Lacrimal Drainage System
Myron Tanenbaum
Clinton D. McCord Jr.
A clinician’s approach to lacrimal disorders should be logical and organized. Just as neurologic disorders must be carefully localized, so too should lacrimal problems be correctly localized and diagnosed before treatment is implemented. A thorough evaluation of the lacrimal drainage system should begin with the eyes, eyelids, and puncta and terminate with the distal nasolacrimal duct and intranasal passages. Haphazard trial-and-error therapies are to be avoided.
This chapter is organized into basic sections on anatomy, physiology, pathology/pathophysiology, diagnosis, and treatment. The content of these sections emphasizes clinically relevant material to aid in understanding, diagnosing, and treating lacrimal disorders.
“Watery eyes” are among the most common lacrimal symptoms. Patients with this symptom have one of two problems (or both): Either they produce too many tears (hypersecretion) or the tears that are produced cannot properly drain (epiphora). Through the understanding of relevant lacrimal anatomy, physiology, pathology/pathophysiology, and diagnostic techniques (none more important than history), a clinician can make such very basic and very important distinctions in patients with watery eyes. The material presented in this chapter thoroughly addresses disorders of the lacrimal drainage system, such as the watery eyes example, and offers clinicians a logical, orderly approach.
Anatomy
An understanding of the anatomic elements of the lacrimal drainage system is necessary to appreciate the operation of the system and how different diseases can affect its function. These elements include the bony conduit, the membranous conduit; and the surrounding soft tissues, tendons, and muscles.
Bony Conduit
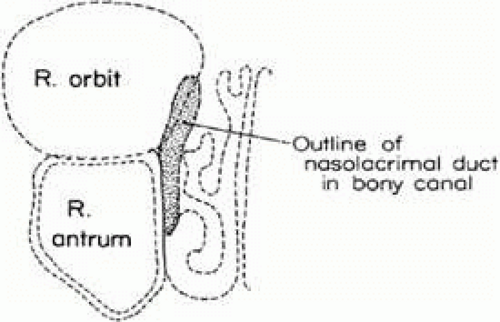
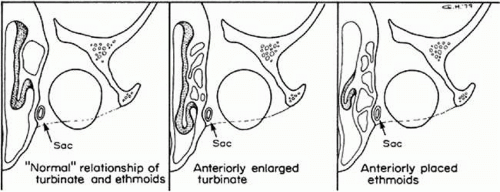
Within the nasofrontal process of the maxilla at the anterior nasal portion of the orbit is the lacrimal fossa, in which is found the lacrimal sac. From there, the nasolacrimal canal, which contains the nasolacrimal duct, extends down inside the lateral wall of the nose and opens under the inferior turbinate or concha in the vault of the meatus of the turbinate. The nasolacrimal canal is approximately 16 mm posterior from the anterior tip of the inferior turbinate and is located between the anterior third and the middle third of the meatus of the turbinate. It exits in the vault of the meatus of the turbinate 17 mm above the floor of the nose laterally. There are, however, normal variations in which the end of the nasolacrimal canal can extend farther down and open at various positions in the lateral wall of the nose. The lacrimal sac fossa is present in the anterior–inferior nasal portion of the bony orbit and is delineated anteriorly by the anterior lacrimal crest and posteriorly by the posterior lacrimal crest. If viewed from the inside of the nose, it would be outlined on the lateral wall of the nose in front of the anterior tip of the middle turbinate (Fig. 13.1). The nasolacrimal canal is not vertical in direction but angulates posteriorly 15 degrees and slightly inward almost 5 degrees to reach its final destination under the inferior turbinate (Fig. 13.2). Two sinuses are in intimate relationship with the lacrimal bony conduit, the maxillary sinus or antrum, which forms the lateral wall of the nose in that area, and the ethmoidal sinus, which borders the posterior edge of the lacrimal sac fossa and the superior nasolacrimal duct. The ethmoidal sinus is a system of mucous membrane-lined air cells that are positioned immediately posterior to the lacrimal sac fossa and the lacrimal bone. It is not uncommon (as many as 80%–90% of patients)1; however, for the ethmoidal air cells to encroach in various degrees into the bone at the posterior lacrimal crest; in some cases, they actually extend as far as the anterior lacrimal crest area separating the lacrimal sac from the lateral wall of the nose and intranasal cavity (Fig. 13.3). Also, the anterior tip of the middle turbinate may extend various degrees anteriorly into the intranasal cavity. These anatomic variations become important considerations when lacrimal surgical procedures are necessary.
