Intraocular Tumors in Adults
Ralph C. Eagle Jr
The ophthalmologist’s primary duty is to preserve or improve vision. Occasionally, however, he or she may elicit symptoms or make observations that can save a patient’s life. Various malignant tumors arise primarily within the eye or can involve its tissues secondarily. Ocular tumors generally are rare, but some have significant mortality. The retinal tumor retinoblastoma predominates in early childhood and is discussed in another chapter. Most tumors in adults affect the uveal tract. Primary nevi and malignant melanomas arise from its constituent population of melanocytes. The uvea, particularly the posterior choroid, is also involved secondarily by distant metastases from primary neoplasm elsewhere in the body, mainly carcinomas, and also is affected by leukemia and lymphoma. Other primary tumors that affect the uveal stroma are hemangiomas and rare entities including leiomyomas, peripheral nerve sheath tumors, and hemangiopericytomas. Other rare intraocular tumors in adults arise from the neuroepithelial layers of the eye. The retina in adults may be affected by vascular tumors, most often capillary hemangiomas or hemangioblastomas, which often are associated with the tumor diathesis Von Hippel-Lindau disease. Acquired neoplasms of the iris and retinal pigment epithelium and the pigmented and nonpigmented epithelia of the ciliary body occur, but are quite rare compared to melanoma. These will be discussed in greater detail later in this chapter.
Melanocytic Tumors of the Uveal Tract
In adults, most intraocular tumors arise from or involve the uveal tract, the eye’s middle coat comprising the stroma of the iris, ciliary body, and choroid. The uvea tract is highly vascular and pigmented. The pigment is contained within the cytoplasm of dendritic uveal melanocytes, which are derived embryologically from the neural crest. A similar number of uveal melanocytes are present in lightly and heavily pigmented eyes. Increasing intensity of uveal pigmentation (and eye color) are caused by a corresponding increase in the size and number of melanin pigment granules or melanosomes in the cytoplasm of the melanocytes.1 The pigment may protect against the development of uveal tumors, because uveal malignant melanoma occurs most often in patients with blue eyes and is rare in heavily pigmented individuals. The choroidal vessels occasionally undergo hamartomatous proliferation forming hemangiomas, and the rich vascular supply of the posterior choroid explains that region’s predilection for blood-borne metastases.
Nevi
Uveal nevi are benign neoplasms derived from uveal melanocytes. Ninety percent are located in the choroid posterior to the equator.2 Like nevi of the skin, uveal nevi are quite common; it has been estimated that they occur in more than 6% to 10% of the population.3 Nevi are often detected during routine fundus examinations (Fig. 20-1). Most choroidal nevi are flat or minimally elevated lesions that usually measure between 1.5 and 5 mm in diameter; most are <2 mm in thickness. Nevi may be pigmented or amelanotic, and they often have an irregular or jagged border. Drusen are quite common on the surface of nevi. Most nevi are stationary lesions that do not change on serial observation. Nevi have been observed to give rise to malignant melanomas, but this is a rare phenomenon. If one assumes the prevalence of uveal nevi to be 5% per year and the incidence of melanoma to be 7 per million per year, it can be estimated that the rate of transformation of nevi to melanoma is only 10,000 to 15,000 per year.4,5
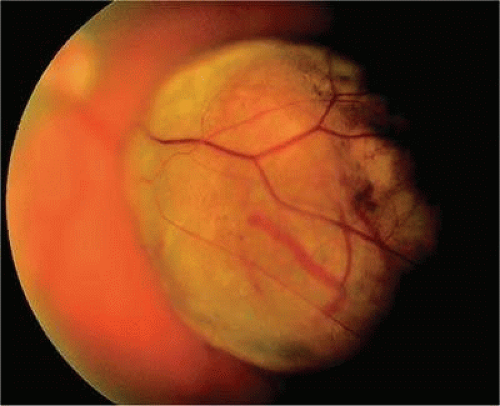
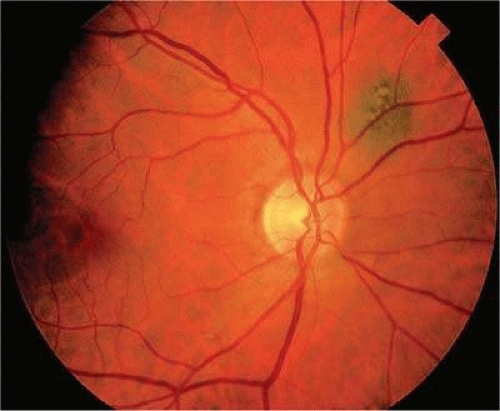 Figure 20.1. Choroidal nevus. Small nonsuspicious pigmented nevus is flat and lacks drusen, orange pigment, and subretinal fluid. |
Dr. Carol Shields has emphasized several clinical factors that assist in differentiating small choroidal melanomas from choroidal nevi clinically.6,7 These can be remembered using the mnemonic TFSOM (to find small ocular melanoma), where T is thickness >2 mm, F is subretinal fluid, S is symptoms, O is orange pigment, and M is margin touching optic disc. Choroidal melanocytic tumors that display none of these factors have a 3% risk of growth into melanoma at 5 years and most likely represent choroidal nevi. Tumors that display one factor have a 38% risk of growth, and those with two or more factors show growth in over 50% of cases. Most tumors with two or more risk factors probably represent small choroidal melanomas, and early treatment is generally indicated.6,7 It may be impossible to differentiate large choroidal nevi from small malignant melanomas clinically. The observation of growth is the only clinical criterion that is helpful in this regard.8
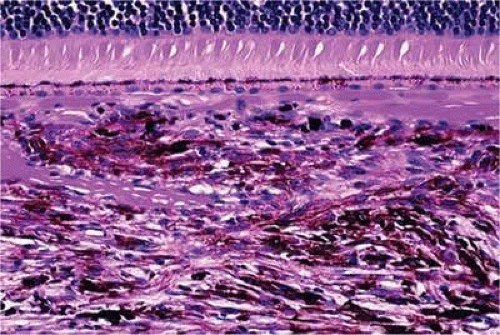
Nevi constitute the benign end of the biologic spectrum of melanocytic neoplasms. Most nevi are bland spindle cell tumors. Histopathologically, the choroidal stroma typically is replaced by a compact infiltrate of slender pigmented or nonpigmented spindle cells (Fig. 20-2). The nevus cells have bland oval or cigar-shaped nuclei that have finely dispersed chromatin and lack nucleoli or nuclear folds. Intranuclear cytoplasmic inclusions are common in some cases. Mitotic activity is absent. In some cases the nevus cells are plump and dendritic in shape.9 Foamy balloon cells that appear to be undergoing lipoidal degeneration are found in 4% of nevi. Maximally pigmented, plump, polyhedral nevus cells constitute the magnocellular variant of nevus called melanocytoma (see below).
Most pigmented tumors of the iris are low-grade spindle cell nevi that remain stationary and do not enlarge on follow-up (Fig. 20-3). The iris freckle is a small, stationary form of iris nevus that is found in half of the adult population.1
Melanocytoma (Magnocellular Nevus)
A melanocytoma is a characteristic type of uveal nevus that is composed of plump polyhedral nevus cells filled with copious quantities of maximally pigmented cytoplasm.10,11 Melanocytomas also are called magnocellular nevi.12 In contrast to most nevi, melanocytoma may be relatively large and may be difficult to distinguish clinically from melanoma.
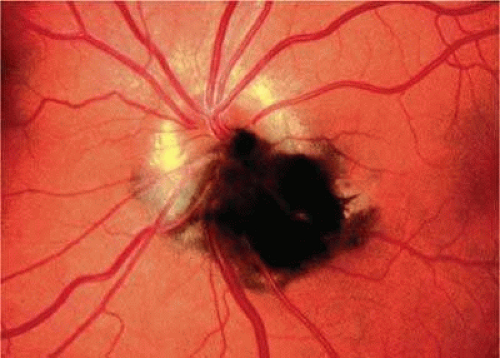
Although best known as a pigmented lesion of the optic disc (Fig. 20-4), melanocytoma can arise from the stroma of the iris, ciliary body, or choroid13,14,15,16,17 (Fig. 20-5). Clinically, melanocytomas are intensely pigmented and often occur in young patients. Unlike uveal melanoma, they do not have a predilection for whites. Thirty-seven percent to 50% of optic disc melanocytomas have been reported in blacks.18 Melanocytomas of the iris are uniformly dark brown in color and often have an irregular friable surface.
