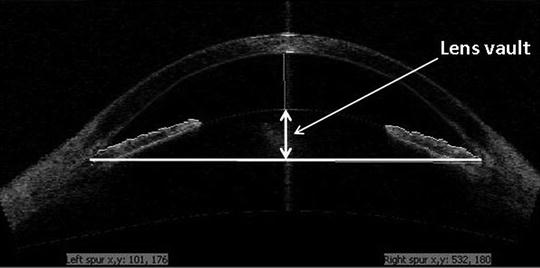
Fig. 4.2
AS-OCT depicting anterior lens vault which may contribute to angle crowding
4.3 Reasons for Lens Extraction in Angle Closure Glaucoma
4.3.1 Evidence for Lens Extraction
Anatomical risk factors associated with PACG are primarily due to a crowded anterior chamber with iridotrabecular contact. Both can be relieved by lens extraction. A number of studies have advocated lens extraction in PACG, with the primary outcome measure being a sustained reduction in IOP. It has been postulated that the anterior chamber shallows with age due to an increase in lens thickness [7]. Consequently, deepening of the anterior chamber achieved by lens extraction may improve aqueous access to the angle, and therefore reduce the IOP [7] (Figs. 4.3a, b). Weak evidence exists as to whether patients without glaucoma receiving cataract surgery achieve any reduction in IOP. One study of 93 patients found an average of 1.5 mm reduction at 3 years [7]. IOP reductions of between 2–6 mmHg have been described following lens extraction in PACG eyes [8–11], notably greater in PACG compared to POAG eyes [12].
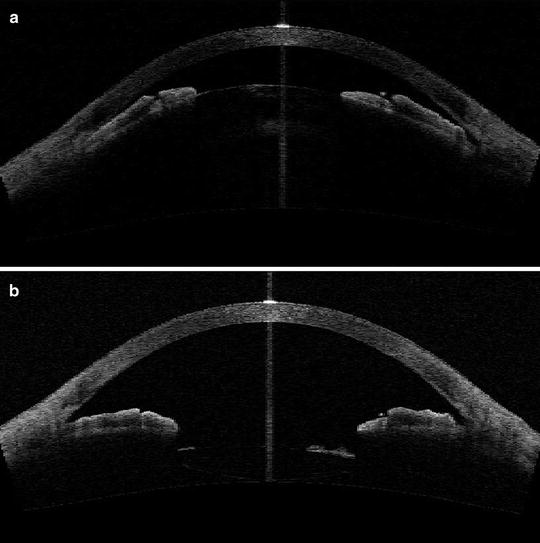

Fig. 4.3
(a) AS-OCT before phacoemulsification surgery showing a shallow anterior chamber and angle closure due to the presence of a cataract with large lens vault. (b) AS-OCT of the same patient as in Fig. 4.3a after phacoemulsification surgery showing deepened anterior chamber with open angles
A recent randomized, controlled trial evaluated laser peripheral iridotomy (LPI) versus lens extraction in patients with acute primary angle closure (APAC), following emergency medical management to lower the IOP. Compared to the LPI group, the group receiving phacoemulsification demonstrated a greater IOP fall. Furthermore, when performed within 1 week of an acute angle closure attack, phacoemulsification was associated with reduced failure of IOP control at 2 years when compared to LPI [13]. A study performed by Lam et al. randomized 62 eyes to LPI or phaco/IOL after medical control of APAC and demonstrated a mean IOP of 12.6 mmHg in the phaco group versus 15.0 mmHg in the LPI group, after 18 months of follow-up [14].
Whilst conventional LPI may open the drainage angle and reduce the IOP initially in APAC, a retrospective review of 65 subjects who had LPI followed up for 5 years revealed that 100 % required medications and 63 % surgery to lower the IOP [15]. Important mechanistic aspects of ACG, such as pupil block are relieved by lens extraction, reducing the risk of CACG. A case series of 21 eyes reported a 19 % reduction in PAS greater than 270° [16] and another purported that the degree of lens vault may be a positive predictor for IOP reduction and degree of angle widening following surgery [17].