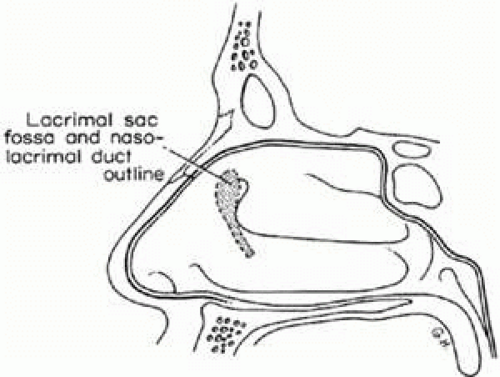 FIGURE 13.1 Outline of the lacrimal sac fossa and nasolacrimal duct in its course under the inferior turbinate as viewed inside the nose looking at the lateral wall of the nose. |
Membranous Conduit
Dimensions
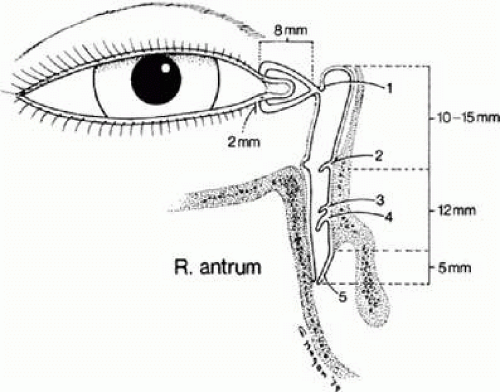
Contained within the bony conduit and the nasal portion of the eyelids are the epithelium-lined tear ducts through which tears pass from the eyelids into the nose. The openings of this membranous conduit in the eyelids are the upper and lower puncta, which are located 6 mm from the inner canthus. The punctal openings in the eyelid are 0.3 mm in diameter. The canalicular portion just beyond the punctum is called the ampulla. It is vertically oriented for 2 mm and balloons out to a diameter of 2.5 mm (Fig. 13.4). The canaliculi then narrow to about 1 mm in diameter and extend nasally 8 mm to enter the lacrimal sac. In more than 90% of people, the canaliculi join before entering the sac at the level of the lower border of the medial canthal tendon. The tear sac itself is 12 to 15 mm in vertical length, and in most instances there is a portion above the entrance of the common canaliculus 3 to 4 mm in height called the fundus (dome), which is usually compressed by the medial canthal tendon. The nasolacrimal duct extends from the inferior portion of the sac through the nasolacrimal canal or bony conduit and travels 12 mm to the position where it opens underneath the inferior turbinate in a space called the meatus (see Fig. 13.4). The membranous conduit in most cases extends 5 mm farther downward and usually is located in the vault portion underneath the inferior turbinate. However, again, there is some variability in the nasolacrimal canal and duct, and it may open farther down on the lateral wall of the nose in a slitlike manner (Fig. 13.5).
Structure
The lining of the lacrimal canaliculi, the sac, and the nasolacrimal duct is pseudostratified columnar epithelium similar to that found in the upper respiratory system. The walls contain much elastic tissue. The canaliculi, in particular, contain large amounts of elastic tissue. In addition, the lacrimal sac and the nasolacrimal duct have collagen, elastic fibers, and amounts of lymphoid tissue in the walls.2,3 The mucous membrane within the sac and nasolacrimal duct is arranged into membranous folds that act as valves. The two most important folds, as best as can be determined clinically, are the valve of Rosenmuller, where the common canaliculus enters into the sac, and the valve of Hasner (plica lacrimalis) at the end of the nasolacrimal duct under the inferior turbinate. Other valvelike folds have been identified, as have valvelike constrictions at the junction of the sac in the nasolacrimal duct (valve of Krause, sinus of Arlt) and within the nasolacrimal duct (spiral valve of Hyrtl and the valve of Taillefer; see Fig. 13.4).
Encompassing Soft Tissue
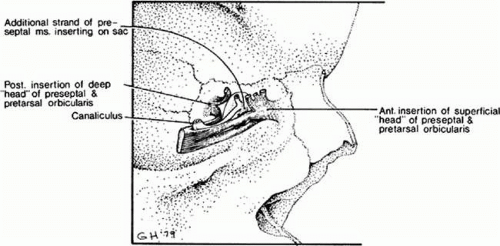
The muscles within the eyelid in front of the tarsus (pretarsal orbicularis), the protractor muscles, are anchored at the lateral canthal tendon and the lateral palpebral raphe. They travel horizontally across the surface of the upper and lower tarsus, and as they reach the medial canthal area they split into a superficial and deep portion or head. The deep head of the pretarsal orbicularis muscle from the upper and lower lid inserts on the lacrimal bone at the posterior lacrimal crest behind the sac and in some cases has been referred to as Horner’s muscle. An additional strand of orbicularis muscle from the preseptal area in the lower lid inserts on the periosteum, which covers the lacrimal sac and its fossa, which extends from the posterior to the anterior lacrimal crest described by Jones.4 The superficial head of the pretarsal orbicularis and preseptal orbicularis fibers insert in a dense conjoined medial canthal tendon anterior to the fundus of the lacrimal sac (Fig. 13.6).
Physiology of Lacrimal Elimination
Much has been written and hypothesized about the mechanism of lacrimal elimination. The abundance of theories and conflicting information in the literature often serves to dissuade practicing clinicians from investigating this topic. A basic understanding of lacrimal physiology, however, is a useful foundation for making treatment decisions about many lacrimal disorders.
The tear film travels across the surface of the globe and eyelids, enters the puncta/ampulla, passes through the canaliculi, and enters the lacrimal sac/nasolacrimal duct/nasal passages. This schema is of course oversimplified, because much of the tear film is likely “eliminated” by direct evaporation or absorption at the level of the lacrimal sac. The first key physiologic point to understand, however, is that the lacrimal outflow system is based on an active, dynamic pumping mechanism. It has long been noted that the blinking mechanism readily drains tears even with the head held in an inverted position. When the palpebral blink mechanism is impaired, however, epiphora is common, such as in patients with facial paralysis.
Although multiple mechanisms may contribute to lacrimal outflow, present evidence suggests that the most important factor is the active palpebral-canalicular pump. This theory is based on the cumulative works of Doane,5 Rosengren,6 Frieberg,7 Chavis et al.,8 and Maurice9; and an analysis of the blink mechanism with high-speed photography. Carbon particles were used to photograph the actual flow of the precorneal tear meniscus. The important findings of this work are summarized as follows. With the eyelids open before the start of a blink, the canaliculi are already filled with tears. As the upper lid descends at the start of a blink, the medial portion of the eyelids around the puncta elevates. The upper and lower puncta come into forceful contact by the time the eyelids are only halfway closed. This important event occludes the puncta such that the remaining portion of the blink acts to compress the canaliculi and lacrimal sac, thus forcing the contained fluid into the nasolacrimal ducts and nasal passages. The volume of fluid within the lacrimal outflow system is at its minimum at the point of maximum lid closure during a blink. As the eyelids begin to open, the muscular compressive force terminates and the elastic walls of the canaliculi and lacrimal sac attempt to restore their original shape. The puncta remain occluded such that a partial vacuum forms within the membranous lacrimal conduit. As the eyelid-opening phase of the blink continues, the two lacrimal puncta pop open and expose the adjacent lacrimal lake to this partial vacuum. Tears rapidly flow into the canaliculi during the 1- to 3-second interval immediately after the blink. Once again, the canaliculi fill with fluid so that the pumping action of the next blink can continue the lacrimal elimination cycle.
In addition to the important role of the active palpebral-canalicular pump, other factors contributing to lacrimal elimination may include physical forces such as gravity and capillary attraction of the tears, reservoir drainage into the lacrimal sac (so-called Krehbiel flow), microcilliation in the nasolacrimal duct, and finally evaporation of tears from the ocular surface and absorption of tears by the lacrimal sac mucosa.
The “lacrimal pump” theory is widely noted in the ophthalmic literature and is based on classic anatomic studies by Jones,10 describing tendinous and muscular insertions exerting their action on and around the lacrimal sac. The Jones theory holds that with closure of the eyelid margin, the eyelid fissure moves nasally and forces tears toward the area of the puncta and interface between the lids, conjunctiva, and caruncle in the area of the lacrimal lake. With relaxation of the eyelids on opening, the canaliculi and ampulla, which have been compressed by the pretarsal deep and superficial muscles because of their elasticity, create a negative pressure in the ampulla-canalicular system, causing tears to be sucked into the puncta. When the eyelids are closed again, the tears, which previously entered the ampulla-canalicular system when the eyelids were opened, are squeezed into the lacrimal sac. It is also theorized that the muscular pull of the preseptal orbicularis muscles on the lacrimal sac creates a negative pressure within the sac. With the eyelids open, the sac is normally collapsed. The valves within the sac and nasolacrimal duct prevent retrograde passage of tear flow in normal situations. This proposed lacrimal sac pumping mechanism is based on anatomic studies and likely does not have a large role in normal lacrimal elimination, because the system functions quite well with the lacrimal sac slit completely open, as is the case after dacryocystorhinostomy (DCR).
Traditional teachings have previously held that the lower canalicular drainage system was far more important than the upper system. This old wives’ tale is completely incorrect. Studies by White et al.11 and Daubert et al.12 have demonstrated equal tear flow between the upper and lower canalicular systems using radioactive dacryoscintigraphy flow studies. Linberg and Moore13 evaluated the clinical symptoms associated with alternate monocanalicular occlusion of the upper and lower puncta. They found that approximately 50% of patients experience mild intermittent symptoms of epiphora associated with experimental monocanalicular obstruction. The symptoms were identical whether patients’ upper canalicular system or lower canalicular system was occluded. Meyer et al.14 studied fluorescein dye disappearance in 20 subjects and found that 90% of patients showed minimal or no impairment with monocanalicular (either upper or lower) obstruction.
A number of important clinical principles can be derived from the physiologic information just provided. Present evidence supports the crucial role of the palpebral-canalicular pump mechanism in lacrimal elimination. All efforts should be made to preserve the lacrimal canaliculi. Repeated instrumentation of the lacrimal system or nasolacrimal duct probings are unlikely to help the underlying pathology and may in and of themselves injure the canaliculi and thus permanently impair lacrimal elimination. Very little can be done to restore scarred fibrosed canaliculi. Frieberg has used a manometer to measure a pressure gradient within the canaliculi and lacrimal sac.7 This pressure gradient cannot be produced if the canaliculus is slit open. This information should caution clinicians against performing overly aggressive procedures on the lacrimal outflow system, such as aggressive “three-snip” punctoplasties or aggressive surgical treatment of canaliculitis. Experimental and clinical studies show that tear elimination is equivalent through the upper and lower canalicular systems.11,12,13,14 Surgeons should thus give equal consideration to a patient with lacerations of either the upper or lower canaliculus. Traditional teachings that upper eyelid canalicular lacerations are unimportant are simply not true.
Embryology
The anlage of the membranous lacrimal conduit is an ectodermal thickening of a groove between the lateral, nasal, and maxillary process in the 12-mm-stage embryo. After this stage, the anlage detaches and becomes buried in the mesoderm. Solid cords of epithelial cells form the anlage of the canalicular system in the eyelids, with a projection downward that will form the nasolacrimal sac and the nasolacrimal duct at the 16-mm stage. Canalization of these epithelial cords starts at the 50-mm stage, or 4 months of gestation, beginning as scattered patches throughout the system and creating a lumen through the system. This lumen finally breaks through its latest stage in the nasolacrimal duct to form a continuous opening just before birth. The lower end of the lacrimal duct is the last to canalize, and in more than half of infants the last portion of this nasolacrimal stem may not completely finalize its patency at birth.15,16 During embryonic development, migrations of epithelial cords can cause various anomalies within the lacrimal system.
Congenital Anomalies
Dacryostenosis
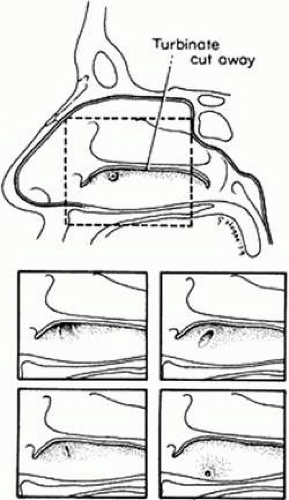
Dacryostenosis is a common condition in which the extreme end of the nasolacrimal duct underneath the inferior turbinate fails to complete its canalization in the newborn period; it produces clinical symptoms in 2% to 4% of newborns. In most instances, the obstruction is a small membrane at the end of the nasolacrimal duct that persists because of failure of complete canalization of the nasolacrimal duct. Other rarer types of congenital occlusion of the nasolacrimal duct and canal may occur if the epithelial cords have migrated within the mesoderm and do not open immediately under the inferior turbinate. Blind pouches can occur within the turbinate itself. A bony obstruction is often found under the inferior turbinate, and in some cases the epithelial cord migrates laterally so that the nasolacrimal canal ends in a blind pouch in the lateral wall of the nose.17,18 There are normal variations in the position of the opening of the nasolacrimal duct under the inferior turbinate. The nasal ostium of the duct, at the highest portion of the inferior meatus within the vault beneath the inferior turbinate, is usually wide open. It may be puckered, slitlike, or grooved down the lateral wall of the nose or have a puncturelike appearance in the vault of the turbinate (see Fig. 13.5).
Absence of Valves
The fold normally present at the end of the nasolacrimal duct or valve of Hasner may be absent, in which case pneumatoceles of the sac may occur and nose blowing may cause retrograde passage of air. If the valve of Rosenmuller is also absent, it is possible to blow air from the nose into the eye, and nosebleeds may produce bloody tears.19
Anomalies of the Sac
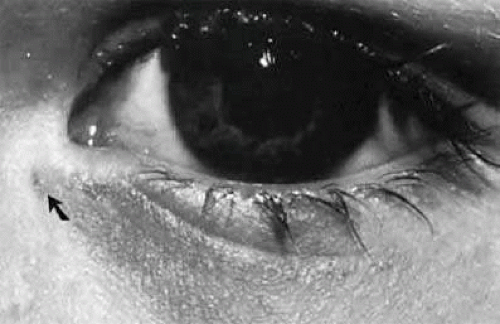
Although diverticuli of the lacrimal sac may occur, a congenital fistula of the lacrimal sac, which has been termed lacrimal anlage duct by Jones, is more common (Fig. 13.7).19 This anomaly has been described by others and in some cases is found to be autosomal dominant and may coexist with thalassemia. It undoubtedly is the result of canalization of a strand of epithelial cords that extends from the sac to the skin surface. The fistulas often have to be completely excised to prevent drainage of tears externally on the skin.
Anomalies of the Puncta
Congenital atresia, supernumerary or double puncta, and congenital slits of the puncta all may occur from aberrations in the location of the epithelial cord and its opening to the surface epithelium. Lateral displacement of the puncta may occur in some congenital syndromes such as blepharophimosis.
Anomalies of the Canaliculi
Atresia or failure of canalization of the lacrimal canaliculi may occur in conjunction with punctal atresia. In many cases, particularly in patients with mesodermal dysplasia, the lacrimal canaliculi and puncta may be absent, and a normal tear sac and nasolacrimal duct may be present but not connected to the eyelid surface.
Lacrimal Problems in Neonates
Neonates have tear secretion at birth, and 96% to 98% have a totally patent and functional lacrimal drainage system.20 The 2% to 4% who do not have a lacrimal drainage system intact have a thin residual membrane at the distal end of the nasolacrimal duct. This membrane spontaneously dissolves in 80% to 90% of patients within the first few months of life.21 Clinical manifestations of congenital nasolacrimal duct obstruction are the following.
Amniotocele
This occurs in neonates as a distention in the lacrimal sac. Amniotic fluid enters the sac, is retained by a nonpatent nasolacrimal duct, and is trapped in the sac by the valve at the common canaliculus, the valve of Rosenmuller. Probing the nasolacrimal duct as an office procedure is usually curative.
Dacryocystitis (Acute Mucocele or Pyocele)
This condition also exhibits acute distention and inflammation in the lacrimal sac region and may occur in the neonatal period. Probing is necessary in newborns with acute dacryocystitis to establish drainage as soon as possible. This procedure is performed with topical local anesthesia only.
Tearing and Mattering
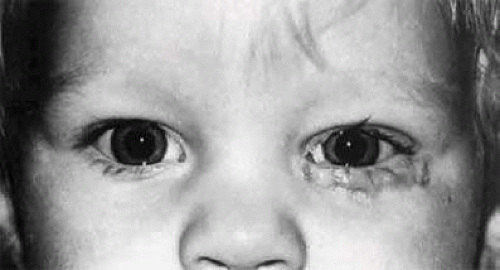
Newborns who have congenital dacryostenosis may not develop acute dacryocystitis with a mucocele or pyocele of the sac in the early neonatal period but may simply have tearing with a chronic mucopurulent discharge, which usually becomes manifest at 2 weeks of age. Topical antibiotics should be administered, and the parents must be instructed in the proper technique of lacrimal sac compression and massage. More than 90% of these cases clear and become asymptomatic with conservative management.22,23 In normal circumstances, these children with mild to moderate symptoms of epiphora and lid crusting can be monitored for the first year of life without serious consequence or sequela (Fig. 13.8). There is rarely any imperative reason to make probing mandatory at an early age (e.g., before 6 months of age). A number of studies have confirmed that probing or silicone tube intubation in children after 12 months of age still has a very high success rate. These techniques are discussed later in the Treatment section in this chapter.
Diseases of the Puncta
Malposition or Eversion
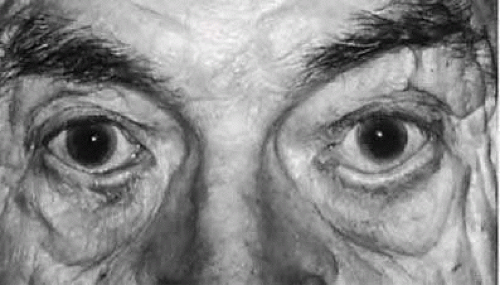
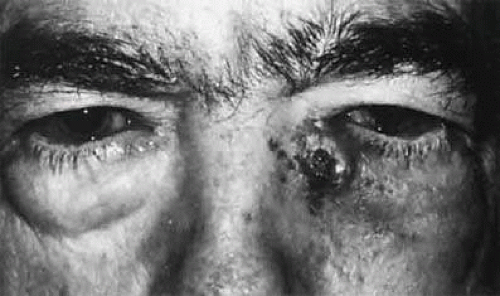
The normal position of the punctum is pointing inward toward the lacrimal lake. It cannot be seen by an observer looking directly toward the eyelid. The punctum can be displaced in a congenital anomaly with eyelid syndromes such as blepharophimosis or congenital ectropion. Malposition more commonly is an acquired anomaly in older individuals with eyelid laxity and senile ectropion (Fig. 13.9). It also may occur after injury or burns with skin shrinkage, which results in turning the punctum outward.
Atresia
Isolated stenosis of the punctum with an intact and patent canalicular system distally may occur congenitally or as a result of cicatrizing, and senile atrophy associated with long-term drying in patients with ectropion. In some patients, the punctum (or puncta) are tightly cicatrized closed. In other patients, the puncta are erythematous, edematous, and functionally swollen closed. It is important to distinguish these differences.
Punctal stenosis is a very common, and commonly overlooked cause of epiphora.
Tumors
Basal cell carcinomas occur most frequently in the inner canthal inflammations, trauma area and often involve the punctum, as may benign growths in this area.
Disorders of the Canaliculi
Obstruction
Obstruction or atresia of the canalicular system may follow any of the conditions discussed next.
Cicatrizing Conjunctivitis
Trauma
Chemical or thermal burns, dog bites, and other lacerations may also cause obstruction or atresia of the canaliculi. At one time or another, every ophthalmologist is called on to repair a canalicular laceration. Acute lacerations of the canaliculi may occur after sharp penetrating wounds or as a result of shearing or ripping wounds of the eyelid caused by hooklike objects or teeth. The location of the lid laceration medial to the lacrimal punctum heightens the examiner’s suspicion of the possibility of a canalicular laceration. If a patient is cooperative, the canaliculus can be gently probed with a lubricated 00 Bowman probe. Visualizing the shiny metal probe within the wound confirms the presence of a canalicular laceration. In children and some adults, the diagnosis of a canalicular laceration can be established only in the operating room or during examination under anesthesia. As with any adnexal injury, the ophthalmologist must perform a complete examination, including dilated retinal examination, to rule out serious ocular or intraocular injury.
Chemotherapy
Canalicular obstruction has been reported in cancer patients undergoing treatment with docetaxel (Taxotere, Aventis, Bridgewater, NJ)27,28 and with 5-Flourouracil.29 Docetaxel (Taxotere) is administered to patients with metastatic breast carcinoma, and is probably the most common chemotherapeutic agent causing canalicular stenosis.
Irradiation
Occlusion of the canaliculi and puncta occurs after irradiation for basal cell carcinoma almost 100% of the time, although intubation with silicone tubing may prevent this problem in some cases.30
Tumors
Skin cancer may involve the canalicular system, but intrinsic canalicular tumors such as papillomas may occur, producing occlusion and secondary inflammation.31
Use of Eye Drops
Echothiophate (Phospholine) iodide has been incriminated as a cause of canalicular stenosis as well as ocular pemphigoid syndrome, and idoxuridine toxicity may cause temporary occlusion of the punctum and canaliculus.32
Repeated Probing
One of the most common causes of stenosis of the lacrimal canalicular system is repeated and traumatic probing of the canalicular system for whatever reason.
Canaliculitis
Inflammation of the canalicular system can occur secondary to dacryocystitis, but isolated bacterial infections of the canaliculus are rare. Perhaps the most common infections are caused by Streptomyces, Actinomyces israelii, and Arachnia propionica (previously mislabeled as Streptothrix). Fungal infections with organisms such as Candida albicans, Aspergillus niger, and Nocardia have been reported. However, actinomycotic infection is by far the most common.33 In the clinical presentation, the lower canaliculus is usually involved and the patient reports epiphora. Swelling and inflammation of the lid medially are noted (Fig. 13.10). The punctum is swollen and red, sometimes described as pouting, and a mucoid and mucopurulent discharge is present. Irrigation may or may not be possible through the canaliculus, and a small probe may encounter gritty resistance. Diagnosis is made on expressing yellow-tinged concretions from the canaliculus. On cytologic examination, they show gram-positive branching filaments.
Allergy
Wojno34 has demonstrated intermittent allergic obstruction at the level of the canaliculus or lacrimal sac. This phenomenon is associated with allergic conjunctivitis and is noted in patients who chronically rub their eyes.
Disorders of the Lacrimal Sac
Infections
Infections of the lacrimal sac are clinically manifested as either acute dacryocystitis, marked by a tender and swollen lacrimal sac, rather severe pain, redness, and tearing; or chronic dacryocystitis, characterized by a smoldering infection within the lacrimal sac without distention of the sac but producing tearing and, in many cases, a chronic unilateral conjunctivitis (Fig. 13.11). Both conditions have as their underlying cause an obstruction to the normal tear passage within the nasolacrimal duct and sac, causing stasis and stagnation of tears and mucus and subsequent infection. The most common organisms in the ensuing infections are pneumococci. Other organisms include streptococci, diphtheroids, Klebsiella pneumoniae, Haemophilus influenzae, Pseudomonas aeruginosa, and mixed organisms. Actinomyces and fungi, such as Candida, are not infrequent, and granules and casts of the nasolacrimal duct and sac may be present in these cases.35 Sarcoidosis producing a chronic recurrent dacryocystitis has been reported in conjunction with systemic sarcoid.36,37
Lacrimal Stones or Casts
Casts or plugs may form within the lacrimal sac or nasolacrimal duct from fungus infections, organized blood clots, or inspissated mucous plugs. The most common organisms causing these seem to be A. israelii and Candida species. The clinical signs are intermittent epiphora and lacrimal obstruction, lacrimal conjunctivitis, recurrent or intermittent dacryocystitis, and variable localized tenderness. The underlying causes of the formation of stones are unclear. However, they occur more frequently in younger (under age 50) individuals and in heavy smokers.35 In many cases, it is possible to express, irrigate, and probe casts through the nasolacrimal duct, but DCR may be needed. A syndrome of acute noninfectious dacryocystic retention may occur when a cast, which may be mobile, plugs the nasolacrimal duct.38 Fluid buildup in the lacrimal sac causes closure of the valve of Rosenmuller at the common canaliculus, and the sac becomes acutely swollen and tender. This can be treated by percutaneous aspiration of the sac, followed by probing and irrigation. Casts may occur from chronic use of epinephrine drops.39
Dacryocystitis
The differential diagnosis of dacryocystitis includes a number of different clinical entities. The presentation of a mass in the lacrimal sac may be caused by a noninfectious amniotocele in a neonate or acute dacryocystic retention in an adult. Lacrimal sac neoplasms, unless attended by infection, are generally not tender and have a slower onset. In neoplastic disease, the mass in many cases extends above the level of the medial canthal tendon, which does not occur in dacryocystitis because of the compression of the fundus of the sac by the medial canthal tendon. Neoplasms that arise extrinsic to the lacrimal sac area include nasopharyngeal carcinomas, orbital rhabdomyosarcomas, and tumors of the antrum. A congenital midline meningoencephalocele may present as a mass in the lacrimal sac area and cause a secondary dacryocystitis. This anomaly may or may not communicate with the intracranial cavity. Dermoid cysts occur quite commonly nasally, although they are more often located in the supra-nasal quadrant and most usually are located at the lateral brow. Ethmoidal and frontoethmoidal mucoceles can occur and produce a firm masslike swelling at the inner canthal area and also a secondarydacryocystitis. Skin cysts and inclusion cysts maysimulate a mass in the lacrimal sac area. Chronicunilateral conjunctivitis with tearing and purulent discharge from the lower canaliculus that occurs with dacryocystitis also can be encountered in isolated canaliculitis.
Tumors
Clinical signs of a lacrimal sac tumor are tearing, painless irreducible swelling in the lacrimal sac area, and secondary dacryocystitis. Bleeding on probing of the lacrimal system is not an infrequent finding in sac tumors. It may be possible to irrigate through the lacrimal sac containing a tumor before the tumor completely occludes the lacrimal drainage system. Extension of tumors outside the sac area may cause intranasal symptoms, and radiographic changes are noted with erosion in the bone. Injection contrast radiography (dacryocystography) of the lacrimal sac may also demonstrate a nonfilling mass but may add little to the diagnosis, which may already be suspected. Historically, series of primary lacrimal tumors have been reviewed by Ashton and colleagues,40 Jones,41 and Radnot and Gall.42 Additional cases have been reported by Ryan and Font,43 Schenck et al.,44 and Stokes and Flanagan.45 In one series, about a third of the “tumors” were pseudotumors and were described as nonspecific granulomas, rhinoscleroma, lymphomatous lesions, sarcoid, syphilis, and fungi.42 In most series of true neoplasms of the lacrimal sac, 50% to 60% arise from the epithelial lining of the sac and are of the papillary carcinoma group, with histologic pictures identical with solid cylindric cell tumors arising from the respiratory epithelium.43
The well-differentiated papilloma group should be treated by aggressive, local excision and have a favorable prognosis, although the rate of recurrence is high. It has been recommended that all of the membranous lacrimal drainage apparatus, including the canaliculi and nasolacrimal duct, should be excised. The papillomas are classified into three histologic types: squamous cell, transitional cell, and mixed cell. They are all resistant to radiation therapy. The carcinomas are histologically squamous and transitional cell carcinomas, and they also may arise from the degeneration of a pre-existing papilloma. In Ryan and Font’s series, the survival rate of these patients after a wide local excision with frozen-section control was “reasonably good.”43 Long-term follow-up is necessary for patients with lacrimal sac papillomatous tumors.
The wall of the lacrimal sac contains lymphoid tissue, and lymphomas are probably the second most common intrinsic lacrimal sac tumor. They are similar to lymphomas occurring in the orbit in that they may be forerunners of lymphoid disease elsewhere in the body or the head and neck. The ultimate prognosis may be improved by noting the presence or absence of follicles, the anaplasia of the cells, and cell surface immunology.46 After biopsy and systemic evaluation, radiotherapy is indicated. Intubation of the lacrimal passages with silicone tubing before irradiation is indicated, and normal lacrimal drainage function for the patient after treatment may be retained.
Other primary lacrimal sac tumors that have been reported are malignant melanoma,47 oncocytic adenocarcinoma,48 neurilemmoma,49 adenocanthoma,50 hemangiopericytoma,51 and fibrous histiocytoma.52
The differential diagnosis should include invasive pharyngeal or sinus carcinoma and mucoceles of the ethmoidal sinus, which can be demonstrated on computed tomography scans. Rarely, orbital rhabdomyosarcomas present in this manner, and other mass lesions, previously mentioned, should be considered.
Disorders of the Nasolacrimal Duct
PrimaryAcquired Nasolacrimal Duct Obstruction
Primary acquired nasolacrimal duct obstruction (PANDO) is the most common clinical syndrome of acquired nasolacrimal duct obstruction in adults. Patients may present with symptoms of chronic epiphora, conjunctivitis, and low-grade infections or with acute dacryocystitis. This clinical syndrome is most common in elderly white women.
Clinical pathologic studies by Linberg and McCormick53 examined the histopathology of the entire membranous nasolacrimal duct in patients with the clinical syndrome of PANDO. These studies have revealed inflammation, vascular congestion, and edema of the nasolacrimal duct in the early phases and, ultimately, fibrosis with complete occlusion of the nasolacrimal duct’s lumen in the late phases.
The specific trigger of this sequence of events is not known. Nonetheless, it is reasonable to postulate that inflammation with partial ductal obstruction leads to accumulation of cellular debris, which aggravates the ongoing inflammation and creates a vicious cycle that leads to permanent cicatrization of the nasolacrimal duct lumen.
Linberg’s studies have demonstrated that in patients with symptoms of relatively short duration (less than 1 year), the inflammation and edema “functionally” occlude the nasolacrimal duct. A potential space does remain within the lumen, however. This pathologic finding lends credence to the hope that PANDO may be reversible in patients with symptoms of short duration. For this reason, as discussed later in the treatment section, this subset of patients may be candidates for either medical therapy with antiinflammatory drugs or nasolacrimal duct intubation with silicone tubes to maintain patency of the duct until the inflammation subsides or has been treated.
Patients with PANDO and chronic symptoms (greater than 2 to 3 years’ duration) demonstrate dense fibrous scar tissue and cicatrization of the nasolacrimal duct as a sequela of chronic inflammation, edema, and stasis of cellular debris. In these patients, the lumen of the nasolacrimal duct is permanently obliterated by scar tissue. This histopathologic finding correlates with the very poor success rate of nasolacrimal duct probing or intubation with silicone lacrimal tubes in adult patients with chronic PANDO symptoms. As discussed later, DCR remains the treatment of choice for this group of patients.
Although most adult nasolacrimal duct obstructions represent the syndrome of PANDO, noninflammatory infiltrative disorders can occlude the nasolacrimal duct. Clinicians must maintain a high index of suspicion in patients with known systemic disorders such as sarcoidosis, lymphoma, or leukemia. In these situations, distal nasolacrimal sac or nasolacrimal duct biopsy is an important part of the DCR surgery. Linberg and McCormick recommend their nasolacrimal duct biopsy technique as a routine part of all DCRs.53 In this manner, important infiltrative causes of nasolacrimal duct obstructions would not be overlooked.
Trauma
Nasolacrimal duct obstructions may occur as a sequela of midfacial fractures involving the bony nasolacrimal canal. Immediate disruption of the nasolacrimal drainage system occurs in some patients. This is typical of severe crushing nasal orbital fractures and the Lefort II and LeFort III fractures, in which extensive damage is sustained by the entire lacrimal drainage apparatus, including the canthal tendons. In other patients, bony fractures initiate an inflammatory, cicatrizing process that results in symptomatic nasolacrimal duct obstructions many years after the original injury. Some investigators have advocated early silicone tube intubation of the lacrimal outflow system in patients with complex midfacial fractures.54 The efficacy of this treatment is not yet fully established.
A number of cases of dacryostenosis have been reported after cosmetic rhinoplasty. It is believed that in most instances, the damage to the membranous nasolacrimal duct occurs during the lateral osteotomy.55 Other sinus and nasal operations may also injure the nasolacrimal duct. This is especially true of Caldwell-Luc procedures and nasoantral window formation. Nasoantral windows are usually created in the most anterior-inferior portion of the maxillary sinus. If they are placed too high or too posterior, however, or if the nasolacrimal canal is in an anomalous position, excision of a portion of the nasolacrimal duct may occur. The Ogura procedure (orbital decompression) with excision of the floor of the orbit and ethmoidal air cells through an antrostomy may also be associated with postoperative epiphora. Again, the likely cause may be the nasoantral window formation.56,57
Prior midfacial or nasal radiation therapy may result in nasolacrimal duct obstructions. In prior generations, radiation treatment was common for conditions such as facial acne and chronic sinusitis. As with some traumas, this occlusive radiation fibrosis effect may be delayed for many years.
Intranasal Disorders
The distal aspect of the nasolacrimal duct may be obstructed as a result of intranasal pathology. Intranasal scarring with inferior turbinate adhesions may occur as a sequela of trauma, radiation therapy, or surgical procedures (e.g., nasoantral window formation). Allergic rhinitis may be associated with nasal mucosal hypertrophy. In some individuals, an abnormally wide nasal vestibule is associated with compensatory hypertrophy of the inferior turbinate that occludes the valve of Hasner (open nasal space syndrome with epiphora).58 Tumors are uncommon and can be benign, such as granulomas or nasal polyps, or malignant, such as squamous cell carcinoma.
Lacrimal Pump Failure: Miscellaneous Disorders
As indicated previously, the lacrimal outflow system may be anatomically patent to irrigation yet be functionally inadequate in terms of normal lacrimal elimination. Facial nerve paresis is among the most common situations in which lacrimal pump failure is present despite patency of the membranous lacrimal conduit. Any condition that impairs the normal contractile and elastic properties of the palpebral-canalicular pump mechanism can cause epiphora. These conditions include scleroderma, radiation fibrosis of the eyelids, and cutaneous burns or trauma of the periocular region. Chronic or recurrent canaliculitis may leave the canaliculi anatomically patent yet functionally impaired.
Diagnosis in Patients with Epiphora
The most common symptom of disorder in the function or anatomy of the lacrimal drainage system is tearing. The blockage or malfunction may occur at many places along the tear elimination route, and the appropriate therapy, surgical and nonsurgical, may be necessary to correct it. Some patients with tearing symptoms truly produce more tears than the normal drainage system can handle, and some people may have conditions that cause more of a sensation of tearing than actual overloading of the drainage system. Table 13.1 shows a classification of the various physiologic and anatomic causes of tearing.
The steps necessary in evaluating a patient with tearing are listed next in the usual sequence. In many cases, not all the steps are needed because the diagnosis may be apparent with some of the more simple tests alone.
History
Slit-lamp examination (noting lid movement and position and integrity of punctum)
Pressure over lacrimal sac
Irrigation of lower canaliculus
Intracanalicular probing (diagnostic)
Dye drainage tests (fluorescein test) for functional blocks
Schirmer’s test
Intranasal examination
Conventional x-ray films and computed tomography
Dacryocystography
Dacryoscintigraphy
A patient presenting to an ophthalmologist with tearing should be evaluated in a systematic manner, and the proper sequence should be observed to promote efficient use of time and to avoid confusion.
History
In evaluating a patient with tearing symptoms, no diagnostic technique is more valuable than taking a careful history. A unilateral watering eye is more commonly a sign of an obstructive process, although bilateral nasolacrimal duct obstructions can occur. Is there actual excess tearing in which tears run down the patient’s cheek(s) and require frequent dabbing, or does the patient have diminished visual acuity and simply refer to the sensation as “my eye is tearing”—a common complaint of patients with macular degeneration.
The primary consideration for clinicians is to distinguish epiphora from hyperlacrimation. Epiphora denotes symptoms of excess tearing due to lacrimal outflow deficiency. Hyperlacrimation denotes that an excess production of tears accounts for the patient’s symptoms. Table 13.1 shows a classification scheme for tearing disorders, including the differential diagnosis of epiphora and hyperlacrimation.
Patients with hyperlacrimation commonly have ocular discomfort. A chronic gritty foreign body sensation associated with excess tearing is typical of patients with keratitis sicca (“dry eye” syndrome). Patients with entropion or trichiasis, blepharitis, corneal abrasions, chronic conjunctivitis, or photophobia and iritis also frequently have hyperlacrimation symptoms. In addition to the comfort of their eyes, patients should be questioned about prior seventh nerve palsy or increased tearing while eating.
Patients with epiphora commonly have “excess tears” as their only symptom. The examiner should carefully question the patient about intermittent redness of the eyes, mucous production or heavy lid crusting in the morning, pain or swelling in the region of the lacrimal sac, or prior episodes of acute dacryocystitis. These symptoms may indicate chronic or intermittent dacryocystitis. Severe, intermittent symptoms of tearing or dacryocystitis that are interrupted by completely normal periods may suggest a “ball valve” disorder such as dacryolithiasis. The clinician should question the patient with epiphora to focus on the key anatomic areas of interest. A history of facial nerve paresis, scleroderma, or lid scarring may indicate a dysfunctional lacrimal pump mechanism. The chronic use of phospholine iodide, idoxuridine, or prior severe conjunctivitis can point toward punctal stenosis as the problem. A history of previous lacrimal therapy may be significant. Repeated probing or instrumentation of the lacrimal canaliculi can result in severe canalicular stenosis. Prior “overaggressive” punctoplasties may actually impair tear elimination. Patients should be questioned about a history of chronic allergies or sinusitis, previous nasal or sinus surgery, and prior midfacial fractures or radiation therapy. Any of these would be pertinent in considering a possible nasolacrimal duct obstruction.
The age of the patient (child, adult, elderly) and the duration of the symptoms (acute, chronic) are relevant. These variables have an important bearing on the likely cause of the problem and thus greatly influence the treatment that is implemented. Childhood lacrimal obstructions are usually due to an imperforate membrane at the distal nasolacrimal duct. Adults, once hyperlacrimation has been ruled out, commonly have the syndrome of PANDO. This type of adult lacrimal obstruction is potentially reversible if the blockage is functional or if symptoms have been of a relatively short duration (less than 6 months). Chronic epiphora symptoms (greater than 12 months) are rarely treatable with any method other than definitive DCR. Specific management of these epiphora problems is expanded on in the treatment section.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree