Subglottic Stenosis
Subglottic stenosis in the neonate is defined as a lumen 4.0 mm in diameter or less at the level of the cricoid. A relevant reference point is the outer diameter of an endotracheal tube. For example, a 2.5 endotracheal tube has an outer diameter of 3.6 mm, and a 3.0 endotracheal tube has an outer diameter of 4.2 mm.
It is important to differentiate congenital from acquired subglottic stenosis. Acquired subglottic stenosis is caused by intubation or other forms of laryngeal trauma; this is often an iatrogenic injury caused by a complication of medical therapy. Acquired subglottic stenosis is generally more severe, requiring aggressive, long-term management (discussed later in this chapter). In the absence of trauma, an abnormality of the cartilage or subglottic tissues is usually considered to be congenital. The cause of congenital subglottic stenosis is thought to be a failure of the laryngeal lumen to recanalize (
3). This abnormality lies on a continuum of embryologic failures that includes laryngeal atresia, stenosis, and webs. In its mildest form, congenital subglottic stenosis manifests with a normal-appearing cricoid with a smaller-than-average diameter, usually with an elliptical shape.
A mild case of subglottic stenosis may produce a clinical picture of recurrent upper respiratory infections, often diagnosed as croup, in which minimal subglottic swelling precipitates airway obstruction. The greatest obstruction is usually 2 to 3 mm below the true vocal folds in a young child.
More severe cases of congenital subglottic stenosis may present with acute compromise of the airway at delivery. If endotracheal intubation is successful, the patient may require intervention before extubation. In more severe cases, when intubation cannot be achieved, a tracheotomy may be lifesaving at the time of delivery. Interestingly, however, infants with congenital subglottic stenosis may have surprisingly few symptoms, and even children with grade III subglottic stenosis may not present for weeks or months.
Congenital subglottic stenosis is often associated with other congenital head and neck lesions and syndromes (e.g., a small larynx in Down syndrome). After the initial management of congenital subglottic stenosis, the larynx will grow with the patient and may not require further surgical intervention.
Congenital subglottic stenosis can be divided histopathologically into membranous and cartilaginous types (
1,
2) (
Table 90.2). The membranous type is usually
circumferential and presents as a fibrous soft tissue thickening in the subglottis caused by increased fibrous connective tissue or hyperplastic mucous glands. It usually includes the area 2 to 3 mm below the true vocal folds, but it may extend upward to include the true cords. The cartilaginous type commonly presents as a thickening or deformity of the cricoid cartilage that creates a shelf-like plate of cartilage on the inner surface of the cricoid ring, extending posteriorly as a solid sheet and leaving only a small posterior opening (
1). Stenosis may also be caused by a trapped first tracheal ring.
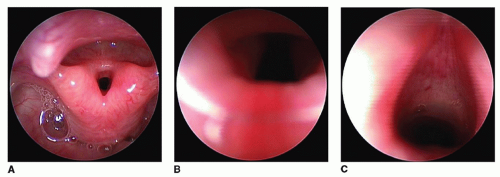
Although the histopathologic picture may vary, the diagnosis of subglottic stenosis is based on endoscopic assessment. The stenosis due to a scar, granulation tissue, submucosal thickening, or a congenitally abnormal cricoid can be differentiated from subglottic stenosis with a normal cricoid, but endoscopic measurement with endotracheal tubes or bronchoscopes is required for an accurate evaluation. Many laryngeal stenoses represent a combination of a congenitally small larynx further traumatized by endolaryngeal intubation or instrumentation.
Radiographic evaluation of a patient’s unintubated airway may give the clinician clues about the site and length of the stenosis. Useful imaging modalities include inspiratory and expiratory lateral soft tissue neck films; fluoroscopy to demonstrate the dynamics of the trachea and larynx; and a chest radiograph. The single most important investigation, however, remains high-kilovoltage airway films. These films are helpful in identifying the classic steepling seen with subglottic stenosis and also in providing a warning of tracheal stenosis; this is usually caused by complete tracheal rings, which may place the child in a life-threatening situation during rigid endoscopy.
Endoscopy is necessary for the diagnosis of laryngeal stenosis. Flexible fiberoptic endoscopy provides information on dynamic vocal fold function. Rigid endoscopy with Hopkins rod lens telescopes provides the best possible examination. Precise measurement of the endolarynx and staging of the laryngeal stenosis can be carried out. The most commonly used system for staging subglottic stenosis is the Myer-Cotton grading scale (
Table 90.3). Most grade I and many grade II lesions do not require surgical intervention; however, grade III and grade IV lesions generally necessitate a surgical approach.