Tham et al. [18, 19] performed two randomized studies comparing phacoemulsification alone to combined phaco-trabeculectomy in medically controlled and uncontrolled chronic PACG. They assessed a number of factors including IOP control, post-operative complications, and the number of glaucoma drops. Combined phaco-trabeculectomy was associated with greater complications, with some necessitating additional surgery. The difference in glaucoma progression was not significant in both groups [19]. Interestingly improved IOP control in the phaco-trabeculectomy group was only marginal when compared to the phacoemulsification group across the 24 months of the study.
Whilst combined phaco-trabeculectomy surgery does offer a small benefit in terms of IOP control, there is little evidence to suggest that this improvement is associated with better long-term outcomes in terms of visual field progression. Therefore, primary lens extraction may be a viable option in the management of angle closure glaucoma. In many circumstances, lens extraction may suffice for glaucoma control—only 2.9 % of patients with controlled PACG versus 14.8 % in uncontrolled PACG receiving lens extraction alone went on to require a trabeculectomy [18, 19].
4.3.2 Additional Considerations for Cataract Surgery in PACG
The patient pathway for conventional phacoemulsification surgery is well established. With infrequent post-operative visits compared to glaucoma filtration surgery, this option may appear more attractive to patients, particularly when the nature, demographics, and geographical setup of the clinical setting are considered. In a scenario where frequent and long-term patient follow-up may be difficult, the option of lens extraction may be favoured above other treatment modalities.
Furthermore, fluctuations in visual performance tend to be less frequent following cataract surgery when compared to glaucoma surgery, the latter often resulting in variations in IOP and blurred vision, which can persist for several weeks. Ocular surface discomfort and inflammation is less pronounced following phacoemulsification surgery [20], and studies have demonstrated improved patient satisfaction following this procedure [20, 21]. Glaucoma filtration surgery can potentially expose patients to long-term risks, including blebitis and bleb-related endophthalmitis which are circumvented by the choice of cataract surgery.
4.3.3 Combined Procedures
4.3.3.1 Lens Extraction with Goniosynechialysis/Viscogonioplasty
Whilst cataract surgery alone may deepen the anterior chamber and open the drainage angle, its effect on the presence of significant peripheral anterior synechiae (PAS) may be limited. PAS formation has been shown to occur even with the presence of a patent PI [22]. In recent years, further evidence has emerged to incorporate goniosynechialysis (GSL) with cataract surgery [23]. In this procedure, PAS are stripped manually under gonioscopic view. This can be performed either directly, using a blunt spatula, or via the application of viscoelastic (also known as viscogonioplasty, VGP), delicately avoiding potential complications such as bleeding, iridodialysis, and fibrinous uveitis. GSL is thought to be most effective in patients where PAS has been present for less than 6–12 months [24].
Whilst the results of randomized controlled trials are awaited, there was a recent cohort study of 51 eyes (17 with APAC and 34 with chronic angle closure glaucoma) that underwent phacoemulsification surgery and GSL. At an average of 25 months follow-up, the mean IOP dropped by an average of 9.67 mmHg for both groups. The average number of medications used fell from 2.51 preoperatively to 0.67 post-operatively [25]. The GSL technique can contribute to deepening the anterior chamber angle [25]. Two retrospective case series have described the IOP effect of phaco-VGP. Varma et al. described, in 15 eyes of patients with refractory PACG, a mean IOP reduction from 27.4 mmHg (on medication) to 14.1 mmHg (off all medications in 14/15 eyes) at 6 months [26]. Another study, in 2008, of 11 patients (11 eyes) with PACG (all of whom had refractory control despite patent LPI) showed a reduction in IOP from 39.4 mmHg preoperatively to a mean of 13.4 mmHg after phaco-VGP at 7.8 months follow-up [27]. Intraoperative complications such as haemorrhage and iridocyclodialysis, previously described with GSL, were not observed in this series.
4.3.3.2 Phaco-Trabeculectomy
A combination of phacoemulsification surgery with trabeculectomy surgery with mitomycin-C should be considered in patients with cataract and advanced PACG or those with glaucomatous progression, with suboptimal IOP control, or those with extensive PAS. One benefit of this approach is that, in most cases, this will result in one visit to the operating room. However, trabeculectomy function may be compromised if cataract surgery is complicated at the same sitting. Post-operative “spikes” in IOP are generally avoided with the combined procedure, and this may be advantageous particularly in advanced glaucoma to reduce the risk of “wipe out”. Tham et al. [19] have demonstrated lower mean IOP and medication use after phaco-trabeculectomy compared to phacoemulsification alone in medically uncontrolled CACG patients. However, the IOP findings were only significant at a few time points, and the increase in post-operative complications in the trabeculectomy group were due to the trabeculectomy part of the procedure [18]. Currently, the long-term results of phacoemulsification alone versus phaco-trabeculectomy are not known.
4.3.4 The Timing of Lens Extraction
Controversy surrounds the optimal timing of lens extraction in both acute and chronic PACG. Whilst some surgeons suggest primary lens extraction days after the resolution of the acute angle closure episode, surgery may be technically less challenging and safer once the corneal clarity and intraocular inflammation have settled after a few weeks. A study by Imaizumi on 18 eyes undergoing cataract surgery within 1 day of medical control of APAC demonstrated no difference in the endothelial cell count compared to a control group of conventional phacoemulsification in normal eyes [11]. Whilst the largest IOP reductions have been observed in medically uncontrolled APAC eyes undergoing phacoemulsification [28], Jacobi et al. demonstrated significantly better IOP control and reduced use of ocular hypotensive medications, at 10 months follow-up, in patients receiving phacoemulsification surgery within 4 days of APAC versus conventional surgical iridectomy [29]. Improved long-term results may be best if surgery is performed before PAS has had a chance to fully form. However, in cases where access to surgery is limited, preparative LPI may be a more viable option.
It has been suggested that 50 % of PACG patients are likely to go on and develop cataracts due to ageing and the effects of glaucoma treatment, and the latter in fact may accelerate cataract progression [30]. However, by the time the cataracts have formed, glaucoma progression may be severe and irreversible. Higher rates of posterior capsule rupture have been associated with dense cataracts, suggesting surgeons may wish to intervene before this stage [31].
The exact timing of intervention will involve planning between the surgeon and patient. A detailed ocular examination, patient demographics, assessing visual symptoms, angle morphology (with the degree of appositional and synechial closure), along with IOP, and importantly, the rate of glaucoma progression, should determine when to consider phacoemulsification. In the presence of mild glaucoma and minimal PAS, phacoemulsification alone is preferred, whereas if extensive and relatively recent PAS is present, cataract surgery may be augmented by GSL or VGP. A cataract/filtration procedure (phaco-trabeculectomy) can be reserved for advanced glaucoma cases in which greater IOP lowering may be required.
4.4 Preparing for Surgery
4.4.1 Cataract Extraction in Angle Closure Glaucoma: Potential Risks
Phacoemulsification is not without its risks: Johnston et al. published an overall posterior capsule rupture rate of 1.92 % in the UK national cataract database of 55,297 cataract operations [32]. The risk of endophthalmitis varies between 0.014 and 0.048 % [33]. Risks associated with surgery may correlate with extreme refractive status, with a higher rate of complications associated with higher ametropia. Lens subluxation may be a precipitating factor for angle closure glaucoma in high myopes, whereas shallow anterior chamber depth may be a causative factor in high hyperopes.
Lens extraction in cases of angle closure glaucoma presents potential additional factors to consider aside from routine phacoemulsification surgery. Significant post-operative complications have been reported. Technically, lens extraction post APAC may pose challenges involving a mid-dilated pupil. Iris atrophy and atonicity may increase the risk of iris prolapse, trauma, and bleeding. The eye itself may still be inflamed and the corneal clarity compromised, therefore obscuring the view. With APAC, inflammation may lead to zonular weakness and lens instability [13]. This may have secondary effects upon fluidic management and anterior chamber stability. In PACG, the lens itself may be bulky and cataractous, increasing the risk of sudden drop in IOP upon removal, and subsequent risk of suprachoroidal haemorrhage. Furthermore, the presence of a shallow anterior chamber may exacerbate endothelial damage, retard visual recovery, and occasionally lead to corneal decompensation. Additional risks in these PACG eyes include malignant glaucoma and post-operative IOP elevation, as well as prolonged post-operative inflammation [9]. Via risk stratification, these cases are therefore best reserved for more experienced surgeons.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


