16 Laboratory Techniques, Special Stains and Immunohistochemistry
SPECIMEN HANDLING IN THE OPHTHALMIC PATHOLOGY LABORATORY
This chapter discusses the basic principles of practical ocular histopathology and summarizes techniques used in the handling, gross dissection, and submission of enucleated eyes and other ocular specimens for routine histopathologic examination. Special histochemical and immunohistochemical stains also are discussed.
Fixation
Routinely, unopened enucleated eyes are fixed for at least 24 hours by total immersion in a relatively large volume (50–100 mL) of 10% neutral buffered formalin. The large volume of fixative is necessary because the solution must penetrate through the sclera. Bouin solution (bright yellow in color) should not be used because it hardens the sclera, making eyes extremely difficult to section. Newer alcohol-based fixatives are also unsatisfactory because they precipitate protein in the vitreous and interfere with gross examination. Enucleated eyeballs should be briefly rinsed in running tap water to remove excess fixative prior to handling.
Specimen Orientation
Determination of laterality (right or left eye) is a prerequisite for proper specimen orientation (Fig. 16-1). The laterality of an eye is determined by identifying key anatomic landmarks. These include the long posterior ciliary vessels, which appear as blue lines on the posterior sclera on either side of the optic nerve in the horizontal plane; the cornea, whose horizontal diameter is usually a millimeter longer than its vertical diameter; and the insertions of the superior and inferior oblique muscles. The inferior oblique muscle lacks a tendon; its fibers insert directly into the sclera in the inferotemporal quadrant. The nasal end of the muscular inferior oblique insertion lies close to the fovea. The shiny tendinous insertion of the superior oblique muscle is located superiorly and temporally and is an excellent landmark for the superior pole of the eye. After the horizontal meridian and the superior pole have been located, the nasal and temporal sides of the eye are readily identified. It is important to remember that both oblique muscles insert on the temporal side of the eye and their fibers run nasally. The temporal arc of the posterior sclera is longer than the nasal arc because the optic nerve enters the eye 15 degrees nasal to the posterior pole. The angulation of the optic nerve stump cannot be relied upon to determine laterality, however, because it often bends temporally after fixation.
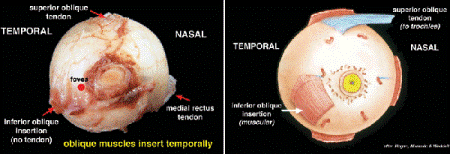
Fig. 16-1. Orientation of globe, posterior landmarks. The temporal insertions of the oblique muscles are helpful landmarks. The inferior oblique lacks a tendon and its muscular fibers insert inferonasally near the fovea. The tendinous insertion of the superior oblique muscle is located superotemporally. The long posterior ciliary vessels are often evident as blue lines on either side of the optic nerve in the horizontal plane.
Measurement
The oriented eye is measured using calipers or a millimeter ruler. Standard measurements include the anteroposterior (AP), horizontal, and vertical diameters of the eye; the length of the optic nerve segment attached; the horizontal and vertical diameters of the cornea; and the size of the pupil (Fig. 16-2A). Measurements should be made and recorded in a standardized sequence, that is, AP, horizontal, and vertical. Important pathologic features such as wounds, lacerations, and tumors should also be measured and their location noted. Most normal human eyes are slightly less than 1 inch in diameter (24–25 mm), and the normal cornea is approximately 1 cm in diameter (11 × 10 mm).
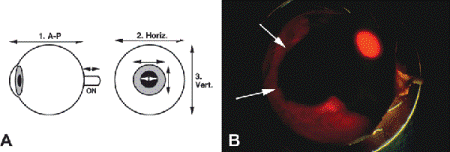
Fig. 16-2. A. Ocular measurements. Standard measurements include the AP, horizontal, and vertical diameters of the eye; the length of the optic nerve segment; the horizontal and vertical diameters of the cornea; and the size of the pupil. B. Transillumination of globe. Transillumination is used to localize ciliochoroidal melanoma in enucleated eye. Arrows point to round shadow cast by choroidal part of pigmented tumor. The pupil glows brightly.
It is easier to section an eye if the optic nerve is removed first. The nerve should be sectioned a millimeter or two posterior to the sclera to avoid “buttonholing” a deeply cupped nerve head. A transverse section of the optic nerve should always be submitted if the nerve has not been cut flush with the globe. The surgical margin should be marked with India ink or an indelible colored pencil before the nerve is removed. The optic nerve segment should always be removed before the globe is opened to preclude potential contamination of the surgical margin with intraocular tumor. This is especially important if the eye contains retinoblastoma, which tends to invade the optic nerve. Surgeons occasionally submit the optic nerve in a separate container. In such specimens, blood staining and crushing serve to identify the true surgical margin.
INSPECTION AND DESCRIPTION
The general consistency and character of the eye are noted. Descriptive terms that are commonly used include normal, soft, collapsed, ruptured, lacerated, hard, rubbery, phthisic, etc. Before it is opened, the external surface of the eye should be carefully and systematically examined with a dissecting microscope, starting anteriorly. The cornea is usually slightly hazy in fixed or postmortem specimens. The presence of corneal opacities, scars, vascularization, ulcers, band keratopathy, wounds, and incisions should be described and measured when appropriate. The shape and size of the pupil, the color of the iris, and the presence and location of iridectomies and surgical iris colobomas should be recorded. If the cornea is hazy, iris defects are readily disclosed by transillumination.
The rest of the globe should be carefully inspected looking for signs of surgical or nonsurgical trauma or extraocular tumor extension. The presence, type, location, and size of wounds, scars, vitrectomy ports, sutures, and prosthetic devices such as retinal implants, explants, and encircling bands and tube shunts should be precisely noted. The use of “clock hours,” for example, “between 1 and 3 o’clock,” is helpful in the description of wounds and other important lesions.
Drawing a diagram of the findings on the pathology protocol facilitates description and permits reconstruction of the gross description if the dictation is lost.
TRANSILLUMINATION
Eyes are transilluminated before they are opened to disclose occult pathology that may not be evident on external examination. Transillumination is performed by holding the eye directly on the surface of a bright light in a darkened chamber. Transillumination is especially helpful during the orientation of eyes that contain intraocular tumors, particularly uveal malignant melanomas (Fig. 16-2B). Pigmented melanomas usually cast a dark shadow on the sclera, which should be localized and measured. When clinical data are absent, transillumination may disclose signs of previous surgery, for example, an iridectomy, that may be an indication for vertical sectioning. Eyes that are filled with blood or contain dense gelatinous exudate often transmit light poorly, as do specimens from heavily pigmented individuals. Staphylomas and cyclodestructive procedures cause increased light transmission.
Microscopic sections are cut from a paraffin block that contains a short cylindrical segment of the eyeball called the pupil-optic nerve or P-O segment. The P-O segment includes most of the cornea, iris, and pupil anteriorly and the optic nerve posteriorly (Fig. 16-3B).
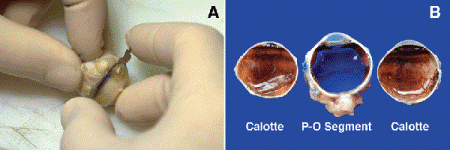
Fig. 16-3. A. Sectioning technique for globe. Globe is sectioned cornea down with a razor blade. B. P-O segment and calottes. The central P-O segment, which is submitted for sectioning includes the pupil and optic nerve.
When tissue is submitted for histopathologic examination, care must be taken to ensure that the microscopic sections will include important lesions such as tumors, lacerations, or incisions. The presence and location of the ocular pathology determine how the eye is opened. It is routine practice to open eyes horizontally if they do not contain wounds or other focal lesions. Horizontal P-O sections include the macula as well as the pupil and optic nerve.
Eyes that have had prior surgery may be opened vertically to include a superior limbal surgical wound in the microscopic sections. This category includes eyes that have had cataract surgery or filtering procedures for glaucoma. If an eye with a small superior incision is opened horizontally, the microscopic sections will not include the wound, and the pathologist will be unable to document its presence.
Traumatic lacerations and intraocular tumors can occur anywhere. Such eyes are opened along the meridian that includes the main part of the lesion. The localization of intraocular tumors by transillumination greatly aids sectioning.
SECTIONING TECHNIQUE
I currently section eyes using one half of a standard double-edged razor blade that has been snapped in half within its protective wrapper. The two ends of the blade are held between the apposed surfaces of the thumb and forefinger, and a gentle sawing and pushing motion is used. During the initial cut, the eye is steadied with the nondominant hand and held cornea side down on the cutting block (Fig. 16-3A). Guidelines drawn on the eye with colored pencil facilitate sectioning and assure that the eye maintains proper orientation. The first cut is begun just external to the dural sheath of the optic nerve and should enter the periphery of the anterior chamber anteriorly. The two dome-shaped caps of tissue that are removed are called calottes (French for visor-less cap). The first calotte should include about one fourth or one fifth of the peripheral anterior chamber. Ideally, the central P-O segment should be about 8- to 10-mm thick. The globe is placed cut surface down on the cutting board during removal of the second calotte. Eye protection (glasses, goggles, or face shield) should be worn during the initial cut to protect against occasional squirts or splashes of fixative and intraocular contents that can occur if the intraocular pressure is high.
Any material that flows from the eye during sectioning (blood, crystals, pigment-tinged fluid, or silicone oil) should be described. Gritty, hard, or even impenetrable intraocular material may be encountered. When this happens, possibilities include intraocular bone, a calcified cataractous lens, or a prosthetic intraocular lens. Intraocular bone is an extremely common finding in phthisical eyes. Eyes that contain bone must be decalcified before they are submitted. In some instances, decalcification is a prerequisite for initial dissection.
Every specimen should be examined carefully and systematically with the dissecting microscope, and macrophotography performed if indicated. Disturbing light reflexes are minimized during gross examination and macrophotography by immersing the specimen in a flat-bottomed cylindrical dish filled with 60% “grossing” alcohol. Placing the flat-bottomed cylindrical dish on a plate of dark blue Plexiglas provides an aesthetic background for the gross photographs, which is enhanced if the bottom of the dish is coupled to the plastic with a thin layer of alcohol. The calottes are retained in alcohol as “wet tissue” and may be retrieved if additional sections or studies are necessary.
ROUTINE HISTOPATHOLOGY: DEHYDRATION, EMBEDDING, AND MICROTOMY
The P-O segment is embedded in a block of paraffin wax to facilitate the cutting of microscopic sections. Because water and wax are not miscible, the aqueous fixative and the water in the tissue must be removed before the specimen can be embedded. This process is called dehydration. Tissue is dehydrated by passing it through increasing concentrations of ethanol until absolute ethanol is reached. The dehydrated tissue is then transferred to xylene, which is miscible with both absolute alcohol and paraffin, and then infiltrated with molten paraffin under vacuum. After infiltration, the specimen is placed in a plastic mold that is filled with molten paraffin. The paraffin is cooled and hardens into a block, which fits into the chuck of a rotary microtome. The support provided by the surrounding paraffin matrix allows the tissue to be sectioned thinly (6–8 m). The thin slices of paraffin and tissue are then floated onto the surface of a bath of heated water, which causes the paraffin to expand. The tissue sections are then mounted on glass slides, which are heated in an oven to promote tissue adherence.
STAINING
Most human tissues are relatively transparent unless they contain endogenous pigment (Fig. 16-4A). To facilitate examination, microscopic slides are stained with dyes such as hematoxylin and eosin (H&E) that color certain tissue components. Because aqueous solutions of these stains usually are used, the tissue sections must be deparaffinized and rehydrated before they can be stained. This is done by immersing the slides in successive baths of xylene and absolute alcohol and then decreasing the concentrations of alcohol and water. After staining, the tissue subsequently is dehydrated and coverslipped.
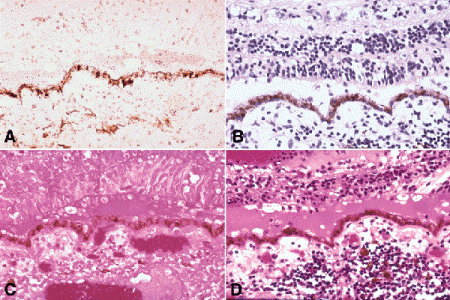
Fig. 16-4. Staining, Hematoxylin and eosin stain. A. Unstained section. Presence of endogenous pigment allows identification of retinal pigment epithelium and choroidal melanocytes. B. Hematoxylin-stained section (same area as that in Fig. 16-4A). Basic dye stains DNA in cellular nuclei, highlighting retinal nuclear layers and inflammatory cells in choroid. C. Eosin-stained section (same area as that in Fig. 16-4A). Acidic dye stains cytoplasm, erythrocytes in choroidal vessels, and proteinaceous subretinal fluid. D. Hematoxylin and eosin-stained section (same area as that in Fig. 16-4A). A shallow retinal detachment and chronic nongranulomatous choroiditis are present. Photoreceptor atrophy denotes chronic process. (A. Unstained section ×100, B. Hematoxylin ×100, C. Eosin ×100, D. Hematoxylin and eosin ×100)
In most laboratories, routine histopathologic sections are stained with H&E (Fig. 16-4D). Hematoxylin is a basic dye that binds to acidic materials including the DNA in the nuclei of cells (Fig. 16-4B). Eosin (tetrabromofluorescein) is acidic and stains basic substances such as proteins pink (Fig. 16-4C).
H&E staining provides helpful diagnostic color cues. Lesions composed of cells with scanty cytoplasm tend to look blue or basophilic during microscopy. Examples of basophilic lesions include inflammatory infiltrates, lymphoid tumors, retinoblastoma, and basal cell carcinoma. Necrosis, calcification, and DNA deposition in eyes with retinoblastoma also appear blue. In contrast, cells that have abundant cytoplasm (e.g., squamous cells or epithelioid histiocytes) appear pink. Squamous cell carcinomas usually are eosinophilic. Normal ocular structures composed largely of connective tissue such as the cornea and sclera appear pink in H&E sections. Protein-rich subretinal fluid, lens material, amyloid, and exfoliation material also are eosinophilic.
The periodic acid-Schiff (PAS) stain is used routinely in most ophthalmic pathology laboratories because it vividly highlights the thick basement membranes of the eye (lens capsule and Descemet membrane). PAS stains materials that contain unsubstituted vicinal glycol groups (CHOH-CHOH). The vicinal glycol groups are oxidized to dialdehydes (CHO-CHO) by periodic acid, and the Schiff reagent (leucofuchsin) reacts with the dialdehydes forming complexes that range in color from red to magenta. In addition to basement membranes and aggregates of basement membrane material such as guttae and cuticular drusen, PAS stains glycogen, some mucins (e.g., conjunctival goblet cells), and many but not all fungal hyphae.
SPECIAL HISTOCHEMICAL STAINS
A variety of special histochemical stains occasionally are ordered in the ophthalmic pathology laboratory to demonstrate the presence of microorganisms and highlight special structures and materials in histologic sections. Standard histochemical stains rely on chemical reactions to demonstrate the presence of substances. All special stains must be used in conjunction with positive control slides that contain the sought-for organisms or materials. These control slides confirm that the special stains are working. In some instances, the use of histochemical stains has been replaced by immunohistochemical stains, which are more specific.
Special stains for microorganisms are ordered most often. These include modifications of the Gram stain for bacteria called the Brown and Hopps and the Brown and Brenn stains (Fig. 16-5A), several acid fast stains for mycobacteria (Fig. 16-5B), and the Gomori methenamine silver (GMS) stain for fungi (Fig. 16-5C). The nonspecific fluorochrome stain calcofluor white can be used to detect fungi and acanthamoeba cysts, but requires an ultraviolet microscope (Fig. 16-5D). The Dieterle and Warthin-Starry silver impregnation techniques for spirochetes, Bartonella, and Legionella occasionally are ordered (Fig. 5-9). Fungi and yeast are impregnated with silver and appear black against a green counterstain when stained with the GMS stain (Fig. 16-5C). The Ziehl-Neelson and Fite-Firaco acid fast stains are used to demonstrate the acid fast organisms that cause tuberculosis, leprosy, nocardiosis, and atypical mycobacterial infections (Fig. 16-5B). The organisms appear red (“red snappers”) against a blue background.
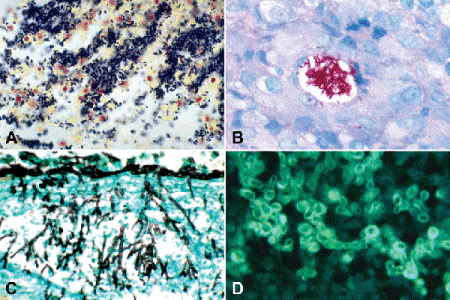
Fig. 16-5. Special stains for microorganisms. A. Tissue Gram stain. Tissue Gram stain discloses myriad Gram-positive cocci in exudate from eye with endophthalmitis. B. Acid fast stain. Stain discloses large colony of atypical mycobacteria within vacuole in granulomatous inflammation. Patient developed infection after blepharoplasty. C. GMS stain for fungus. Branching septate Aspergillus hyphae in choroid of patient with endogeneous endophthalmitis are stained black by silver impregnation. D. Calcofluor white. Yeast stained with nonspecific fluorochrome stain calcofluor white fluoresces vividly during ultraviolet microscopy. (A. Brown and Hopps ×250, B. Ziehl-Neelson ×250, C. GMS ×250, D. Calcofluor white ×400)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