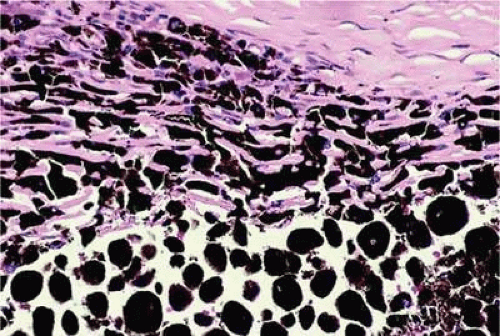
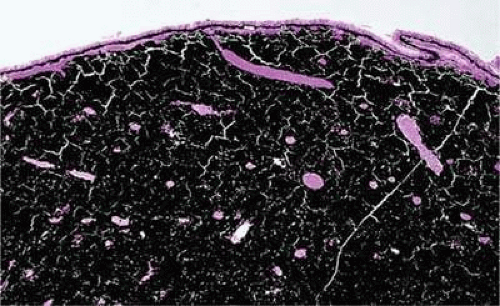 Figure 20.5. Ciliary body melanocytoma. Ciliary epithelium covers intensely pigmented tumor. Melanin pigment obscures nuclei. (Hematoxylin-eosin, X 50.) |
Most melanocytomas of the optic disc have a feathery margin caused by infiltration of the nerve fiber layer. In approximately one half of cases, a juxtapapillary component in the choroid identical to a choroidal nevus is present.16,17 Most patients are asymptomatic with normal vision or slight visual loss. Nine percent have an afferent pupillary defect.19 Visual field examination may show enlargement of the blind spot or nerve fiber bundle defects.19 About 10% to 15% of optic disc melanocytomas enlarge on followup.16,17 Rare cases of malignant transformation have been reported.20,21,22,23 The incidence of malignant transformation is estimated to be 1% to 2%. Central retinal vein occlusion has been reported in a patient with a necrotic optic disc melanocytoma and does not always imply malignant transformation.24 Primary malignant melanomas of the optic nerve head have been reported.25 Retinal pigment epithelial adenoma may mimic optic disc melanocytoma.26
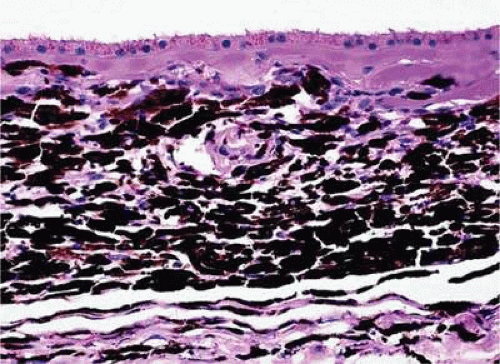
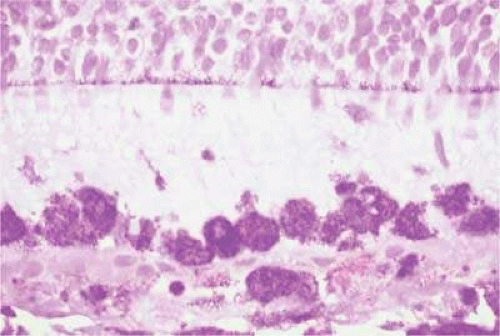
Histopathologically, melanocytomas are composed of cells that have copious amounts of maximally pigmented cytoplasm that often obscures nuclear details, making microscopic assessment impossible11 (Fig. 20-5). Bleached sections, which are a requisite for examination, show that the cells have cytologically bland nuclei and a low nuclear cytoplasmic ratio (Fig. 20-6). Nucleoli usually are inconspicuous, but there are exceptions to the rule. Electron microscopic studies also have revealed a second population of spindle cells.15,27 Melanocytoma cells resemble the benign melanocytes found in the uvea of patients with ocular melanocytosis.
Melanocytomas are predisposed to undergo spontaneous necrosis.14 One is much more likely to find extensive tumor necrosis in a melanocytoma than a melanoma of comparable size, and totally necrotic melanocytomas occasionally are encountered. Numerous plump melanophages typically are found when necrosis is present. It may be difficult to differentiate the melanophages from magnocellular nevus cells in cytologic preparations obtained by fine-needle aspiration biopsy.
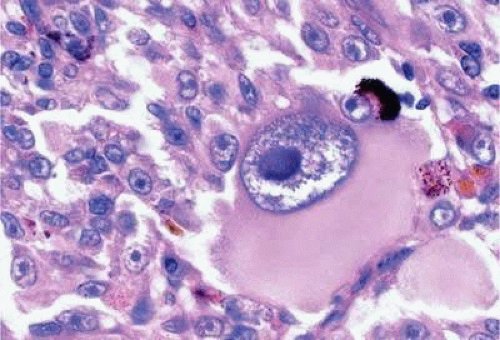
A rare type of secondary glaucoma called melanocytomalytic glaucoma may develop in patients who have partially necrotic melanocytomas of the iris and ciliary body.28,29,30 In such cases, the open angle is obstructed by macrophages that have ingested melanin pigment released by the necrotic tumor (Fig. 20-7).
Uveal Melanoma
On a worldwide basis, uveal melanoma is the most common primary intraocular tumor in adults. In the United States and Europe, uveal melanoma is also the most common primary intraocular tumor. In most of the rest of the world, the most common intraocular tumor is the pediatric retinal neoplasm retinoblastoma.31
Uveal melanoma is rare, even among persons of European ancestry. Fewer than 2,000 cases occur yearly in the United States where the incidence is estimated to be 7 cases per million per year, which is <10% the incidence of lung cancer.32
Although pediatric and even rare congenital cases33,34 have been reported, uveal melanoma generally occurs in older persons. The mean age of patients eligible for treatment in the COMS I125 brachytherapy study was 59 years.35 Fewer than 0.8% of cases occur in patients younger than age 20 years.36 In general, patients who are older tend to have larger tumors and are more likely to die from disseminated melanoma after enucleation. Recent data from the COMS Study indicates that uveal melanoma affects both sexes equally.35
Predisposing Factors
Race is an important predisposing factor. Uveal malignant melanoma has a definite predilection for Europeans who have blue or lightly pigmented eyes.37,38 In a recent study in Florida, the risk of uveal melanoma in non-Hispanic white men was 72 times that in black men.32 Uveal melanoma also is less common in Latin and Native Americans32,39 and Asians. The incidence of uveal melanoma in the United States is 25 times greater than that in Taiwan (i.e., 7 per million versus 0.28 per million, respectively).40 The prognosis of melanoma in black and white patients is similar. Tumors in black patients typically show more necrosis and heavy pigmentation.41
White patients who have congenital ocular or oculodermal melanocytosis (nevus of Ota) are especially at risk to develop uveal malignant melanoma.42 It has been estimated that about one in 400 white patients with oculodermal melanocytosis will develop uveal melanoma in their lifetime.43 This risk is about 25 times greater than the risk in unaffected patients. Melanomas have developed in young patients with oculodermal melanocytosis.36,44,45,46 Ocular and oculodermal melanocytosis are relatively common in Asians,47 but do not appear to predispose them to melanoma. Several blacks with nevus of Ota who developed uveal melanoma have been reported.48,49
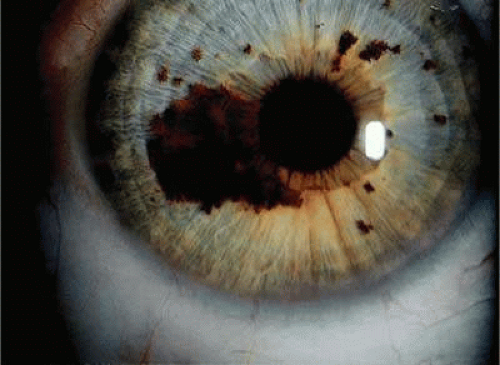
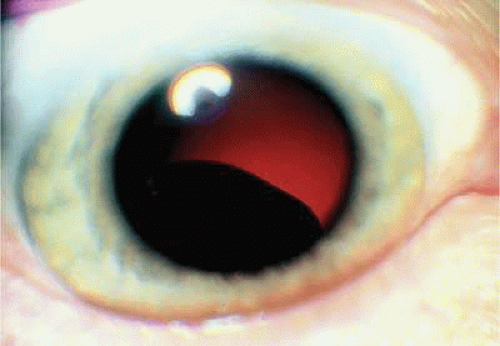
The substrate for the development of melanoma in white patients with ocular melanocytosis is a diffuse nevus that affects all or part of the uveal tract. Clinically, the nevus typically is evident as hyperchromic heterochromia iridum, patchy slate gray epibulbar pigmentation and a darker aspect of the fundus compared to the fellow eye (Fig. 20-8). Only a sector of uvea is affected in some patients, however, and the nevus may spare the iris. The uvea in congenital melanocytosis is thickened by an increased number of heavily pigmented nevus cells similar to those found in melanocytomas (Fig. 20-9). Retinal pigment epithelial abnormalities such as drusen often develop on the surface of the thickened choroid.50 The sclera also contains patchy foci of pigmentation. The slate gray conjunctival pigment reflects the presence of dendritic melanocytes on the epibulbar tissues deep to the conjunctiva. Nevus of Ota does not predispose to conjunctiva melanoma. However, patients rarely may develop melanomas of the orbit51,52,53 and even leptomeninges.1,54,55 White patients who have congenital ocular or oculodermal melanocytosis should be followed periodically because of the increased risk of uveal melanoma.
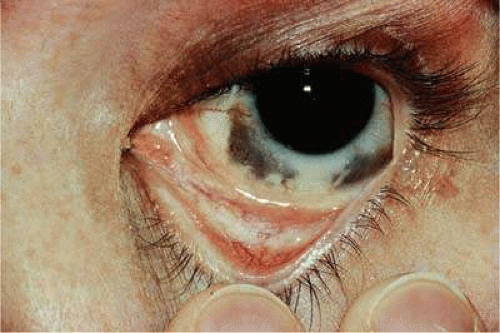 Figure 20.8. Nevus of Ota. White patient who developed choroidal melanoma has dark brown iris and slate-gray pigmentation of sclera and periocular skin. |
Uveal melanoma occasionally arises from localized uveal nevi, but the estimated incidence of malignant transformation is quite low. Nevuslike cells observed histopathologically at the base of many uveal melanomas probably are a compression artifact and do no represent a pre-existing nevus.56 Cases of uveal melanoma have been reported in patients with von Recklinghausen neurofibromatosis type I46,57,58,59,60 and the dysplastic nevus or familial atypical mole-melanoma syndrome.61,62,63 The tumor’s propensity for light-eyed individuals and the exposed inferior part of the iris suggests that exposure to ultraviolet light could be a predisposing factor.37,38 Conjugal melanoma (i.e., the occurrence of tumors in both husband and wife) may be coincidental, but could be indicative of the role of shared environmental factors. An increased incidence of uveal melanoma has been reported in a group of chemical workers.64
Uveal melanoma can arise in patients who have a rare paraneoplastic syndrome called benign diffuse uveal melanocytic proliferation or BDUMP syndrome.65,66,67 Unknown factors produced by a nonocular cancer stimulate diffuse proliferation of benign-appearing nevoid or spindle cells throughout the uveal tract of both eyes. Often occult, the primary tumor is typically a poorly differentiated ovarian or uterine carcinoma in women or lung carcinoma in men. Patients typically present with rapid visual loss and usually have cataracts. Fundus examination shows multiple pigmented and nonpigmented placoid nodules and reddish patches in the choroid, which show striking early hyperfluorescence during intravenous fluorescein angiography (IVFA), as well as serous retinal detachment.66 One patient presented with nonocular pigmentation typical of the Peutz-Jeghers syndrome.67
Clinical Features
Clinical Presentation
The clinical signs and symptoms of uveal malignant melanoma depend largely on the location of the tumor and the extent of the disease when the patient initially seeks medical attention. More than a century ago, Fuchs68 described four characteristic stages in the natural history of uveal melanoma. These include a relatively asymptomatic stage, a symptomatic stage marked by glaucoma and/or inflammation, a stage of extraocular growth, and finally, distant metastasis. Today, most patients are encountered in the early stages of the disease. Overt metastatic disease is almost never evident when the patient initially presents or is treated.69 However, many patients are now thought to have clinically undetectable micrometastases.70
Totally asymptomatic tumors occasionally are detected on a routine ophthalmoscopic examination. However, most choroidal melanomas present with painless visual loss. In the great majority of cases, visual loss is caused by serous or solid detachment of the retina. In rare instances, the pathway of light to the fovea is physically blocked by the mushrooming apex of an actively growing, anteriorly located tumor. Ciliary body melanomas frequently do not produce retinal detachment and therefore may remain relatively asymptomatic for some time. Ciliary body melanomas cause loss of vision by directly encroaching on the visual axis, or by impinging on the lens causing visually significant distortion, subluxation, or secondary cataract formation. Peripheral melanomas also can cause decreased vision by inducing cystoid macular edema. Dilated episcleral and conjunctival vessels called sentinel vessels often mark the site of an underlying ciliary body tumor.
Vitreous hemorrhage is an extremely rare cause of visual loss in uveal melanoma because the retina serves as a barrier that prevents the blood from entering the vitreous. Retinal perforation is a requisite for vitreous hemorrhage. This occurs most often when highly elevated tumors are situated near the optic disc or ora serrata where the retina is attached. Perforation of the retina also can lead to extensive seeding of the vitreous and the inner surface of the retina.71 In rare instances, malignant epiretinal membranes of seeded tumor cells can produce a pseudoretinitis pigmentosa-like picture.72
The ready visibility of many iris melanomas leads to their diagnosis at an earlier age and a smaller size (Fig. 20-10). Iris melanoma typically is discovered when a patient or his or her physician detects a focal pigmented blemish. Higher-grade iris melanomas, or ciliary body tumors that invade the anterior chamber, can seed the iris and anterior chamber causing acquired hyperchromic heterochromia iridum (Fig. 20-11). Secondary glaucoma frequently accompanies the change in iris color. Not infrequently, the underlying diagnosis may be overlooked and the patient treated for glaucoma, especially when a melanoma is nonpigmented or confined to the periphery of the iris or the trabecular meshwork.
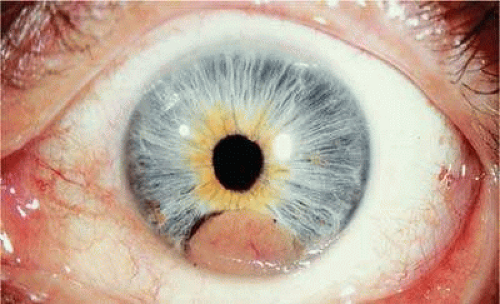 Figure 20.10. Iris melanoma. Amelanotic tumor fills inferior part of anterior chamber. Gonioscopy and transillumination disclosed no involvement of ciliary body. |
Most eyes with untreated uveal melanoma eventually develop glaucoma.73,74 Anterior uveal tumors such as high-grade iris melanomas or ciliary body melanomas that invade the anterior chamber typically cause secondary open-angle glaucoma by seeding or directly infiltrating the trabecular meshwork (Fig. 20-12). The latter often occurs in a circumferential or ring fashion. Posterior melanomas generally cause secondary angle-closure glaucoma by stimulating iris neovascularization or by causing pupillary block. The pupil is blocked when large tumors, or tumors that have caused highly elevated total retinal detachments, displace the lens iris diaphragm forward (Fig. 20-13). The same mechanism also occurs in eyes with other neoplasms that often produce high bullous retinal detachments such as uveal metastases, choroidal hemangiomas, and exophytic retinoblastomas. In such cases, severe pain often prompts a patient with a blind eye to seek medical care. More than a century ago, it initially was emphasized that a glaucomatous eye with a total retinal detachment harbors a intraocular tumor, typically a melanoma, until proven otherwise. Imaging studies such as ultrasonography are mandatory prior to therapy in such cases, especially if evisceration of a blind painful eye is contemplated instead of enucleation.
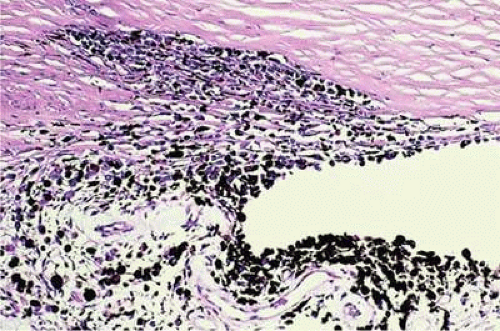 Figure 20.12. Diffuse iris melanoma with secondary open angle glaucoma. Tumor cells infiltrate trabecular meshwork and seed its inner surface. (Hematoxylin-eosin, X50.) |
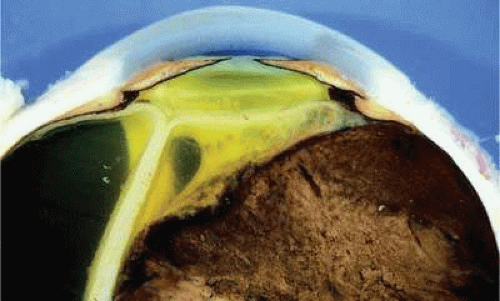 Figure 20.13. Large ciliochoroidal melanoma with total retinal detachment and secondary closed-angle glaucoma caused by anterior displacement of lens–iris diaphragm. |
Rarely, patients with uveal melanoma may present with extraocular extension of their tumors.50,75,76 Epibulbar foci of tumor from ciliary body melanomas that have grown out of the eye through anterior emissarial canals or Axenfeld nerve loops can be confused clinically with conjunctival melanomas, but are not attached to the conjunctival epithelium (Fig. 20-14). Although some posterior melanomas infiltrate directly through the sclera, transscleral extension typically occurs along the emissarial canals of vessels or nerves (Fig. 20-15). Rarely, a posterior nodule of extrascleral extension develops that is much larger than the primary intraocular part of the tumor that spawned it.50,76,77 Such patients may present with ocular proptosis. In exceptional circumstances, patients with chronically neglected tumors, which have filled and destroyed the eye, will present with an orbital mass.
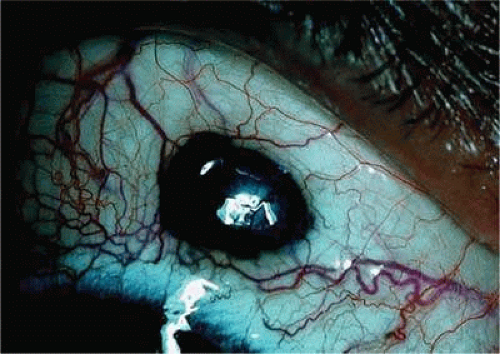 Figure 20.14. Anterior epibulbar focus of transscleral extension from ciliary body melanoma. Conjunctiva covers tumor, which has grown through emissary canal. |
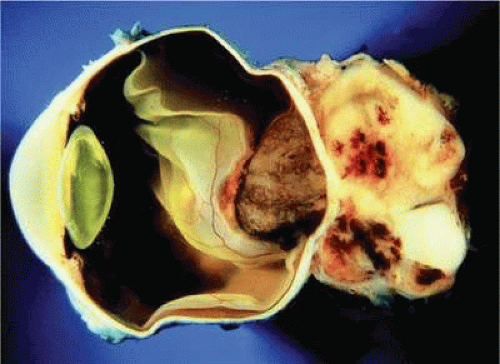 Figure 20.15. Massive posterior extrascleral extension, uveal melanoma. Pigmented choroidal melanoma has extended extrasclerally, forming large nonpigmented orbital mass. |
Necrotic melanomas may present with severe orbital and adnexal inflammation that can mimic infectious cellulitis or idiopathic orbital inflammation.78 Extensive necrosis typically is found in eyes with rapidly growing high-grade tumors. Marked elevation of intraocular pressure in eyes with secondary glaucoma may cause infarction of the tumor and other intraocular structures.
Unsuspected melanomas occasionally are discovered when blind painful eyes with opaque media are examined pathologically. Before the advent of ultrasonography as many as 10% of blind painful eyes were said to harbor previously undiagnosed tumors.79 The possibility of a tumor should always be entertained when cataract is unilateral or asymmetric. It is quite embarrassing to diagnose a melanoma during cataract surgery.
Clinical signs
Ophthalmoscopic examination of a choroidal melanoma typically reveals a sessile or dome-shaped pigmented mass located deep to the retina (Figs. 20-16, 20-17 and 20-18). There often is an associated secondary nonrhegmatogenous serous detachment of the retina. Orange pigment may be located on the surface of smaller lesions.80,81,82 Tumors vary in pigment content and may be heavily pigmented or totally amelanotic. The differentiation of a totally amelanotic melanoma from uveal metastasis, hemangioma, lymphoma, or osteoma may be difficult. Compared to metastasis, melanoma usually is more highly elevated, and drusen and retinal pigment epithelial proliferation more evident on its surface. Ophthalmoscopy also typically shows well-defined vessels within melanoma.50 Ancillary studies such as fluorescein angiography and ultrasonography may be necessary to make the diagnosis.
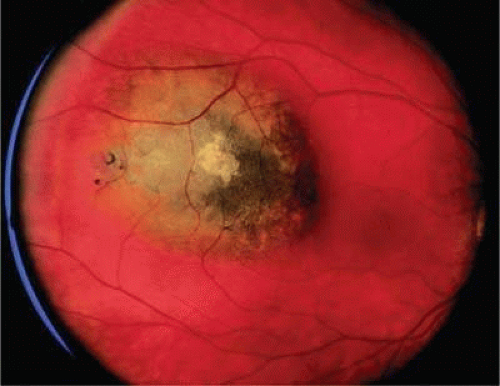 Figure 20.16. Small pigmented tumor thought to be choroidal nevus found on routine ophthalmoscopic examination. Nevus is slightly elevated, and RPE changes are present. |
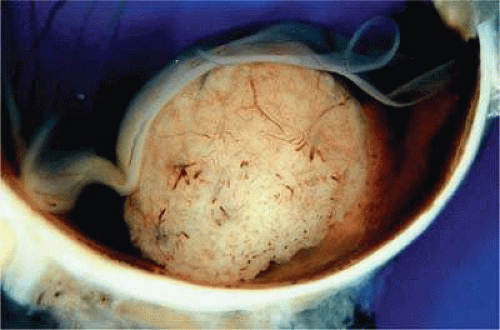
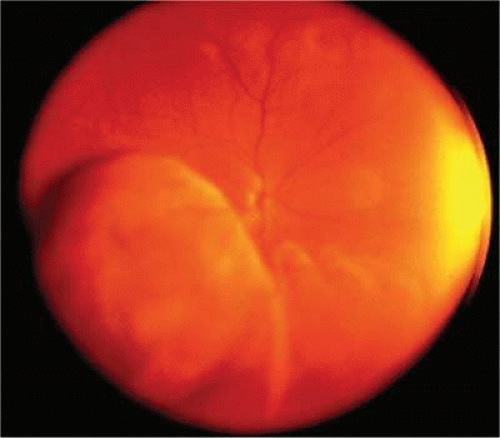 Figure 20.18. Large ciliochoroidal melanoma. Wide-angle photo shows extent of lesion and associated inferior retinal detachment. |
Many choroidal melanomas have a characteristic mushroom or collar button configuration that results when the tumor breaks through Bruch’s membrane (Figs. 20-19, 20-21 and 20-22). In such cases there is often secondary choroidal, subretinal or vitreous hemorrhage. Dilated, intrinsic vessels often are evident in the mushrooming dome of nonpigmented melanomas. The vessels are obscured in more heavily pigmented tumors.50
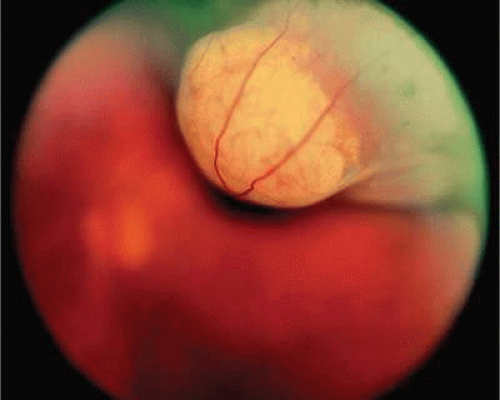 Figure 20.19. Fundus photo of mushroom-shaped choroidal melanoma with nonpigmented dome. Configuration indicates that tumor has ruptured through Bruch’s membrane. |
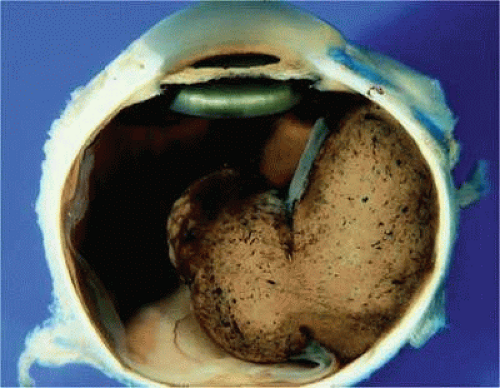 Figure 20.21. Gross photo, large mushroom-shaped ciliochoroidal melanoma. Pigmented tumor is highly elevated. |
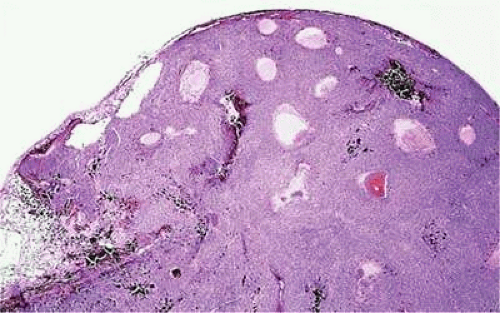 Figure 20.22. Mushrooming head of choroidal melanoma contains dilated vessels that are located anterior to constricting gap in Bruch’s membrane. (Hematoxylin-eosin X10.) |
Although the diagnosis of melanoma often can be made by careful indirect ophthalmoscopy, ancillary studies such as intravenous fluorescein angiography, indocyanine green angiography, ultrasonography, computed tomography, magnetic resonance imaging, and fine-needle aspiration biopsy occasionally are required to support or confirm the diagnosis.50
The characteristic fluorangiographic features of uveal melanoma include mottled hyperfluorescence in the vascular filling phases and diffuse late staining of the mass and surrounding subretinal fluid. A characteristic double circulation in which both the retinal vessels and the choroidal vessels in the tumor are readily evident may be shown by larger amelanotic tumors, particularly those that have perforated Bruch’s membrane. The prominent blood vessels seen in amelanotic mushroom-shaped melanomas may impart a pseudoangiomatous appearance to the lesion.50
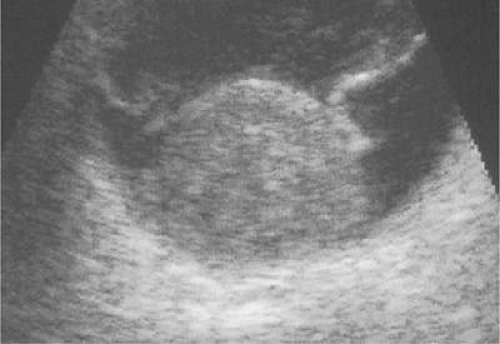
Ultrasonography can support the diagnosis of ciliary body or choroidal melanoma and is a particularly helpful way of disclosing tumors in eyes with opaque media.50 Ultrasonographic studies can also demonstrate areas of extrascleral extension. A scan ultrasonography shows a high internal spike and low internal reflectivity. B scan ultrasonography shows characteristic acoustic hollowness and choroidal excavation (Fig. 20-23). Hemangiomas and metastases usually appear acoustically solid because they contain multiple acoustic interfaces.
Transvitreal fine-needle aspiration biopsy (FNAB) occasionally is performed if the diagnosis remains uncertain after routine tests, and choice of therapy requires an accurate diagnosis.83,84 Potential candidates for FNAB include the patient who has a history of breast cancer and presents with a solitary amelanotic choroidal tumor that could be a primary amelanotic melanoma, or the patient who is thought to have a choroidal metastasis but has no history of cancer.84
Gross pathology
Choroidal melanomas initially arise in the stroma of the choroid. In early cases, the profile of the sectioned tumor is oval or almond-shaped and its tissue usually appears relatively cohesive after fixation (Fig. 20-24). Although some tumors diffusely infiltrate the uvea, most uveal melanomas are relatively well-circumscribed tumors with distinct margins. In many cases the growing melanoma perforates Bruch’s membrane and enters the subretinal space where its apex typically assumes a spherical shape that often is likened to a mushroom or collar button (Figs. 20-19, 20-20, 20-21 and 20-22). Dilated vessels often are found in the mushrooming head of the tumor because the ends of Bruch’s membrane exert a compressive cinchlike effect on the waist of the tumor (Fig. 20-22). Rupture of Bruch’s membrane was present in 87.7% of 1,527 large or medium-sized melanomas examined in the COMS study.85 Retinal invasion was present in nearly half (49.1%), and tumor cells were found in the vitreous body in one fourth.
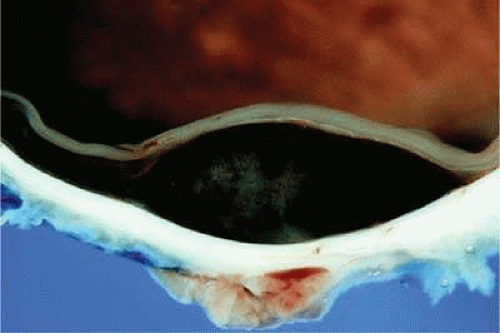 Figure 20.24. Cut surface of heavily pigmented choroidal melanoma is almond-shaped. Bruch’s membrane is intact. |
Uveal melanomas vary markedly in their pigment content. Some tumors are totally amelanotic; others are maximally pigmented. In some instances, distinct clones of tumor cells that vary in pigment content are evident on gross examination, or even clinically (Fig. 20-25). A friable or granular appearance of the cut surface of a melanoma noted during gross examination may be indicative of necrosis or a tumor rich in poorly cohesive epithelioid cells. Melanomas that contain cystic cavities occasionally are encountered.86
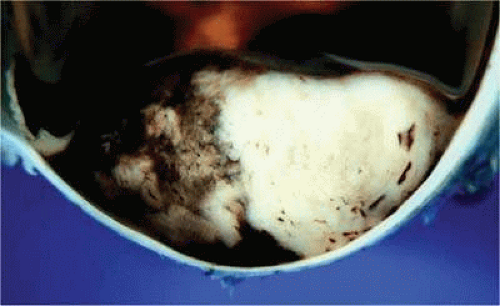 Figure 20.25. Large dome-shaped ciliochoroidal melanoma is partially amelanotic. Bruch’s membrane is intact. |
About 3% of melanomas have a diffuse growth pattern. These relatively flat tumors grow laterally without thickening the choroid.87,88 Most are about 2 mm in thickness. Diffuse melanomas often are of mixed cell type and, compared to localized lesions, are more apt to infiltrate the sclera and invade the optic nerve or orbit (Fig. 20-26). Delayed diagnosis or misdiagnosis is common.
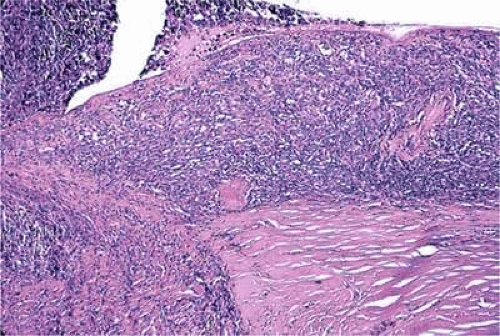 Figure 20.26. Diffuse choroidal melanoma thickens choroid. Massive invasion of optic nerve is seen at left. (Hematoxylin-eosin X50.) |
Choroidal melanomas are classified as small, medium, or large based on the tumor’s largest basal diameter (LTD). Small choroidal melanomas are 10 mm in diameter or less and appear as focal discoid or oval areas of choroidal thickening. Medium-sized melanomas measure 11 to 15 mm, and large tumors are more than 15 mm in largest basal tumor diameter. Larger tumors are more likely to have ruptured through Bruch’s membrane.
In the COMS study, medium-sized tumors were located more posteriorly than large tumors.85 Presumably, more anteriorly situated ciliary body tumors are larger when they are first detected because they remain hidden behind the iris, cause late retinal detachment, and hence remain asymptomatic. Ciliary body melanomas (Figs. 20-27, 20-28 and 20-29) are less common than choroidal melanomas and tend to be more spherical in shape. As noted above, ciliary body melanomas can cause unilateral cataract, lens molding, or subluxation (Fig. 20-28). In addition to causing secondary glaucoma by angle seeding or circumferential infiltration, ciliary body melanomas that invade the periphery of the anterior chamber often displace the iris root centrally, causing a solid iridodialysis. Heterochromia iridum also is possible if there is extensive chamber seeding. A ciliary body tumor that perforates the iris initially may be misdiagnosed as a small iris tumor that in reality is the “tip of the iceberg” (Fig. 20-29).
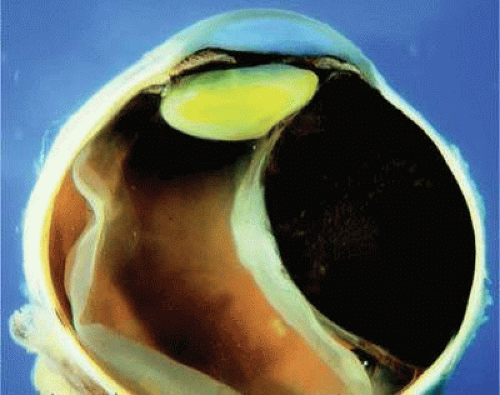 Figure 20.28. Large, heavily pigmented ciliochoroidal melanoma. Dome-shaped tumor is causing lens subluxation. |
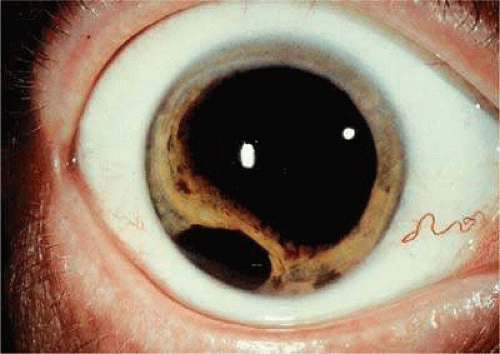 Figure 20.29. Anterior chamber invasion by ciliary body melanoma. Part of the tumor is seen through pupil behind the iris. |
Retinal detachment is the most common cause of visual loss in eyes with posterior melanoma. Initially solid, the detachment becomes exudative as serous fluid accumulates. Visual loss initially may be caused by induced hyperopia as a subfoveal tumor displaces the retina anteriorly. The exudative detachment initially is localized; fluid accumulates in the subretinal space tented up by the growing tumor. Failure to find a retinal hole in a patient with retinal detachment should always raise the possibility of a choroidal tumor or some other choroidal process known to produce an exudative detachment. Total retinal detachments are found in some eyes with choroidal melanomas, generally those with larger or neglected tumors (Fig. 20-13). Melanoma-bearing eyes with total retinal detachments often have secondary closed-angle glaucoma caused by pupillary block or iris neovascularization.73,74 Clumps of orange pigment may be seen in the subretinal space overlying the tumor.
During gross examination, the external surface of an eye harboring a melanoma should be inspected carefully for signs of extraocular extension. The latter may be evident as an epibulbar nodule or a vortex vein that is dilated and heavily pigmented. Tumor infiltration of scleral emissarial canals may be evident on the cut surface of the globe at the base of the tumor.
Treatment effects are evident in eyes whose tumors have been treated previously with radioactive plaque brachytherapy or transpupillary thermotherapy (TTT).89,90 Prior plaque therapy frequently causes prominent areas of diffuse chorioretinal depigmentation and atrophy. TTT scars usually are white and sharply demarcated like a coloboma and often are rimmed by heavily pigmented foci of retinal pigment epithelium (RPE) hyperplasia.90
Histopathology
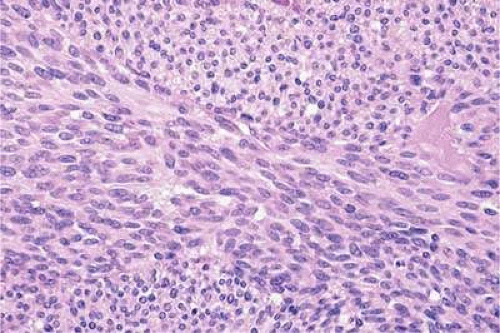
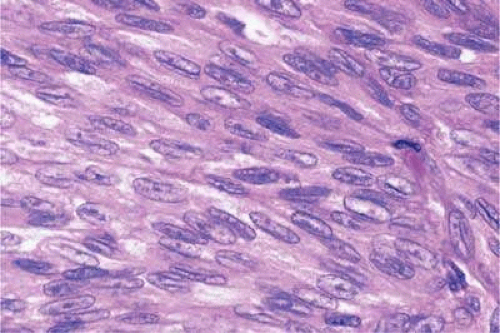
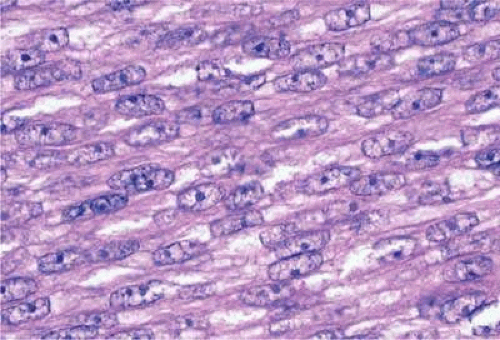
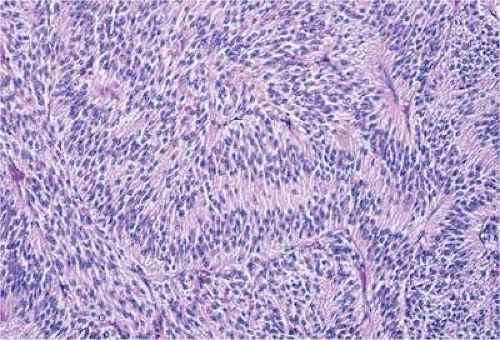
The cells composing uveal melanoma constitute a biologic spectrum comprising bland spindle A melanoma cells at one end and wildly anaplastic epithelioid cells at the other. The term spindle cell is derived from the fusiform or spindled configuration of the cells’ cytoplasmic outline. They are bipolar in shape, and many have long tapering processes that occasionally are highlighted when individual pigmented cells are seen in a largely amelanotic tumor. Spindle cells grow in a syncytial fashion forming interweaving fascicles of parallel oriented cells (Fig. 20-30). The cells can be pigmented or nonpigmented. Two types of spindle cells are recognized; spindle A and spindle B. These are distinguished by their nuclear characteristics. Spindle A nuclei are tapering ovals or cigar shaped and have finely dispersed chromatin (Fig. 20-31). If a nucleolus is present, it usually is inconspicuous. Many spindle A cells have a longitudinally oriented chromatin stripe that actually reflects a fold in the nuclear membrane. The nuclei of spindle B cells have distinct nucleoli and coarser chromatin and tend to be plumper and more oval (Fig. 20-32).
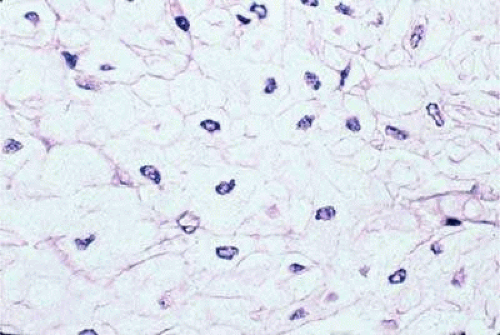
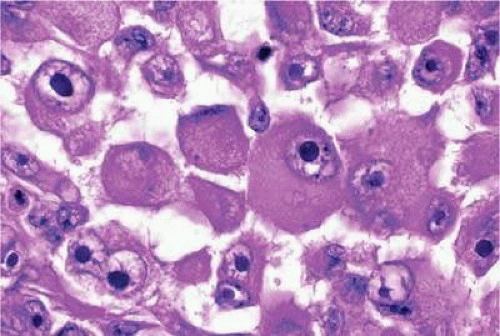
Epithelioid melanoma cells constitute the poorly differentiated end of the cytologic spectrum. Melanomas that contain epithelioid cells have a poorer prognosis. The term epithelioid, meaning epithelial-like, denotes the fanciful resemblance of the tumor cells to the cells of simple epithelia. Epithelioid cells have abundant cytoplasm and are often polygonal in shape (Fig. 20-33). They have distinct cytoplasmic margins, are poorly cohesive, and do not grow as a syncytium. The nuclei of epithelioid cells are usually round or oval and often appear vesicular owing to margination or clumping of the chromatin along the inner side of the nuclear membrane. Epithelioid melanoma cells also have prominent nucleoli that are often large and reddish-purple. Variants of epithelioid cells include relatively uniform small epithelioid cells (Fig. 20-34) and bizarre tumor giant cells that may appear wildly anaplastic (Fig. 20-35).
Melanoma cells are classified by their nuclear characteristics. Spindle-shaped cells that have epithelioid nuclei occasionally are encountered; such cells are classified as epithelioid. In recent years the term intermediate cell has been used increasingly. Intermediate cells are cells that have nuclear characteristics that are intermediate between spindle B and epithelioid. For example, one might apply the term intermediate cell to a spindle B cell that has a nucleus that is somewhat large and has a fairly prominent nucleolus.
Occasionally, the spindle cells in a melanoma are arranged in a radial fashion around vessels or fibrovascular septa (vasocentric pattern), or the nuclei form rows that resemble the Verocay bodies or the Antoni A pattern seen in schwannoma (Verocay pattern). Melanomas are called fascicular if these patterns dominate4 (Fig. 20-36). Fascicular melanoma was a category in Callender’s initial classification91 that was removed from the 1983 modification by McLean et al.92
Varying degrees of necrosis may be found (Figs. 20-37 and 20-38). Necrosis tends to be more prominent in rapidly growing high-grade tumors or tumors that have had prior brachytherapy. The necrosis may be patchy and focal, or may involve extensive parts, or even all of the tumor. Aggregates of melanophages typically are found in the necrotic areas. Total infarction of the tumor (and other intraocular structures) may occur in eyes with severe secondary closed-angle glaucoma. As mentioned previously, melanocytoma is prone to spontaneous necrosis. The latter diagnosis should always be considered when a totally necrotic, heavily pigmented tumor is found.
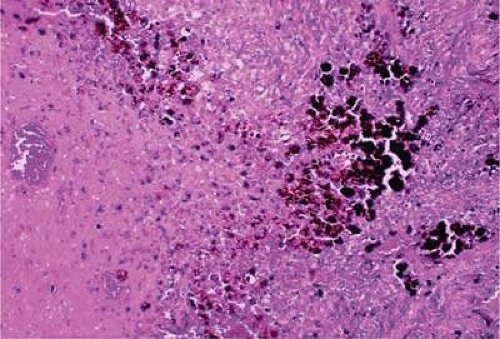 Figure 20.37. Focus of necrosis in choroidal melanoma. Pigmented melanophages have accumulated at interface between viable tumor and necrotic focus (below). (Hematoxylin-eosin X100.) |
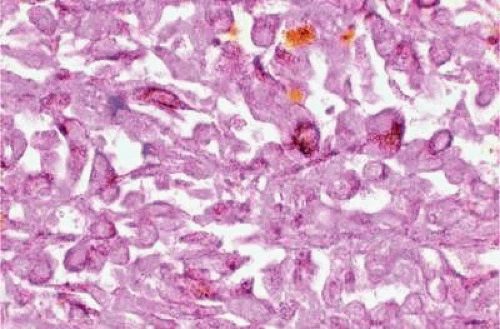
Choroidal melanomas produce abnormalities in the overlying retinal pigment epithelia including atrophy, hyperplasia, and the formation of drusen and drusenoid material.93 The overlying retina often shows photoreceptor loss and may develop cystoid edema. The latter tends to be more common over slower-growing lesions, especially choroidal hemangiomas. After Bruch’s membrane has ruptured, the vessels located in the mushrooming head of the tumor are often quite prominent, reflecting vascular stagnation caused by the compression at the waist of the tumor (Fig. 20-22). Aggregates of macrophages that have ingested PAS-positive lipofuscin pigment and melanin from the damaged retinal pigment epithelium can be found in the subretinal fluid (Figs. 20-39, 20-40 and 20-41). These are evident ophthalmoscopically as clumps of orange pigment that serve as a clinical marker for an actively growing neoplasm.82,94
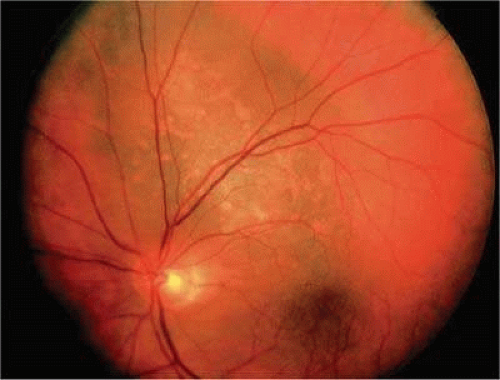 Figure 20.39. Juxtapapillary melanoma with orange pigment. Orange pigment is a clinical marker for an actively growing tumor. |
 Figure 20.40. Orange pigment. Clumps of orange pigment adhere to outer surface of detached retina overlying actively growing melanoma. |
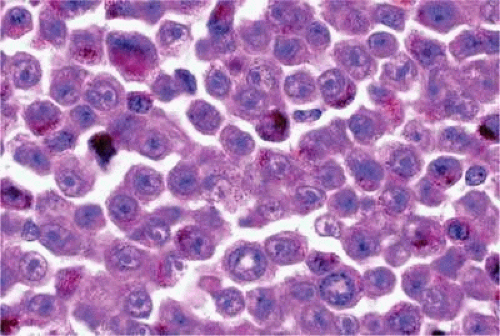
Melanocytic tumors of the uvea are classified into four groups on the basis of cytology. Tumors composed entirely of spindle A cells or even blander nevus cells are classified as spindle cell nevi. Tumors composed of a mixture of malignant spindle A and spindle B cells are called spindle melanomas. Melanomas of mixed cell type contain a mixture of spindle and epithelioid melanoma cells (Fig. 20-42). Some laboratories specify the predominant cell type found in a mixed cell melanoma, for example, reporting mixed cell, predominantly spindle if only a few epithelioid cells are present. Epithelioid melanomas are composed predominantly of epithelioid cells. They are relatively rare and have the poorest prognosis. Most medium and large-sized melanomas contain a mixture of spindle and epithelioid cells. In the COMS histopathology study, 86% of the posterior melanomas were classified as mixed cell type; 8% were of spindle cell type, and 5% were epithelioid.85 The association between cytology and mortality is known as the Callender classification.91 (See the section on prognostic factors later in this chapter.)
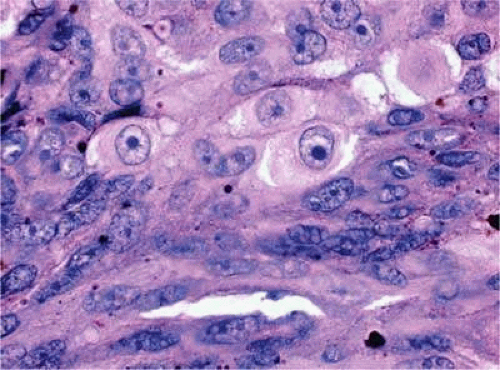 Figure 20.42. Uveal melanoma, mixed cell type. Mixed cell melanomas are composed of a mixture and spindle and epithelioid cells. (Hematoxylin-eosin X250.) |
Iris melanoma is relatively rare, constituting between 4% and 15% of uveal melanomas in various series.95 The studies with higher reported incidences may reflect inclusion of nevi. Iris melanomas differ in some respects from tumors of the posterior segment. Most are low-grade spindle cell tumors (Fig. 20-43). However, iris melanomas with epithelioid cells occasionally are encountered (Fig. 20-44). Many pigmented iris tumors actually are nevi,96 and relatively few of these enlarge when observed. Several studies have shown that the risk of enlargement in 5 years is only 5% to 6% after a subset of promptly treated lesions is excluded.97,98 One study showed that iris tumors are likely to be considered melanomas and be treated promptly if the basal diameter is >3 mm, pigment dispersion or prominent tumor vascularity is present, the intraocular pressure is elevated or there are tumor-related symptoms.98 The mean age of patients with iris melanomas is about 10 years younger (age 43 years) than the age of patients with posterior segment melanomas.99 The prognosis of iris melanoma is also relatively favorable compared to tumors of the posterior segment. Shields et al99 studied 169 patients with histologically confirmed iris melanoma and found that distant metastases developed in 5% at 10 years’ follow-up. The relatively small size of most iris melanomas probably is a major factor in their good prognosis. The prognosis of iris melanomas actually may be similar to posterior segment tumors of similar size and cell type.
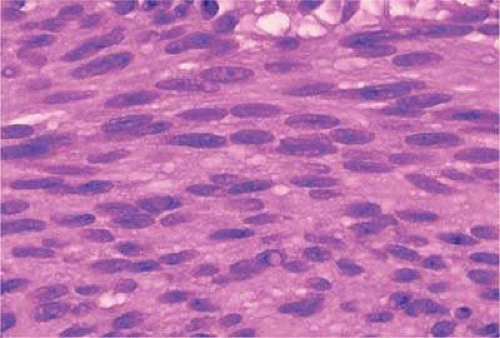 Figure 20.43. Low-grade spindle cell iris melanoma. Cytology is very bland. Only a few small nucleoli are present. (Hematoxylin-eosin X250.) |
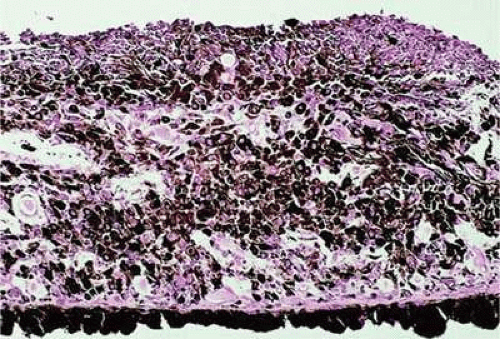 Figure 20.44. Iris melanoma, mixed cell type. Pigmented infiltrate replacing iris stroma contains many epithelioid cells. (Hematoxylin-eosin X250.) |
Diffuse iris melanomas that cause heterochromia iridis and secondary glaucoma are a rare but clinically important group of iris tumors.100,101 Many diffuse iris melanomas are higher-grade tumors that contain epithelioid cells, which are poorly cohesive and prone to aqueous dispersal. Patients are often misdiagnosed clinically and undergo filtering surgery for glaucoma. The latter invariably fails and puts patients at greater risk for extraocular extension and metastasis.101,102
Tapioca melanoma is a rare variant of iris melanoma with an usual clinical growth pattern marked by the presence of amelanotic nodules or spherules that have been likened to the pearl form of tapioca used to make tapioca pudding.103 Two types of tapioca melanoma are recognized. The more common solitary nodular variant resembles a conventional iris melanoma in most respects but has a multinodular tapioca-like surface. Multicentric or multinodular tapioca melanoma is a variant of diffuse iris melanoma characterized by multiple amelanotic or lightly pigmented tapioca-like nodules that are diffusely dispersed in the anterior chamber and iris surface. Affected patients typically present with severe glaucoma and may have heterochromia.
Prognostic Factors
About one half of patients with choroidal and ciliochoroidal malignant melanomas die from their tumors.104,105 Because the eye and orbit lack lymphatics, uveal melanoma spreads via the bloodstream. Metastasis to the liver occurs most often; more than 90% of cases with metastatic melanoma have liver metastases, and they are the first metastases detected in 80%. Other common sites of metastatic uveal melanoma include the lung (24%) and bone (16%).106 Multiple sites are found in 87%. Unfortunately, once distant metastases are manifest clinically, therapy generally is ineffective, and more than 50% of patients who have metastatic uveal melanoma die within 1 year. In recent years there has been an effort to identify prognostic factors that could identify patients at high risk for metastatic disease who, it is hoped, might benefit from prophylactic chemotherapy or immunotherapy.107
Standard Histopathologic Factors
The association between the cytologic characteristics of uveal melanoma (cell type) and mortality was initially reported in 1931 by Major George Russel Callender91 who examined a series of cases on file in the Registry of Ophthalmic Pathology at the Army Medical Museum in Washington, D.C. Callender reported that melanomas were composed of two types of spindle cells that he designated spindle A and B and less differentiated epithelioid cells. He found that tumors that contained epithelioid cells had a poorer prognosis.
Ian McLean et al92,108 at the Armed Forces Institute of Pathology (AFIP) modified Callender’s original classification in 1978. Spindle A and Spindle B melanomas, which formed separate categories in Callender’s original classification, were combined into the single category of spindle melanoma. Very low-grade spindle cell tumors were classified as spindle cell nevi. Mclean also introduced the concept of intermediate cells (i.e., cells with nuclear characteristics that were intermediate between spindle B and epithelioid).
The presence or absence of epithelioid cells is an extremely important prognostic factor in uveal melanoma. In 1982, Mclean et al105 reviewed a series of 3,432 cases of malignant melanoma of the choroid and ciliary body on file in the AFIP’s Registry of Ophthalmic Pathology and found that 56% were mixed cell tumors composed of a mixture of spindle and epithelioid cells. The 15-year mortality of patients with melanomas of mixed cell type was three times that of patients whose tumors were composed solely of spindle cells. Tumor size, measured as the largest tumor diameter (LTD), was also highly correlated with mortality.
Cell type remains one of the prognostic mainstays of surgical pathologists because assessment is relatively rapid and requires no special stains or equipment. However, application of the Callender classification unquestionably has limitations. First, determination of cell type is highly subjective and diagnostic accuracy can vary with the expertise and experience of the pathologist. A masked study showed that even experienced ophthalmic pathologists disagree about their classification of individual tumor cells.109 Second, melanoma cells constitute a continuous biologic spectrum that includes extremely bland spindle A melanoma cells at one end and highly anaplastic epithelioid cells at the other. Despite this, only three categories—spindle, mixed or epithelioid—are available for the classification of a given tumor, and tumors in a single category (e.g., mixed cell type, can vary significantly in their apparent degree of differentiation).
Extrascleral extension was found histopathologically in 8.2% of 1,527 enucleated globes with uveal melanoma evaluated in the Collaborative Ocular Melanoma Study85. Coupland et al110 found that extraocular spread correlates with increased mortality because it is associated with increased tumor malignancy and, in the case of posterior tumors, more advanced disease.
Morphometry and Nucleolar Parameters
The limitations of the Callender classification prompted a search for more objective and reliable criteria for the histopathologic assessment of the malignant potential of uveal melanomas. Gamel et al111,112,113 performed morphometric analysis on series of tumors with known survival and showed that certain nucleolar parameters, most notably the inverse of the standard deviation of the area of the nucleolus, were useful predictors of death from metastatic melanoma. Using the combination of nucleolar parameters and largest tumor diameter, Gamel et al114 were able to correctly predict the clinical course of 88% of cases.114 The technique subsequently was applied to 340 cases with known outcome from two independent laboratories and successfully subdivided patients into groups that suffered a sixfold difference in mortality.113,115
Gamel’s laboratory subsequently developed another simpler objective method of nucleolar assessment based on the measurement of the 10 largest nucleoli (MTLN).116 This was more reproducible than cell type and had substantial cost and labor advantages over the original computer-controlled system required to measure the standard deviation of nucleolar area (SDNA). The authors also used a digital filar micrometer to measure nucleoli. The latter method for measuring MTLN required <11 minutes per case, compared to an average of 28 minutes for the original method.117 The technique compared favorably with LTD, cell type, and SDNA in the prognostic assessment of a large series of patients with melanoma.118 Other researchers have had less success with MTLN, probably because of technical factors.119,120
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree