Intraocular Lenses
James A. Davison
Guy Kleinmann
Yoel Greenwald
David J. Apple
This chapter describes the evolution, chemistry, and clinical characteristics of pseudophakic and phakic intraocular lenses (IOLs). Surgical strategies and complications are also discussed, as well as future trends. Cataract surgery, secondary lens implantation, IOL power calculation, and pediatric lens implantation are discussed in other chapters.
HISTORICAL OVERVIEW
The history of the IOL is interesting and colorful. It is a classic example of the improvement of medicine with the active cooperation of science and industry. It involves a reciprocating but overlapping evolutionary relationship of cataract removal technology with IOL design. Cataract surgery evolved through extracapsular cataract extraction (ECCE), intracapsular extraction (ICCE), machine-assisted ECCE, phacoemulsification by external nuclear attack, and phacoemulsification-assisted internal nuclear disassembly. For IOL fixation, the evolution has been posterior chamber, anterior chamber (AC), pupil and iris, iridocapsular, ciliary sulcus, asymmetric placement, and capsular bag. As with any evolutionary process, this has been and still is a leapfrogging phenomenon, so that at any one point in time several cataract surgery strategies and IOL implantation techniques can be considered good science and good medicine. The process continues as microincision phacoemulsification procedures gain sophistication in search of an IOL to be inserted through a sub–2.0-mm incision.
Sir Harold Ridley
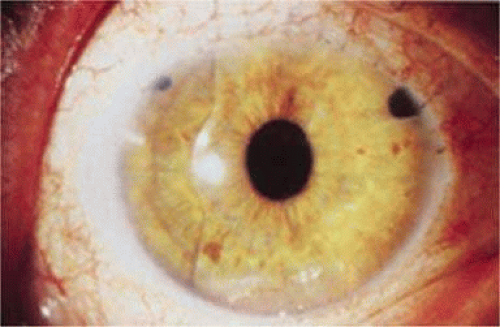
Credit for the invention and first implantation of the IOL is given to Sir Harold Ridley of London (Fig. 11-1)1,2,3,4,5,6,7,8 Details regarding Ridley and his invention are provided in a beautifully written and illustrated biography. Ridley’s first IOL surgery was accomplished as a two-step procedure. The ECCE was performed on November 29, 1949. He waited for the eye to become quiet and stable and implanted the IOL secondarily a few months later on February 8, 1950. During the next 12 years, approximately 1,000 Ridley IOLs were implanted. These operations were described as successes in 70%, failures caused by dislocation in 20%, and secondary glaucoma in 10%, which sometimes required explantation.9
Ridley had been inspired by the tolerance of British fighter pilots’ eyes to plastic fragments, which had lodged in them after their canopies, made of polymethylmethacrylate (PMMA; Perspex), had shattered. He worked with the Rayner Company and Imperial Chemical Industries, both in Great Britain, to develop Perspex CQ, a more purified “clinical quality” PMMA. He used the human lens as his model and selected similar radii of curvature to create a biconvex disc while using approximately half the thickness and weight (∼5 mm thick and 230 mg for the human lens). One of his original lenses made by Rayner, a 23.00 diopter (D), was measured at 8.5 mm in diameter and 2.4 mm thick, with a weight of 108 g. Modern analysis has demonstrated that this IOL passes modern optical bench examinations10 (Fig. 11-2).
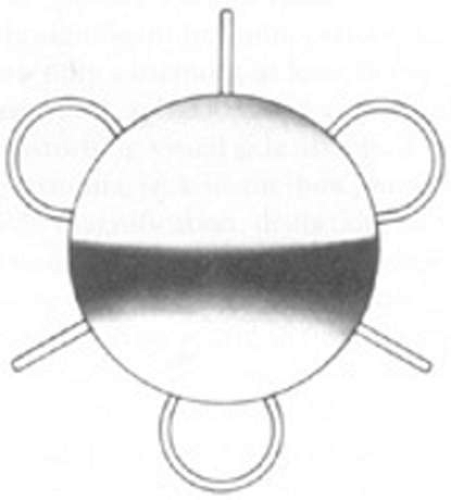
 Figure 11-2. This scanning electron micrograph (SEM) of an original Ridley IOL interestingly shows compound relatively square edges. |
Ridley said that the cataract operation “without a replacement lens was an incomplete, only half-finished operation”11 and that he would like to be remembered as the man who cured or at least initiated the cure for aphakia. He saw aphakic vision as a highly significant but unnecessary disability. Now only a memory, at least in the modern world, aphakic vision contained many disturbing visual side effects. The roving scotoma, jack-in-the-box phenomenon, 30% magnification, distortion, loss of side vision, and extreme spectacle dependence were disabling for many patients. Because of this, even as late as the mid-1970s, surgery was typically performed only when the patient’s vision decreased to 20/70 or worse in the better eye, with the fellow eye undergoing operation 3 days after the first eye.
Mr. Ridley performed the first intraocular lens implantation in the United States in Chicago in 1952 after delivering a lecture to the Chicago Ophthalmological Society. The audience included Dr. Warren Reese of Philadelphia, who immediately became enthusiastic about the idea. Mr. Ridley gave him five lenses at the meeting; Dr. Reese flew back to Philadelphia, and the next day became the first American to perform intraocular lens implantation.10 Despite that early beginning, it would take more than 25 years for IOL implantation to become the dominant and standard accepted method of curing aphakia in the United States.
The Ridley lens was placed in the posterior chamber after ECCE (Fig. 11-3). The anterior capsulectomy of the day was very large, and thus zonular support was poor. Some Ridley lenses dislocated into the vitreous because of poor zonular support and partially because of their weight, which was approximately eight times that of current IOLs.
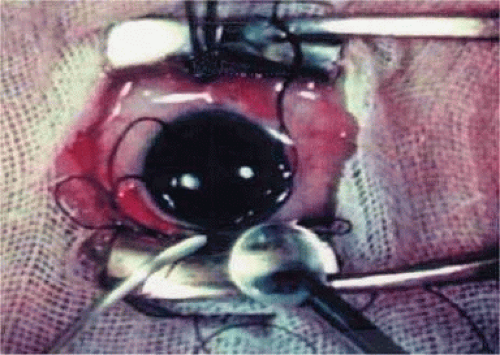 Figure 11-3. Photograph of Sir Harold Ridley’s intraocular lens implant operation taken from his original movie filmed during surgery. (Courtesy of David J. Apple, M.D., Charleston, SC.) |
Ridley’s achievements were finally and belatedly celebrated in numerous tributes. His first and perhaps most-prized honor was his election to the Royal Society in 1986. Dr. Apple presented him his first University Doctorate at the Medical University of South Carolina in 1988. He received another honor at the Science Museum in London on November 29, 1999, the 50th anniversary of the first part of the first IOL implantation. In the Flight Room, with airplanes suspended overhead, Ridley was honored by fellow pioneers and colleagues from around the world, as well as by the Rayner Corporation and government representatives from the United Kingdom and United States. The same year, the American Society of Cataract and Refractive Surgery (ASCRS) honored him as one of the 10 most influential ophthalmologists of the 20th century. In 1990, he was guest of honor at the Annual Meeting of the American Academy of Ophthalmology. In recognition of his unique efforts in IOL development and implantation, Ridley was knighted on February 9, 2000, by Queen Elizabeth II. On May 25, 2001, at the age of 94 years, Sir Harold died in Salisbury, England, after a cerebral hemorrhage.
Because of the difficulty with posterior chamber IOL placement, pioneering surgeons would spend the next two decades trying to find a better place to fixate the IOL. The AC lens, pupil-fixated IOL, iris-fixated IOL, and iridocapsular IOL would be placed in large numbers, only to return to the posterior chamber in the 1970s. This made for very interesting surgical residencies during these times. We were always learning something completely new. For example, during J.A.D.’s 3-year residency starting in 1977 at the Mayo Clinic, we evolved through cryoextraction ICCE using no IOL, cryoextraction ICCE with Medallion iris-sutured IOLs, machine-assisted ECCE with iridocapsular fixation using metal and then plastic clips through the iridectomy, and machine-assisted ECCE with posterior chamber IOL implantation.
Anterior Chamber and Pupil-Supported Lenses
Apple et al12 have described in detail the evolution of the IOL, describing six generations. AC IOLs were generation two.
The first AC IOL was implanted by Baron of France in 1952. This lens failed primarily because of excessive anterior vaulting, which caused contact with the corneal endothelium. Mr. Ridley’s good friend and long-time defender, Peter Choyce, also of the United Kingdom, was one of the developers and certainly the greatest champion of the AC lens (Fig. 11-4). These lenses could be placed after ECCE or ICCE procedures. His models were very successful with his last models, the Mark VIII and Mark IX, used until the mid-1970s in the United States.
Other AC lenses had problems as well. Ellingson’s uveitis, glaucoma, and hyphema syndrome13 was associated with their use. Chronic irritation of the delicate structures of the angle caused this problem, and even without it, pain could sometimes be elicited by simply touching the eye. Later, similar rigid AC IOLs would have similar problems. Some had to do with sizing difficulties; others were problems of poor finish and the misapplication of polypropylene haptics in the AC, where they underwent ultraviolet (UV) degradation.
Ultimately the flexible AC IOL was developed, most commonly the Kelman Multiflex (Fig. 11-5). This style of IOL features rather broad area smooth footplates, which can be placed in the angle without causing the chafe and erosion that small-sized loop-shaped haptic IOLs did. The Multiflex-type lens has provided excellent performance with similar and sometimes lower long-term corneal endothelial cell loss in secondary implantation than has the sutured ciliary sulcus posterior chamber IOL.14
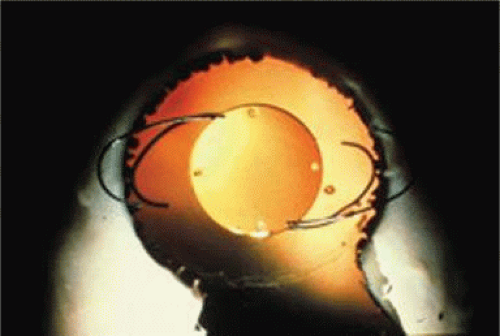
Taking another path, Cornelius Binkhorst of Holland developed a lens that involved pupil fixation with pairs of horseshoe-shaped haptics in front of and behind the iris (Fig. 11-6). This lens was associated with total dislocation into the AC or vitreous after pupil dilation, so miotics were many times given on a prophylactic basis indefinitely. Also, after ICCE, there was so much iridodonesis, especially with the initially used metal haptics, that the anterior aspects of the anterior haptics could touch the endothelial surface of the cornea, leading to localized corneal decompensation. Also, the weight of metal haptics could erode the iris. It was this style of lens that was implanted by early American implant surgeons Jaffe, Hirschman, Byron, and Kwitko in 1967.15
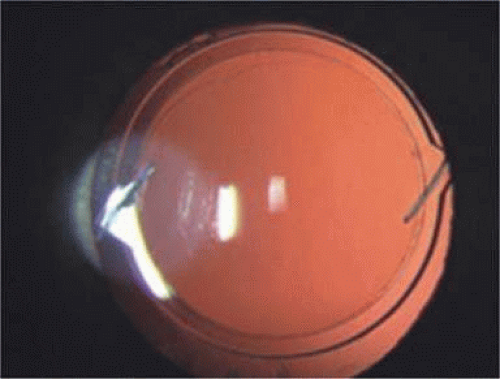
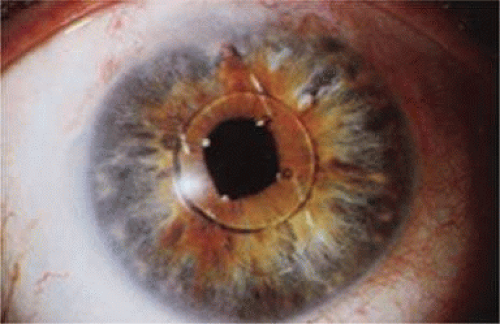 Figure 11-6. Binkhorst lens with PMMA haptics in front of and behind the iris. A Prolene suture has been placed through the superior anterior haptic to prevent dislocation. |
Introduced in 1967, Stanislav Fyodorov’s “Sputnik” IOL design achieved stabilization without larger anterior haptics (Fig. 11-7). In the mid-1970s, the Worst medallion IOL also featured an anterior optic with two horizontally oriented horseshoe-shaped looped posterior haptics similar to the Binkhorst structure. A Prolene suture was passed horizontally through the superior iris and then threaded between through two small holes in the superior optic. Because iris suture had to be preplaced, it always seemed to become entangled with the haptics during insertion, making it difficult to place smoothly under air after ICCE, which featured a 180-degree corneal incision and an unprotected anterior hyaloid membrane. The suture was tied loosely so that the optic was secured to the superior iris. This would still allow the posterior haptics to dislocate anteriorly (embarrassingly, many times after engaging in sexual activity) or create a partial pupillary capture, but at least it prevented total dislocation. Because surgical manipulation was expensive and carried the risk of infection, we would spend hours positioning patient’s heads and bodies after pharmacologic weak dilation so that gravity would reposition the IOL. Then, when the IOL fell into position, we would reposition the patient and administer a topical miotic to capture the appropriate part of the IOL with the pupil. Aside from partial dislocation, this type of lens performed very well, but eye movement after ICCE still generated substantial iridopseudophakodonesis.
ICCE was difficult and time-consuming and carried a high risk. Even when done well, the procedure left an eye not able to support an IOL in stable fashion. The structural diaphragm of the posterior capsule after ECCE was rediscovered and appreciated because it contained the vitreous and created compartmentalization and stabilization of the AC, iris, and posterior chamber. Iridocapsular fixation was the next step. The lenses designed for this technique had a small platinum or plastic rod attached to the superior optic that could be clipped to the superior posterior haptic through a superior peripheral iridectomy (Fig. 11-8). The inferior haptic would be inserted, and eventually fibrose, between a leaflet of remaining anterior capsule and the posterior capsule. When properly secured, the pupils of these eyes could be dilated without fear of superior or inferior IOL dislocation. Because of capsule fibrosis and stabilization, there was also a substantial reduction in pseudophakodonesis. Surgeons who had taken up phacoemulsification, which had been introduced in 196716 and adopted by a fair number by 1972, could enjoy the small-incision control aspect of cataract removal but still had to enlarge the incision for IOL insertion. They were able to preserve larger leaflets of anterior capsule to make capsular fixation possible in both haptics (Fig. 11-9). Things were improving.
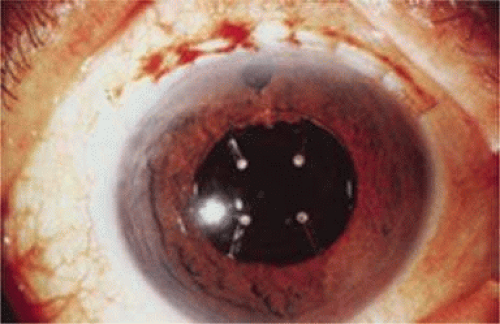 Figure 11-8. Worst iridocapsular lens with PMMA clasp clipped through the peripheral iridectomy to the superior posterior haptic. |
Return to Posterior Chamber Lenses
John Pearce of the United Kingdom returned implantation to the posterior chamber by developing a rigid tripod-shaped PMMA IOL designed for implantation there,17 but it was Steve Shearing who would have the greatest influence on the future development of posterior chamber IOLs. In March 1977, he introduced his IOL, which featured flexible posterior haptics (Fig. 11-10).18 His original intention was to have the haptics placed within the capsular bag remnant. The lens had a 5-mm optic (because that was the size of the Binkhorst optic, which was already in production at the time) and an overall length of 12 mm and was flat. Of course, the potential value of the patents of the day was substantial. So much so that another ophthalmologist claimed that he had already implanted an IOL that looked like that described in Dr. Shearing’s patent for the flexible posterior J-loop haptic. He said that he had cut off a portion of both of the posterior haptics in a Binkhorst-style lens, thus modifying it to look like the “J” appearance.
That conflict was resolved by exhuming the body of the patient in whom he had claimed to have implanted the modified lens. But after exhumation, an autopsy failed to support his assertion that he had used amputated haptics and his claim was dismissed (personal communication, Steve Shearing, 2004).
This lens design revolutionized the concept of IOL placement, but the need for a supportive capsular bag architecture was not fully appreciated, nor had the techniques to preserve it been developed. Most dislocations were incomplete (i.e., nonintravitreal). For example, the “sunset syndrome” (Fig. 11-11) resulted from occult anterior radial capsular tears (ARTs), which extended through the equator or inferior zonular disruptions and allowed the inferior haptic to sink through the defect. However, in most inferior dislocations, the IOL optic was still contained within the ciliary sulcus. The IOL’s overall length was too short for symmetric sulcus fixation, so the whole IOL structure gravitated inferiorly with sulcus placement. The “windshield wiper syndrome” resulted from sulcus contact inferiorly but none superiorly. Surgeons tried to retain at least a leaflet of anterior capsule inferiorly in which to place the inferior haptic. When that was accomplished, the IOL stayed fairly well centered. However, if asymmetric placement occurred with the inferior haptic contained within the capsular bag remnant and the superior haptic loose in the ciliary sulcus, there would be opportunity for another type of decentration, the “sunrise syndrome.”
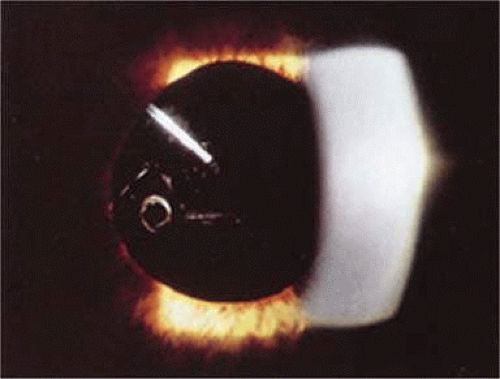 Figure 11-11. Sunset syndrome with posterior chamber IOL sinking through an inferior zonular defect. |
Because the capsular bag was such a hard place to find, the IOL was soon modified to include a 6-mm optic and 13-mm overall length. Even then, fixation was not perfect, and pupillary capture, just as Dr. Shearing had experienced in his fourth case, was not rare (personal communication, Steve Shearing, MD, 1998).
However, an important step had been taken, and during the next two decades, the Shearing posterior chamber IOL would evolve into three basic styles. The first was designed primarily for ciliary sulcus fixation but could also be used for capsular bag fixation. It was the result of the tremendous influence of two popular, committed, prestigious California phacoemulsification surgeons who had trained together at Duke University, Dick Kratz and Bob Sinskey. The second was the short haptic diameter modified C-loop IOL designed for capsular bag placement. The third featured larger haptic and optic diameters and was designed for surgeons still practicing planned ECCE who might place the IOL within the sulcus or asymmetrically with one haptic in the sulcus and one in the capsular bag.
The Kratz IOL was compatible with Kratz’s phacoemulsification technique, which involved a prolapse of the superior nuclear pole into the iris plane for external attack phacoemulsification (Figs. 11-12–11-14). In this technique, remaining anterior capsule was a problem because it could produce asymmetric placement and decentration. Therefore, anterior capsulotomies were large so that the IOL could be placed in the ciliary sulcus. Kratz developed a “tap test” that involved tapping the eye over the ciliary sulcus with a Weck sponge to see whether the IOL would move, which would indicate that the haptic was in contact with the structures of the ciliary sulcus and not hung up in the capsular bag remnant. Through the Precision Cosmet Company (Minneapolis, Minnesota), Kratz introduced a 10-degree optic posterior angulation as an effective way to prevent pupillary capture. The resistance to compression of the haptics was softened significantly by a side optic mounting modification with the overall length of 13.5 mm. Actually, before that, more than a few surgeons had been bending the Shearing haptic at the optic haptic junction to try to reduce compression resistance. Sinskey’s lens was very similar and was introduced at about the same time but by a different company, IOLAB (San German, Puerto Rico). Because of the nearly simultaneous introduction and similarity, the lens ultimately has been called the Kratz-Sinskey IOL (Fig. 11-15).
 Figure 11-15. The Kratz-Sinskey IOL involved haptic anchoring to the optic sides and a more gentle modified J-loop haptic. |
Sinskey’s phacoemulsification technique was basically one-handed and allowed a more generous anterior capsular remnant to exist after nucleus removal. Symmetric capsular bag fixation was possible a fair amount of the time. Optic posterior angulation was incorporated in his lens to prevent pupillary capture, which was still possible.
The concept of intracapsular phacoemulsification and the desire to fixate the IOL within the capsular bag ultimately led to the second type of posterior chamber IOL, the minimally compression-resistant, short haptic configuration created for capsular bag fixation that exists today.19
The third style was a transitional lens that was oversized and was to be used with planned ECCE. It eventually measured 14 mm in haptic diameter and 7 mm in optic diameter. It was large and stiff in its eventual one-piece design. The problem was that after ECCE, the capsular bag did not exist in a structural sense. With broad contact, at least one haptic would be captured by some partially unrolled capsular flap remnant, usually the inferior. If it were asymmetrically placed, which was the expectation, the lens was so long that capsular fibrosis would not decenter the very large optic too much because of the contact created in the superior ciliary sulcus by the high resistance to compression. Because of their large size, these lenses were very difficult to place symmetrically within the capsular bag but are still in use today as ciliary sulcus–placed secondary IOLs in patients with very large eyes.
As an aside, glass IOLs were marketed for a short period. Even in aqueous, they were heavier than plastic. Glass also broke if hit by the neodymium:yttrium-aluminum-garnet (Nd:YAG) laser (Fig. 11-16).20 Plastic IOLs inadvertently hit by the YAG laser during capsulotomy developed small cracks or pits that did not affect vision and did not completely fracture. Glass was eventually abandoned because of its weight and YAG intolerability. The right to use polyamide framing and haptic material was purchased from Lynell Optics by STAAR Surgical (Monrovia, CA) and is in use today by that company as a haptic material combined with silicone optics.
Endothelial Cell Damage, Viscoelastics, Anterior Capsular Tears, and Sutureless Closure
With the introduction of clinical corneal endothelial photography in 1976, Bourne and Kaufman21 heightened our awareness of quantitative and qualitative endothelial damage associated with IOLs. In 1980, Miller and Stegmann22,23, working with the Pharmacia Company of Uppsala, Sweden, introduced the first viscoelastic, 1% sodium hyaluronate (Healon). This not only protected the corneal endothelium during IOL implantation but also made anterior capsulotomy much easier to perform. The control of the anterior capsular surface with viscoelastic, pushing it back and making it flat, was an important aid in the prevention of unwanted ARTs during the can-opener capsulotomy process.
The number of central corneal endothelial cells after IOL implantation decreases at rates greater than those of healthy unoperated corneas 24,25,26,27,28,29 (normal loss ranged from 0.3%–1.0% per year).
The only randomized trial of lens implantation, the Oxford Cataract Treatment and Evaluation Team, found a higher rate of cell loss in eyes with implants than in those without in the first 3 postoperative years.30 Ultimately, the type of surgical technique and IOLs (ICCE and Binkhorst four-loop lens) are no longer being used.
A 5-year prospective study from the Mayo Clinic31 reported 23% to 28% endothelial cell loss after cataract surgery over the 5-year postoperative period. The rate of cell loss was not found to be influenced by the surgical technique (ICCE vs ECCE), implantation of an IOL, and type of IOL implanted (medallion iris suture IOL, transiridectomy clip implant, or posterior chamber IOL). Correlation was found between the endothelial trauma judged at surgery and the long-term endothelial cell loss. Extension of the follow-up time to 10 years32 demonstrated that the eyes continued to lose endothelial cells from the central cornea at an average rate of 2.5% per year (2.5–8.0 times the rate in healthy unoperated eyes); also in this study the type of IOL implanted did not influence the rate of cell loss. These studies had two major caveats: First, a significant percentage of patients were lost to follow-up; and second, the study represents the early cases in the implant experience of the surgeons (1976–1982) and thus does not represent the current surgical technique and IOL technology.
In the early days of the phacoemulsification, the corneal cell loss rate was high because of the long phaco time and high energy that were commonly used. In recent years, damage to corneal endothelial cells during cataract extraction has been minimized as a result of better instrumentation,33,34 newer viscoelastic materials,35,36,37 and improved surgical technique such as phaco chop,38 which aims to reduce machine-measured phaco time.
Several preoperative and intraoperative parameters can influence the risk for endothelial cell loss after phacoemulsification. A high nucleus grade,39 old age, long phaco time,40 and short axial length41 are associated with an increased risk for endothelial cell damage. Ravalico et al42 reported that AC IOL implantation did not appear to alter corneal endothelial function during 5.2 years of postoperative follow-up period. As a summary, it seems that the endothelial cell loss after cataract surgery is primarily related to the surgical process, maybe to the absence of the crystalline lens, and in some but not most situations to the intraocular lens itself.
Surgeons who performed one-handed phacoemulsification were preserving the capsular bag better than two-handers. In 1981, John Graether,43 a left-handed one-hander, designed the first one-piece all PMMA IOL that was 12.25 mm in overall length designed specifically for implantation in the capsular bag (Fig. 11-17). He had invented a “collar button” retractor and described a way to use it by pushing on the superior optic-haptic junction to compress the inferior haptic and dial the IOL into the capsular bag, the method still used today.43 Later, a reduction in the amount of nuclear prolapse into the iris plane made it possible for surgeons who used the two-handed procedure to retain more anterior capsule by creating only one anterior radial tear (ART) most of the time.44 Even if two superior tears were noted, posterior chamber IOLs could be placed in what was left of the capsular bag with minimal or no decentration.45 Sometimes lenses with broader haptics, as designed by Bill Simcoe, were used to bridge the gap created by ARTs in the capsular bag equator. Even though surgery and implantation had improved, significant IOL optic decentration, with its attendant optical and physical complications, was not uncommon (Fig. 11-18). Given the anatomy of the eye with possible multiple ARTs after iris-plane phacoemulsification or ECCE, two schools of thought developed. The first embraced larger optic and haptic diameters, which provided greater stability and increased resistance to compression and were designed in an attempt to center IOL optics within the capsular bag, ciliary sulcus fixation, or asymmetric fixation and prevent pupillary capture (Fig. 11-19). The other school fashioned 6.0-mm optics and reduced 12.0-mm haptic diameters with a very low resistance to compression to try to keep them within the capsular bag or capsular bag remnant (Figs. 11-20 and 11-21).19 In an attempt to custom tailor IOLs to patient anatomy, J.A.D. created the “graduated length method” through the D.G.R. Company of Clearwater, Florida, in 1990. This IOL line featured shorter 12.0-, 13.0-, and 14.0-mm haptic structures for small, medium, and large eyes.
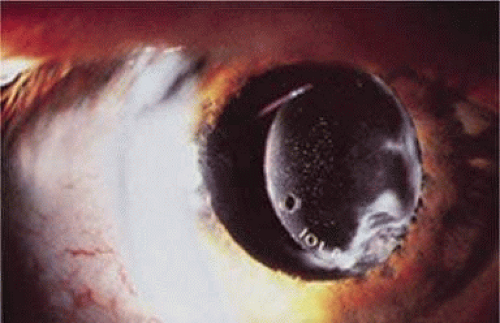 Figure 11-18. Asymmetric bag-sulcus posterior chamber lens fixation with capsular remnant and anterior optic precipitates. |
Because of these compounding developments, David Apple began his important work with Randy Olson and in 1983 founded the Center for Intraocular Lens Research at the University of Utah (Fig. 11-22). They were a uniquely qualified combination. Dr. Apple was board certified as both a pathologist and ophthalmologist; Dr. Olson was an academic ophthalmic surgeon. Much like Harold Ridley and Peter Choyce, they began their dedicated quest to improve the condition of pseudophakia. They solicited autopsy specimens and wrote innumerable articles about IOL design, centration, and complex IOL-ocular interactions. One of their most important articles, published in 1985, was a position article citing the advantages of capsular bag placement and recommending it over ciliary sulcus placement, still a controversial issue at the time.46 The eyes submitted to them that year demonstrated capsular bag fixation in 31%, ciliary sulcus fixation in 11%, and asymmetric bag–sulcus fixation in 58%.47 At that time, 85% of surgeons still preferred planned ECCE as their surgical technique.48
While in Utah, Apple and Olson saw the problems created by ARTs solved with the invention of capsulorrhexis, almost simultaneously reported in 1986 by four surgeons from around the world. Two presented articles at the Welsh Cataract Congress in Houston that year, and two reported their techniques independently. The discovering surgeons in alphabetical order are Drs. Calvin Fercho (Welsh Cataract Congress, Houston, 1986), Howard Gimbel (video presentation at the annual meeting of the ASCRS in Boston, 1985),49 John Graether (Welsh Cataract Congress, Houston, 1986) (Fig. 11-23),50 and Thomas Neuhann (video presentation at the meeting of the German Ophthalmological Society in Heidelberg, Germany, 1985).49 With this technique, and the creation of an approximate diameter of 5 mm, symmetric placement of any IOL could be guaranteed. It still took several years for most ophthalmologists to incorporate continuous curvilinear capsulorrhexis (CCC) into their surgical routines.
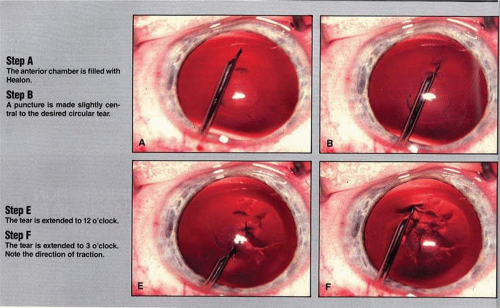 Figure 11-23. From John Graether’s original 1986 Ocular Surgery News (OSN) article describing anterior continuous curvilinear capsulotomy. (Courtesy OSN and John Graether.) |
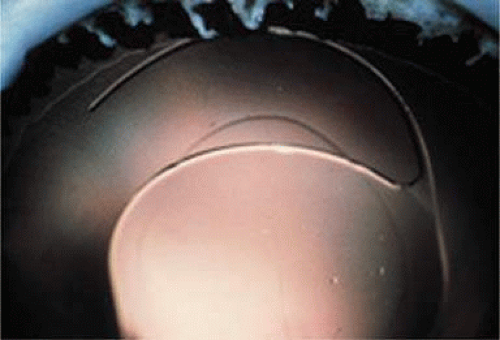
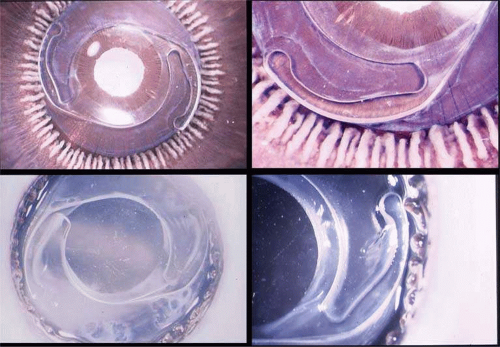
In 1988 Dr. Apple relocated the laboratory to the Storm Eye Institute at the Medical University of South Carolina in Charleston. Reflecting new challenges, his laboratory name would be changed to the Center for Research on Ocular Therapeutics and Biodevices. From there, he and his staff, residents, and research fellows continued to receive autopsy specimens from around the world and eventually demonstrated, in the largest autopsy specimen study to date, a decline in asymmetric placement to only 10% of eyes with foldable lenses submitted in 1998.47 Early in his work there, Dr. Apple recruited Dr. Kensaku Miyake’s retrociliary photographic analysis method to locate and analyze IOL placement within the eye (Fig. 11-24). With increasing video sophistication and use of the process by Dr. Apple and his colleagues, Dr. Miyake himself generously expanded the name of the procedure to be called the Miyake-Apple technique.
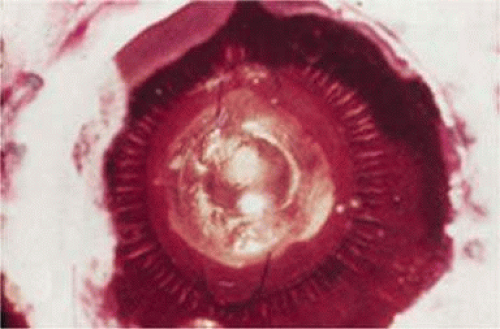 Figure 11-24. First picture from Dr. Miyake of a Shearing lens in the ciliary sulcus. Note the profound capsular bag shrinkage and abundant ring of Soemmering. |
After cancer was diagnosed in Dr. Apple and he was successfully treated, the center was relocated to Salt Lake City in 2002, where it has been permanently designated as the David J. Apple, MD, Laboratories for Ophthalmic Devices Research.
In the days when the laboratory was in Charleston, Dr. Apple worked with industry representatives and surgeons to refine IOL design to ensure that capsular bag residence would be as consistent as possible, thereby reducing lens contact with other eye structures in both routine and complicated situations (Figs. 11-25 and 11-26). J.A.D. had the great pleasure of working with him in his laboratory to help improve an already sophisticated haptic configuration in a one-piece all-PMMA IOL. At that time, we thought that the entire haptic should be C-shaped so that even its distal end could be recruited for capsular equatorial support (Fig. 11-27). We studied resistance to haptic compression, attempting to make it softer and more uniform through diameter reductions from 13.0 to 11.5 mm (Fig. 11-28). These efforts contributed to the development of the Pharmacia model 811, which, along with others of its day, may have represented the height of single-piece PMMA IOL development (Fig. 11-29).51
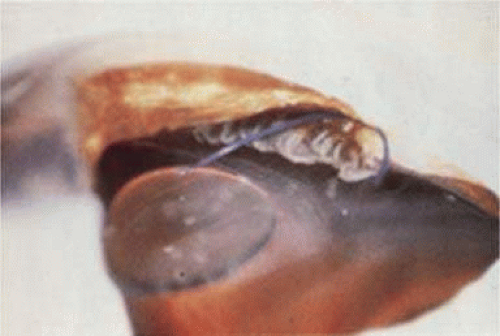 Figure 11-25. Sulcus placement required long haptic structure and posterior angulation to avoid pupil capture. |
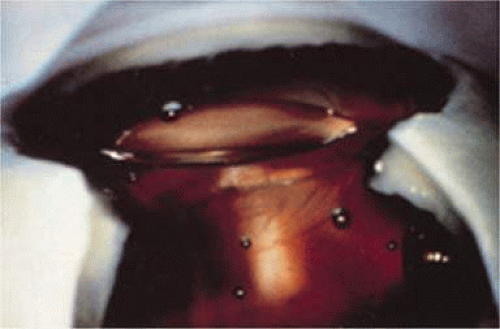 Figure 11-26. The shallow disc of a posterior chamber IOL fills only the anterior portion of the capsular bag. |
Inevitably, with the new technique of capsulorrhexis, the improved haptic architecture was actually not as critical as it had been earlier, when ARTs were universal. It was still helpful in difficult situations in which capsular support was anomalous (e.g., pseudoexfoliation and trauma). Coatings of heparin and Teflon were developed to increase the biocompatibility of PMMA IOLs.52 However, the benefits of the sophisticated one-piece PMMA capsular bag design would soon be replaced by the even greater benefits of foldable IOLs, the use of which would overtake PMMA in 1998.48
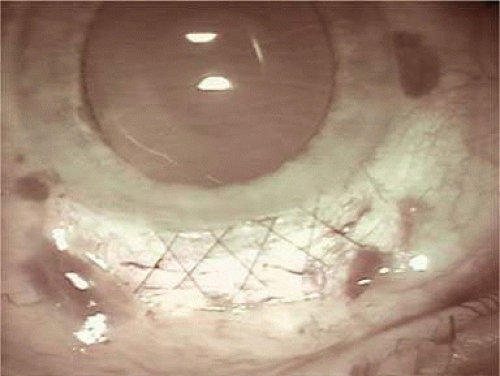
Sutured superior corneoscleral incisions with round PMMA IOLs were standard throughout the 1980s (Fig. 11-30) until 1991, when Mike McFarland53 discovered that they could seal themselves without sutures if properly constructed. In an attempt to integrate the smaller phacoemulsification incision and PMMA technology, ovoid posterior chamber IOLs had been produced since 1980 and reached their peak in popularity (35%) in 1991 (Fig. 11-31).48 But, because of their truncated edge, ovoid IOLs introduced a higher incidence of glare, streaks, and halos54 (pseudophakic dysphotopsia); for this reason, they never reached great popularity.
Foldable Lenses, Clear Corneal Incisions, and Topical Intracameral Anesthesia
In 1984, Mazzocco et al55 introduced the first foldable plate haptic silicone lens produced by STAAR Surgical. It became known as the “Mazzocco taco” because of the way it appeared when folded. It was delivered through an injector, whose cartridge went through a 3-mm incision, allowing the IOL to unfold in the posterior chamber (Fig. 11-32). This changed cataract surgery forever because it made possible the full appreciation of phacoemulsification. Cataract surgery had become the first microscope-accomplished, machine-assisted small-incision surgery in medicine. All of the benefits of improved surgery safety, improved postoperative state quality, and shorter patient recovery were realized.
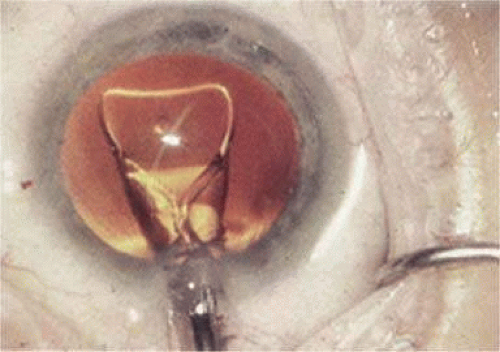 Figure 11-32. Plate haptic silicone lens unfolds into the capsular bag after it leaves the injector cartridge. |
This IOL did not fixate well in the sulcus, and because of currently evolved surgical technique, asymmetric bag–sulcus fixation was the standard of the day, which did not work for this lens either. An intact or almost intact (one or two opposing ARTs) capsular bag was necessary for acceptable centration performance. Concerned about this, the designers made the earliest models too long, so that when they were well fixated within the capsular bag they could actually wrinkle centrally because of capsule contraction, causing the “Z phenomenon.” The silicone material was hydrophobic and did not bond to the capsule during fixation, so that if asymmetric placement occurred, it could be squeezed in one direction out of the bag. Significant decentration was likely when only anterior capsular leaflets remained for fixation. Intravitreal dislocation was also possible after YAG laser capsulotomy.
The revolutionary and long-anticipated marriage of small-incision phacoemulsification surgery and small-incision IOL implantation had finally been consummated This created an accelerated reciprocating increase in popularity for the complementary technologies, especially in the United States. Ultimately ease of implantation through a cartridge using an injector and relatively low price would continue to sustain the IOL’s popularity over the next two decades.
Next, three-piece foldable silicone optics with polypropylene haptics emerged. These IOLs centered better and could be used in cases with ARTs. Popular small-incision cataract surgery had finally arrived. Surgeons no longer had to enlarge the incision much after cataract removal to accommodate the IOL, so one of the main advantages of phacoemulsification could be realized. However, increased AC reaction, capsular fibrosis, and optic decentration compared with single-piece PMMA IOLs kept some surgeons from using early three-piece silicone IOLs.56
The objections to silicone optics were obviated by another Charles Kelman invention, the introduction of the Phaco Fit PC-28LB IOL manufactured by AMO,57 which featured opacified winglike sections constituting two of the edges of its optic. (Fig. 11-33) Two side portions of the optic had opaque silicone wings, which would fold on top of the PMMA central optic during insertion through a 3.5-mm incision and then unfold in the eye. It was hoped that this design would reduce the edge glare seen with ovoid PMMA IOLs. Many years later, Howard Fine would recommend using such a textured finish in plate haptic silicone IOL haptics, not for optical reasons, but to enhance capsular fixation to prevent IOL movement within the capsular bag.
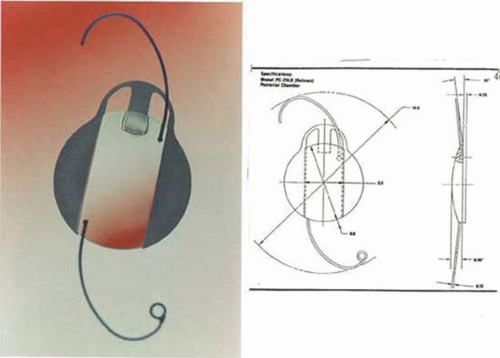 Figure 11-33. AMO Phacofit IOL designed by Charles Kelman featured translucent wings that folded over the clear optic for small-incision insertion. (Courtesy Abbott Medical Optics.) |
In 1992, Kimya Shimizu reintroduced a clear corneal incision only 3 mm wide to accommodate phacoemulsification and foldable IOL insertion.57 One suture was placed. That same year, Howard Fine showed that a properly constructed temporal clear corneal incision could be left unsutured and consistently perform well, with an extremely low incidence of incision complication and very little astigmatic consequence58 Although this initial incision was thought not to be as strong as one created with an additional scleral shelf,59 it was improved over the years by studying its structural integrity (Paul Earnest et al in ASCRS60) and reducing its width to 2.4 mm (Figs. 11-34–11-37). Foldable silicone lenses could be placed without using scissors, cautery, or sutures.
Cataract surgery using only topical anesthesia was introduced by Richard Fichman in 1992 and was improved with the addition of intracameral anesthesia by Jim Gills61 in 1995. Surgery was not very easily accomplished at its introduction in 1992 because most surgeons were still using superior corneoscleral incisions. But it quickly became the procedure of choice with the adoption of temporal clear corneal incision.
Another landmark in IOL history occurred in 1995 when the foldable acrylic IOL (AcrySof, Alcon Surgical, Fort Worth, Texas) was approved for use in the United States. In 2000, the single-piece AcrySof IOL was introduced (Figs. 11-38 and 11-39). The easily deformable low resistance to compression haptics (Fig. 11-40) made insertion easier through the 2.4-mm incision using the Monarch D cartridge (Fig. 11-41). The bulky 0.4-mm-tall three-dimensional haptics (Fig. 11-42) would provide resistance to rotation within the capsular bag, making it an ideal platform for its toric iteration that would be introduced in 2005. Other manufacturers would follow with their versions of three and single-piece foldable acrylic IOLs (Fig. 11-43). With self-sealing temporal clear corneal incisions, topical intracameral anesthesia, and foldable acrylic IOLs available, surgeons who had objected to foldable lenses made of silicone found their last obstacle to small-incision surgery removed. However, the acrylic lens had to be withdrawn temporarily because of glistenings within the optic.62 These turned out to be water vacuoles, which were associated with packaging materials. When the lens was repackaged, the glistenings were reduced.63 Through manufacturing improvement, the glistenings continue to be reduced to lower levels, but they still may be present in 60% of cases to small degree, but without visual consequences.64
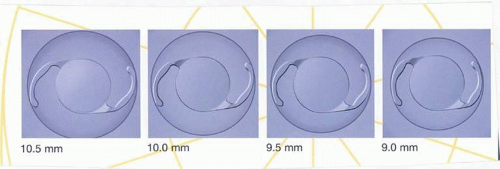 Figure 11-40. The AcrySof single-piece IOL features haptic structure with low resistance to compression at all capsular bag diameters. (Courtesy Alcon Surgical.) |
 Figure 11-42. SEM of AcrySof single-piece IOL components including detail of the optic-haptic junction and distal haptic. |
An important part of the evolution of the IOL has been the propagation and dissemination of information as well as the continuous development of educational processes, ensuring that the latest techniques and materials were available to surgeons. In the 1960s and 70s, the use of intraocular lenses was very controversial and they were not discussed in any formal academic setting. To learn more from each other and advance medical science, the International Intraocular Implant Club was founded and its first meeting held on July 14, 1966, with Mr. Ridley presiding in the United States. IOLs were almost removed from the market through a U.S. Senate committee hearing process in the late 1970s; patients and doctors testified, and fortunately, availability continued. There was a tremendous need in the United States for a forum to provide the free exchange of information on cataract surgery and IOLs. Recognizing that need, the American Intraocular Implant Society was founded in 1974 by its first president, Kenneth Hoffer, M.D., of Santa Monica, California (Fig. 11-44). Even though he had been in practice for only 2 years, he had the vision to recognize that the open and unrestricted sharing of thought and personal experience without bias or preconception was key to the development of modern IOL science. The Implant Society was a tremendous success and in 1985, its name was changed to the American Society of Cataract and Refractive Surgery. In 1988 its annual meeting outgrew its traditional location, the Century Plaza Hotel in Century City, California. In 1996, the organization combined its journal publication with that of the European Society of Cataract and Refractive Surgery. It. has grown to 5,000 U.S. and 2000 international members. The open climate of information sharing of the ASCRS and ESCRS has benefited science and patients greatly.
Cataract Surgery 2010
The changes in cataract removal technique and IOL implantation have been gradual, with considerable overlap of individual preferences. As can be seen in any evolutionary activity, there is never just one right answer. That is, at any one point in time, there exist multiple materials and surgical techniques that have their individual and combined inherent advantages and disadvantages. In fact, as of 2004, there were 1,548 IOLs and nonoptical implants from 33 different manufacturers available to surgeons.65 In 1998 many surgeons still preferred PMMA (33%) and superior corneoscleral incisions, but momentum was shifting away from those methods toward the use of silicone (22%) and foldable acrylic (42%)19 and clear corneal incisions. By 2003, PMMA had decreased to 6%, silicone had stabilized to 21%, and foldable acrylics had grown to 69%.66 Acrylic IOLs have evolved and are now made by manufacturers in addition to Alcon including Rayner, the manufacturer of the original Ridley lens (Fig. 11-45). The Alcon acrylics continue to evolve as well (Fig. 11-46).
 Figure 11-45. The Rayner single-piece IOL has architectural characteristics that are similar to the AcrySof single-piece and Graether Scroll Flex IOLs. |
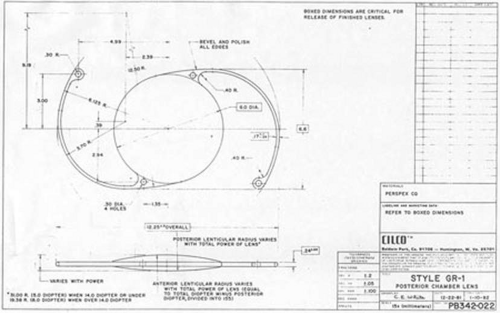
Substantial industry consolidation has occurred so that, in the United States at least, there were four major manufacturers with commercially available IOLs in 2004: Alcon Surgical of Fort Worth, Texas; Abbott Medical Optics of Santa Ana, California (AMO); Bausch and Lomb of Clearwater, Florida; and STAAR Surgical of Monrovia, California. Although many company executives and factory representatives from earlier companies have been integrated into the contemporary organizations, gone are the days of the prominent IOL lines of the 1980s and 1990s: CooperVision, Ciba Vision, O.R.C., Cilco, IOLAB, Precision Cosmet, Lynell Optics, and Pharmacia.
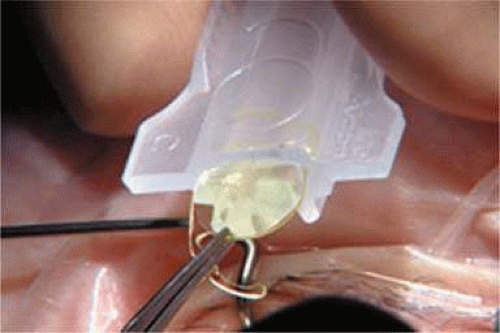
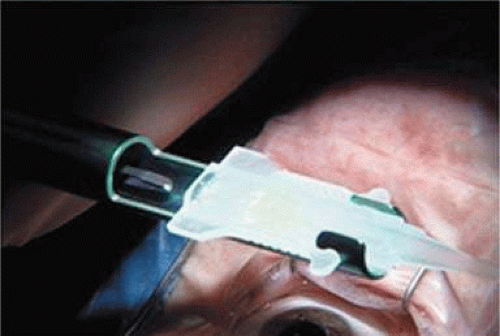
Modern lens replacement surgery consists of removal of the nucleus, cortex, and as much remaining lens epithelium on the posterior capsule as possible, while avoiding other ocular structures. It should leave a central circular anterior capsular opening so that the anterior capsule remnant can overlap the peripheral IOL optic by approximately 0.25 to 0.5 mm for a complete 360 degrees. Surgery is usually accomplished under topical intracameral anesthesia through a temporal clear corneal incision of less than 3.0 mm in length, permitting a foldable IOL to be placed using an adjunctive viscosurgical device. If well executed with either forceps (Fig. 11-47) or more commonly with an injector using a disposable cartridge (Figs. 11-48–11-50), this strategy provides good IOL centration and minimal unwanted side effects regardless of the foldable IOL material or design used (Figs. 11-51 and 11-52). Preloaded injectors are becoming available (Fig. 11-53) and will eventually be driven by a device connected to the phacoemulsification machine.
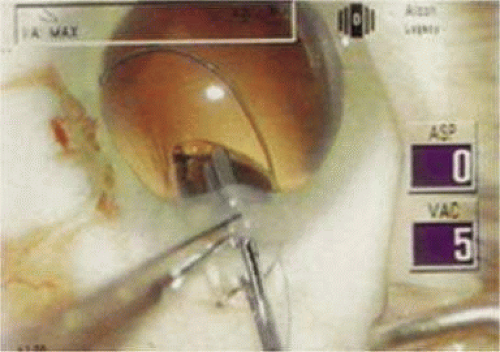 Figure 11-47. Using a Buratto direct-action forceps, the acrylic lens is placed through a self-sealing temporal clear corneal incision. |
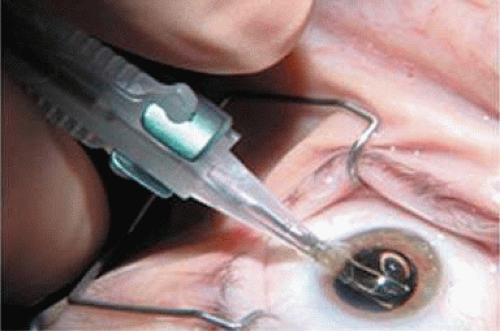 Figure 11-50. The curled SN60WF opens slowly as it is injected through the end of the cartridge to the capsular bag. A small air bubble is visible just to the left of the IOL. |
INTRAOCULAR LENS MATERIAL CHEMISTRY
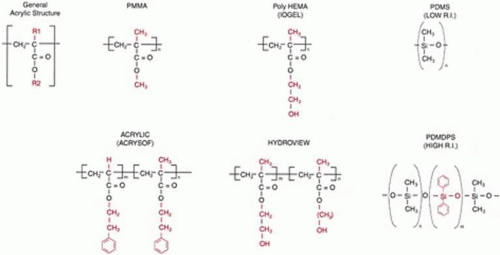
Various materials have been used for IOL optics. Excluding glass, the materials used for IOL manufacture have been several types of plastics, also known as polymers. The name polymer is derived from Greek poly (many) and mer (unit). A polymer is a long chain structure, composed of many units. The properties of polymers are derived both from the units in the structure and the relation of the chains to each other.
Polymerization is the process by which the repeat units (the monomers) that form a polymer are linked by covalent bonds, which produces a stable bond between each pair of monomers. For example, the methyl methacrylate monomer is used for the manufacture of PMMA. PMMA (poly[methyl methacrylate]) is a rigid, linear acrylic polymer. When different monomers are polymerized together, the process is called copolymerization. Three-dimensional, flexible acrylic polymers can be created by using appropriate monomers and a cross-linker that connects polymer chains together. Each currently available foldable acrylic lens type is manufactured from a different acrylic copolymer, with different refractive index, glass transition temperature (above which the polymer exhibits flexible properties and below which it remains rigid), water content, mechanical properties, and so forth.
The polymeric materials currently used for the manufacture of IOL optics can be divided into two groups based on their flexibility: rigid versus flexible. Within the flexible group, the subgroups of materials are silicones, hydrophobic acrylics and hydrophilic acrylics (Fig. 11-54).
RIGID MATERIAL
The rigid material for IOL optic usage is PMMA. PMMA is a “glassy” material at room and body temperature because it is both rigid and brittle. It exhibits these glassy properties because of its structure: The individual polymer chains are inflexible, and the chains are tightly packed together. Foldability can occur above the glass transition temperature (about 105°C [221°F], but raising a PMMA lens to this temperature prior to and during implantation is not practical.
FLEXIBLE MATERIALS
Various different flexible polymers have been used for IOLs. They fall into three categories: silicones, hydrophobic acrylics, and hydrophilic acrylics (i.e., hydrogels). The flexibility of each of these materials is based on three factors in the polymer’s structure: flexibility of the molecular chain, interchain flexibility, and flexibility as the result of the presence of other materials. The glass transition temperature for silicone optic materials is approximately -100°C, whereas those for hydrophobic acrylics are generally in the range of 5° to 15°C and 25°C for hydrophilic materials.
The three flexible material types constitute the basis of foldable IOLs implanted worldwide today.
Silicones
The first foldable IOLs were manufactured from silicon. Silicone polymers are any of a large class of polysiloxanes, which are very stable over a wide range of temperatures. They possess an alternating silicon–oxygen atom backbone with organic side groups attached to the silicon atoms. The first silicone material used in the manufacture of IOLs was poly(dimethylsiloxane) [PDMS], which has a refractive index of 1.41. This was termed first-generation silicone. Poly(dimethyldiphenylsiloxane) [PDMDPS] is the later, second-generation silicone IOL material in use today. It has a higher refractive index of 1.46, so IOL optics manufactured from it can be thinner.
Silicones derive their flexibility from both their chain structure, alternating silicone–oxygen bonds that create large bond angles allowing rotation about the silicon–oxygen bonds, and their intermolecular structure in which tight chain packing is not possible. Substitutional modifications along the chain silicon atoms can be made to modify overall material properties, most notably flexibility and refractive index. These are the only nonhydrocarbon polymers in general use and were developed primarily because of their ease of fabrication and thermal stability. They incite little inflammatory reaction and are used in scleral buckling implant materials, heart valves, shunts, and other surgical devices.
Major manufacturers of silicone materials include AMO (SI40 series and Clariflex); STAAR Surgical (AA4203); and Bausch & Lomb (SoFlex series).
Acrylics
“Acrylic” is actually defined as any compound that can be considered to be derived from acrylic acid. In general terms, it is used to apply to any type of plastic made from acrylic monomers. For example, the PMMA of Ridley’s first implant was designated as an acrylic material.
With the introduction of the AcrySof IOL, the term acrylic was expanded to define a new type of foldable IOL optic material, one composed of acrylic monomers, and also to distinguish it from the other foldable material of the time, silicone. The two adjectives hydrophobic and hydrophilic are used to modify the term acrylic as it pertains to IOL chemistry. The modifying terms are used based on the, wet ability or more accurately, the contact angle measurement, of the material and the water content of the material.
Flexible Hydrophobic Acrylic Polymers
Flexible hydrophobic acrylic polymers are structurally similar to the rigid PMMA used for IOLs for many years. However, substitutional changes to the monomers used to fabricate the chains, the use of different units in a controlled composition, and control of the intrachain structure result in the desired flexibility of these acrylic materials. Hydrophobic acrylic lenses have very low water content, usually less than 2%.
Major manufacturers of hydrophobic acrylic materials include Alcon Laboratories (AcrySof), AMO (Sensar), and Hoya (AF Series; Hoya, Japan), but recently other manufactures also started to use hydrophobic material.
Flexible Hydrophilic Acrylic Polymers
Hydrophilic acrylic polymers contain hydrophilic monomers and water. These polymers have the ability to swell like a sponge in water and retain a significant amount of water in their structure while not dissolving because their polymer chains are cross-linked. Their equilibrium water content depends on their composition and dictates their bulk and surface properties.
The currently available hydrophilic acrylic lenses are manufactured from acrylic copolymers (contain two primary acrylic monomers) with water contents ranging from 18% to 38%. One exception is represented by a lens manufactured in Brazil (Acqua, Mediphacos, Belo Horizonte, MG, Brazil), which has a water content of 73.5%. This expandable lens, based on the concept of the full-sized lens,67,68 is inserted in the dry state and attains its final dimension of the original crystalline lens within the capsular bag after hydration and expansion.
The hydrophilic lenses got off to a slow start in the United States because some of the early designs brought to the international market were poorly designed and fabricated and inadequately tested. Some of these designs had unanticipated surface and interior calcification. This has largely been eradicated by careful contemporary manufacture performed by established companies. Some lenses never had the problem of primary calcification of the material. For example, the acrylic polymer material used to fabricate the Rayner Center C-flex IOL (Rayner, London East Sussex, England) series has not produced any cases with this complication in almost 14,000,000 implantations over 50 years.
Five hydrophilic lenses have been available in the United States. The Bausch & Lomb Hydroview IOL (18% water); the Carl Zeiss Meditec MemoryLens (La Rochelle, France) (20% water); the Bausch & Lomb Akreos and the Rayner C-Flex (26% water); and the STAAR Surgical hydrophilic acrylic designs. The Collamer material (34% water), is essentially composed of 2-hydroxyethyl methacrylate (HEMA) monomer and porcine collagen.
The Rayner C-flex IOL (Rayner) (26% water) design is presently under Food and Drug Administration (FDA) investigation in the United States. Clinical and laboratory studies, as well as preliminary results of the FDA study, have shown excellent results with low rates of posterior capsule opacification (PCO).
Other major manufacturers of hydrophilic acrylics include Rayner Intraocular Lenses Ltd., Brighton Hove, East Sussex, England (C-Flex IOL, formerly Centerflex); Bausch & Lomb, Rochester, New York (Hydroview); STAAR Surgical (Collamer IOL); Ioltech (MemoryLens); and manufacturers of a wide variety of European lenses that are not available in the United States.
Light Filters
Two classes of UV-absorbing chromophores are used for the manufacture of pseudophakic IOLs: benzotriazoles and benzophenones. The UV cutoff for a particular chromophore is determined by the unique structural features it contains.
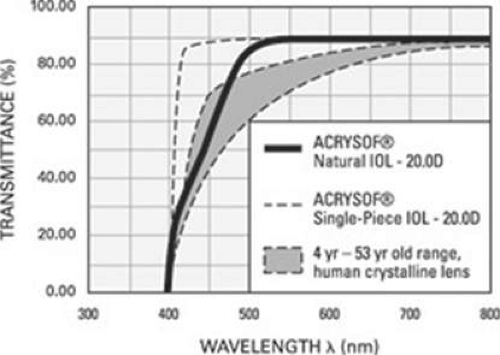
In 2003, IOLs that filter both UV and short wavelength visible violet and blue light were introduced in the United States (section on Light Normalizing Intraocular Lenses). A yellow chromophore is incorporated into the IOL optic to more accurately mimic the light transmission characteristics of the normal crystalline lens by partially blocking the transmission of presumably excessive amounts of short wavelength visible light.
Haptic Materials
Four materials are currently used for the manufacture of the haptic component (loops) of three-piece lenses: PMMA, polypropylene (Prolene), polyamide (Elastimide), and polyvinylidene fluoride (PVDF).
Material Summary
The following is a summary of some of these materials:
Polymer: a compound of high molecular weight derived from the addition of many small molecules (monomers) resulting in chain formation.
Elastomer: a material that returns to its original shape after folding.
Acrylic resin: any of a group of thermoplastic resins formed by polymerizing the esters of acrylic or methacrylic acid.
Acrylics: polymers incorporating methacrylic or acrylic esters, including PMMA, hydrogels, and other foldable high-water- and low-water-content materials (Fig. 11-54).
PMMA: poly(methylmethacrylate), a hydrophobic acrylic, with R1 = CH3 and R2 = CH3 (Fig. 11-54).
Acrylic IOLs are acrylic in nature, even though the term “acrylic IOL” has come to define a foldable IOL with an acrylic structure, such as the Alcon AcrySof series. Technically, acrylics are composed of methacrylate and acrylate co-monomers (2-phenylethyl acrylate and 2-phenylethyl methacrylate for the AcrySof material) (Fig. 11-54). The addition of the phenyl ring is key for producing a high refractive index of 1.55. Low-water-content acrylic IOLs like the AcrySof have a glass transition temperature of +12°C, which accounts for their slow folding at room temperature and faster unfolding at body temperature. The unfolding rate is considerably slower than that of silicone IOLs, which have lower glass transition temperatures and significantly different structure. Acrylic IOLs are hydrophobic, with a water content of less than 2%.
Hydrophilic acrylic (hydrogels): a family of polymers that swell in water and retain a significant amount of water in their hydrated structure without dissolving. These polymers of methacrylate esters mostly consist of a hydrophilic monomer with a hydroxyl functional group such as HEMA, or 6-hydroxyethyl methacrylate (HOHEXMA). The presence of this hydroxyl group is responsible for the material absorbing and retaining water. These materials may include a second hydrophobic co-monomer such as methyl methacrylate. The ratio of hydrophilic to hydrophobic co-monomer determines the water content of the hydrogel, and it may vary from 18% to 38%. Its formula is based on the acrylic polymer formula (Fig. 11-54), where R1 is CH3 and R2 is CH3, CH2CH2OH, or (CH2)6OH. There are many other variations of hydrogel IOLs used outside the United States.
HEMA: 2-hydroxyethyl methacrylate (Fig. 11-54). The original IOGEL lens was composed of HEMA, called poly-HEMA, and is a hydrogel of high water content (38%). This hydrophilic material damages corneal endothelial cells to a lesser degree than PMMA on contact69 and incites less giant cellular reaction on their surfaces.70
HOHEXMA: 6-hydroxyhexyl methacrylate, combines with HEMA in the formation of the Hydroview IOL.
Hydroview: a hydrogel-type IOL made by Bausch & Lomb Surgical. It has a low-water-content hydrophilic optic of 18% and PMMA haptics. The optic contains HEMA, HOHEXMA, and 1, 6-hexanedioldimethacrylate (as a cross-linker) (Fig. 11-54).
Elastimide: a polyamide haptic structure used in three-piece silicone optic IOLs manufactured by STAAR Surgical; originally used by Lynell Optics to frame and provide a haptic for its glass implants.
Silicone: elastomer of silicon–oxygen chains containing organic side groups, such as methyl or phenyl (Fig. 11-54). Polysiloxane with two methyl groups per silicon atom forms polydimethylsiloxane, the first-generation silicone material. If the organic side groups are two phenyl structures, alternating with two methyl groups, then the second-generation silicone is created, poly(dimethyldiphenylsiloxane). The refractive index is 1.41 for the first generation (STAAR AA4203VF), which accounted for IOLs with relatively thick optics. Because of the addition of the phenyl groups, the refractive index of the thinner second-generation IOLs is 1.47 (AMO SI40). Silicone lenses have a glass transition temperature of approximately -100°C, making them very flexible at room temperature. They are hydrophobic in nature.
UV blockers: benzophenone and benzotriazole structures.
Collamer: Trademarked by STAAR Surgical, this hydrophilic hydrogel is composed of poly-HEMA, containing 34% water and 0.3% porcine collagen with a benzophenone UV blocker. The porcine tissue component was chosen to enhance biocompatibility. The collagen is not subject to biodegradation because of the high degree of material cross-linkage.
INTRAOCULAR LENS TYPES, CLINICAL CHARACTERISTICS, AND USE
Polymethylmethacrylate
Until 1997, PMMA was the preferred optic material for IOL manufacture. In 1998 foldable acrylic became the preferred optic material and the PMMA IOL was the second most popular choice (33%) for use in primary implantation.46 As of 2003, PMMA was preferred by only 6% of the surgeons.71 It is available in an almost infinite variety of haptic configurations. It is available as a one- or three-piece IOL with PMMA or polypropylene haptics usually available in a modified-C configuration. It is available in positive and negative optic powers and because of its hardness can be piggybacked without optical degradation, which may be encountered in foldable IOLs.72
Optic sizes range from 5 to 7 mm. The 5-mm optic has been used by some surgeons in carefully crafted clear corneal incisions. A 5.5- to 6-mm optic is standard for those who want to place the IOL through a traditional superior corneoscleral incision. The usual overall haptic length of the IOL for intracapsular fixation is 12 to 13 mm. PMMA optics may be used for primary implantation in some very highly myopic patients and are even available in negative power.73 These are available in larger 13.5- to 14-mm diameters and are good choices for ciliary sulcus-fixated secondary IOL implantation in very large eyes in which a sufficient posterior capsular structure still exists; when it does not, these larger lenses can be secured within the sulcus and fixated with sutures anchored internally to the iris or externally to the sclera.
Silicone
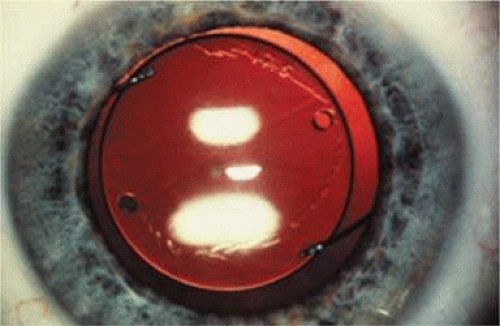
Effective since its introduction in 1984, silicone polymers, the original foldable IOL materials, are still very popular, preferred by 22% of American surgeons in 1998 for use in small-incision cataract surgery48 and by 21% of American surgeons in 2003. It is found in two designs, the plate haptic and the modified C-loop haptic. The early silicone plate designs appeared to be difficult to manufacture, and problems with molding edge finishing, optic opalescence, and surface irregularities were not uncommon. By the late 1980s, the design and manufacture had markedly improved and modern foldable plate IOLs, first characterized by the presence of small positioning holes on either side of the optic, emerged. With an overall length of 10.8 mm (11.2 mm measured corner to diagonal corner), the plate haptic lens produced by STAAR Surgical is designed specifically for use within an intact capsular bag. It originally had small 0.3-mm-diameter round fenestrations in each fixation plate. Apart from the main body of the lens itself, the plate design had no specific haptic elements designed for fixation. They were therefore not only prone to decentration within the capsular bag but also occasionally underwent significant dislocations when the surrounding capsular bag was disturbed, in particular after Nd:YAG laser posterior capsulotomy. The peripheral holes have been enlarged to 1.15 mm in diameter in these plate haptic silicone IOLs to promote a more significant fibrosis within them and produce greater long-term IOL stability within the capsular bag.74,75 The haptic surfaces have also received a textured finish to promote capsular fixation and rotational stabilization, which is important for the toric model (AA-4203TL) (Fig. 11-55)
This IOL is commonly delivered through an injector device using viscoelastic and a disposable plastic cartridge. An incision size of 2.8 mm is necessary.
The plate haptic IOL is not recommended for placement in eyes that have anterior radial capsular defects because of its strong tendency for clinically significant decentration and dislocation in such situations.
Although anterior capsule opacification (ACO) is a common occurrence with a potential complication of phimosis and decentration, the rate of central PCO as measured by the Nd:YAG laser posterior capsulotomy rate is relatively low (15.2%).76
An important modification of this design is the toric IOL. For this design to be effective, the lens should not rotate within the eye after implantation.
For the three-piece modified C-loop silicone IOL, available haptic materials include polypropylene, polyamide, PMMA, or PVDF in overall haptic diameters of 12.5 to 14.0 mm overall lengths. These 5.5- to 6.3-mm optic diameter lenses can also be delivered with an injector through a 3-mm incision or folded and placed with instruments through a 3.2-mm incision. Care must be taken with the haptics so they are not overly deformed or broken.
The posterior surface of silicone optics immediately fogs when in contact with intravitreal gas. Silicone optics create an interface with silicone oil, which makes fundus observation impossible during pars plana vitrectomy.77 Condensation compromises postoperative checks as well. Because of this difficulty, silicone lenses are not generally recommended for patients in whom vitrectomy may be more likely.
Earlier generations of three-piece silicone lenses seemed to produce greater and longer-lasting inflammatory changes. Increased cells and flare and chronic long-term uveitis seemed more common. PCO and capsule contraction appeared to be more common as well. All of these problems were more prevalent in patients with blood-aqueous barrier defects. Second-generation, silicone preparations (RMX 3 for STAAR and SLM2 for AMO) seem to produce these problems less often, and the AMO SI40 IOL has been reported to produce similar PCO rates as Alcon Surgical’s acrylic IOL, the AcrySof.78 A study of Nd:YAG laser posterior capsulotomy rates of human eyes obtained postmortem by Dr. Apple and his group showed that the PCO rate of the earlier AMO SI-30 design was 23.3% in total, whereas the PCO rate noted with the AMO SI40 design was 14.5% in total.76
Acrylic
1998 was the first year that the low-water-content foldable AcrySof (Alcon Surgical, Fort Worth, TX) acrylic IOL became the first choice (42%) among American surgeons,48 and it was increased to 69% by 2003.71 The popularity of foldable acrylics (high and low water content) had increased in 2009.66 Acrylic IOLs provide all the advantages of foldable optics with none of the problems associated with silicone. Optically, they have all the attributes of PMMA. They can be placed with folder instruments or through injectors. Because of the material, capsular contact with it, and squared posterior optic edge, capsular opacification and capsule contraction are generally reduced, compared with PMMA and silicone optics. The substantial advantage of this IOL is the reduction in the epithelial inflammatory consequences of capsular opacification and contraction. It is quite usual for the AcrySof models to demonstrate glistenings, which are condensed water droplets visible in the AcrySof hydrophobic optic.
Hydrophobic Acrylic IOLs
As of 2009, the hydrophobic acrylic IOLs available to U.S. surgeons were the AcrySof (Alcon Surgical) and the Sensar AR40 and AR40E (AMO Surgical). What about the hydrophobic Tecnis? What about the Hoya IOL? They received the FDA approval in 2008—they are not selling in the US?
Introduced in 1995, the AcrySof IOL is manufactured from hydrophobic acrylic materials with cross-linked polymers or copolymers of acrylic esters and is less than 1% water. It is available as a three-piece in 5.5-, 6-, and 6.5-mm optics with bonded PMMA modified-C haptics in overall lengths of 12.5 and 13.0 mm (the MA series: 30, 60, and 50), and as a single-piece lens (the SA and SN series). All IOLs feature a nonreflective surface of the optic edge. This treatment is applied to the haptics in the single-piece design as well. There is a 5.50-D base curve on the posterior surface, with the remaining refractive power applied to the anterior surface.
Alcon has incorporated two additional features as combined options into its AcrySof platform: a light-normalizing blue blocker (SN) and the posterior aspheric surface (WF). The AcrySof single-piece toric IOLs were approved by the FDA in 2005. The AcrySof IQ Toric (aspheric and toric IOL) was approved by the FDA in March 2009. The FDA approved the diffractive-refractive pseudoaccommodative IOL (ReSTOR D3), and the D1 model with lower add and aspheric features became available. Both multifocal IOLs are available in three-piece and single-piece designs.
In the initial models, the high-index plastic and squared edge combined to cause an increased incidence of unwanted photic phenomenon, such as glare, rays, and temporal dark shadows (dysphotopsia) in a few patients. The glare and rays have almost been eliminated after Alcon engineers placed the power curve on the anterior optic surface and made the optic edge thinner and nonreflective (frosted square edge). The incidence of temporal shadows or temporal obscurations of vision do not seem to have been influenced as much by those design modifications. It was hypothesized that the corneal edema associated with a beveled temporal incision contributes to this transient negative dysphotopsia.79
In 2000, the FDA approved the second foldable acrylic IOL, the Sensar AR40 (AMO Surgical) three-piece foldable IOL. This IOL featured a round edge optic. The edge was modified (AR40e) incorporating a PCO inhibiting posterior square edge and a compound rounded middle and anterior edge design to reduce the incidence of light reflections. On October 2007 AMO received the FDA approval for the Hydrophobic Tecnis IOL.
Another Hydrophobic IOL that received the FDA approval on September 2008 is the Hoya Spheric Model YA-60BB Intraocular Lens.
Hydrophilic Acrylic IOLs
For the last three decades IOLs fabricated from hydrophilic materials have occupied a back seat to silicone and hydrophobic acrylic because of technical complexities, varying degrees of bioincompatibility, and several cases in which the whitish discoloration on the optic surface or within the optical component occurred and required explanation. It is now known that these problems occurred only in poorly fabricated designs. Well-manufactured IOLs, for example, the series of Rayner IOLs, properly manufactured with their Rayacryl material, have not had that problem. More than four million of these lenses have been implanted over the past 10 years without a report of primary calcification that had given this category of lenses a poor reputation.
The high-water-content acrylic hydrogels are the descendants of the 38% high-water-content IOGEL composed of HEMA, which was not approved by the U.S. FDA (circa 1992) because of its tendency for intravitreal dislocation after YAG laser capsulotomy. It should be emphasized that the demise of this IOL and some others in this era was not because of lack of biocompatibility, but rather design problems that precluded good fixation of the IOL.
Hydrophilic acrylic IOLs are packed in fluid (wet packed). They are packed in a vial containing distilled water or balanced salt solutions; thus they are already in the hydrated state in their final dimensions within the container. These lenses are flexible, enabling the surgeon to fold and insert/inject the lens through small incisions.
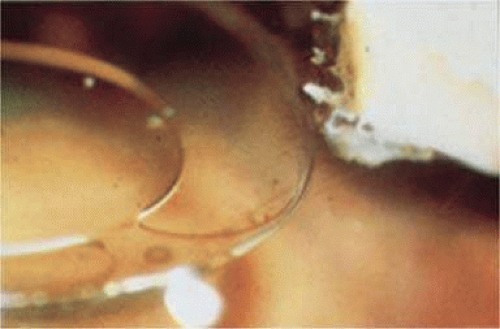
The Bausch & Lomb Hydroview entered the international market in 1995. It was the first hydrophilic acrylic IOL to be approved by the U.S. FDA. However, in May 1999, the manufacturer first received reports about clouding of a small number of Hydroview IOLs. Analysis performed at Dr. Apple’s laboratory revealed that the optical surfaces of the IOLs were covered almost completely by a layer of granular deposits on both anterior and posterior surfaces. The granules noticed on the optical surfaces stained positive with several calcium stains (1% Alizarin red, von-Kossa) (Fig. 11-56). Surface chemistry studies performed by Bausch & Lomb identified the lens deposits as a layered mixture of calcium phosphate, fatty acids, salts, and small amounts of silicone. This model, according to the manufacturer, revealed a migration of silicone from the gasket in the lens packaging onto the surface of the IOL. The models also indicated a possibility that in addition to silicone, fatty acids needed to be present to attract calcium ions to the lens surface. A compromised blood-retinal barrier also seemed to be associated with the appearance of calcified deposits. Therefore, fatty acids and silicone, perhaps in association with a metabolic disease in the affected patient, could result in the calcification.
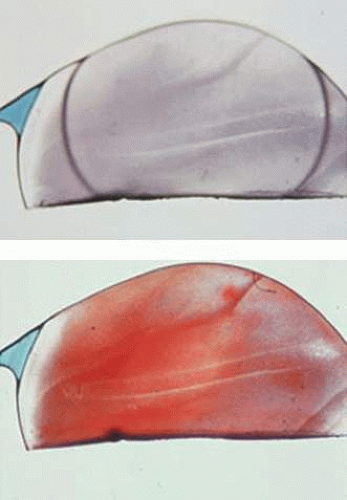 Figure 11-56. Hydroview IOL. A. Extensive surface calcification. B. Calcification shown by positive staining with alizarin red. |
In May 2001, on the basis of this evidence, the manufacturer changed the packaging of the Hydroview. The new packaging retained the ease of use of the previous SureFold components, but it was sealed with a gasket made from a perfluoroelastomer. The company states that calcification has not been reported in any of these IOLs. (I am not sure that it is still available; in the website of B&L, I could not find the IOL.) There were more opacification cases in England after the company had changed the gasket80
The MemoryLens by Carl Zeiss Meditec (formerly Ioltech, formerly by Ciba Vision, Duluth, GA) was introduced in 1989. The thermoplastic properties of the IOL are unique. The polymer used for the manufacture of the optic of this lens contains 59% HEMA, 16% methyl methacrylate, 4% 4-methacryloxy 2-hydroxy benzophenone UV absorber, and 1% ethylene glycol dimethacrylate. The haptics are made of polypropylene (Prolene). The lens is prefolded and remains glassy and stiff at room temperature. The prefolded MemoryLens can be implanted directly from the container without any requirement of folding instruments. The container with the lens is kept at a temperature of 8°C. After intraocular insertion, and under the influence of body temperature, the lens unfolds slowly (∼15 minutes) providing an atraumatic and controlled implantation.
Reports on granular deposits on the optical surface component of the MemoryLens led Ciba Vision to voluntarily withdraw the lens from the market in April 2000. Analyses performed in Dr. Apple’s laboratory on some of those lenses demonstrated that the deposits were in part composed of calcium/phosphate. Analyses performed by the manufacturer revealed that a biofilm, composed of different proteins, in addition to calcium/phosphate, was covering the optic surfaces of the affected lenses. The company modified their tumbling process used in lens polishing, thus changing the surface characteristics of the lens in the hope of avoiding the unwanted biofilm. No new cases of this problem have been reported with this new lens design. After identifying and correcting the problem, the manufacturer received approval from both the U.S. FDA and the European regulatory authorities to return the lens to the market. Ciba Vision re-released the MemoryLens as model CV232. This maintains the same basic characteristics of the previous models, but in addition has an incorporated square posterior optic edge for PCO prevention. The CV232 model allows surgeons to place this IOL through a 3.2-mm incision. In 2005, few cases of cleft, cavitation, or schisis of the IOL were identified by Dr Apple’s laboratory, the speculated mechanism leading to include a variation in the polymer and/or tighter rolling of the IOL after its relaunch than in the earlier version, in which no such occurrence was observed. During the folding process, the pressure applied to the IOL causes the anterior portion of the IOL to expand laterally while the posterior portion of the IOL compresses centrally. This may result in opposing forces inside the IOL, especially at the central portion, which can lead to movement of the anterior and posterior portions of the IOL in opposite directions. Changes in the manufacturing process of the IOL may result in an IOL material that is not cohesive enough to withstand these conflicting forces. This may result in an internal crack or cleft in the IOL, such as observed in Dr Apple’s laboratory. In a later stage, after implantation, this cavity may fill with aqueous humor.81,82
The STAAR Surgical Collamer IOL (CC4204BF) is a plate haptic, single-piece foldable lens manufactured from a collamer material. The overall length of the IOL is 10.8 mm (11.2 mm corner to diagonal corner) with an optic diameter of 6.0 mm. The haptic design has two 0.9-mm fenestrations to facilitate capsular fixation. These fenestrations are smaller than those of 1.15 mm incorporated into STAAR’s silicone plate haptic IOL.
This acrylic IOL is composed of a hydrophilic collagen polymer (copolymer of 63% hydroxyl-ethyl-methyl-acrylate, 0.3% porcine collagen, and 3.4% of a benzophenone for UV absorption), with a water content of 34%, a light transmission of 99%, and a refractive index of 1.45 at 35°C. It is also available as a three-piece model with a square posterior optic edge (CQ2015A).
The Rayner C-flex, formerly Centerflex (Rayner Intraocular Lenses Ltd.), is a single-piece, hydrophilic acrylic IOL, which began FDA trials in the United States in 2004 and gained U.S. FDA approval in may 2007. Studies in Dr. Apple’s laboratory have confirmed that this IOL and its basic polymer material have not been associated with the opacification/calcification and decentration problems seen with the previously mentioned IOLs, as well as the others that unfortunately have, until now, given this category of material a bad name. The C-flex lens has extended closed loops or haptics that render a general configuration of standard one-piece modified C-loop IOL designs. This type of platform helps provide stability of the lens optic in all three axes, an advantage that should be useful as new refractive or other specialized elements are added to this basic lens. The optic size of this lens is 5.75 mm with an overall diameter of 12.00 mm. The lens is made of a copolymer of hydrophilic and hydrophobic methacrylates with a water content of 26%, namely, HEMA and methyl methacrylate. Its material incorporates a benzophenone UV-absorbing agent, and it is inserted into the eye by means of a disposable cartridge-injector system (Fig. 11-57).
The original Centerflex model had a square edge around most of the optic edge, but the optic edge remained continuous with the haptics, and therefore, it was rounded and continuous where the haptics attached to the optic (Fig. 11-58). Dr. Michael Ammon (Vienna, Austria), in observations of clinical cases, and Dr. David J. Apple, in observations of pathologic specimens, postulated that the optic-haptic junction might represent a structural vulnerability for epithelial cell invasion, or as termed by Dr. Apple, an Achilles heel, in which the lack of the 360-degree square optic edge may inhibit the IOLs ability to block ingrowth of lens epithelial cells (LECs) over the visual axis.83 Both of these authors have furthermore postulated that the continuation of the square edge in the region of the optic-haptic junction would provide a complete 360-degree physical barricade against invading epithelial cells that cause PCO.
 Figure 11-58. Scanning EM of Rayner Centerflex IOL shows a smooth physical transition from haptic to optic. |
In rabbit studies completed at Dr. Apple’s laboratory, it was noted that the subsequently improved model 570C featuring a discontinuous enhanced edge provided the best barrier effect against cell migration/proliferation and PCO formation.65 Analyses of the scanning electron photomicrographs demonstrate this model’s complete ridge or enhanced edge extending for 360 degrees around the lens optic (Fig. 11-59). It is this enhanced-edge model 570C that underwent FDA trial. Another advantage of the C-flex IOL is its ability to serve as a drug delivery system for the fourth-generation fluoroquinolones after presoaking the IOL in the antibiotic solution.84,85
Another hydrophilic IOL, the Bausch & Lomb Akreos Adapt is a one-piece lens, with four fixation points, made of the unique Akreos hydrophilic acrylic material. The biocompatible lens material has a long-term safety record, with more than 2 million implants since 1998. Akreos Adapt is implanted with its single use Hydroport injector (PS27) for a 3.2-mm standard injection.
Faulty hydrophilic designs have been largely weeded from the market, and more recent experiences such as noted with the Rayner design have altered negative attitudes about this category.
PSEUDOPHAKIC INTRAOCULAR LENSES WITH SPECIAL FEATURES
LOW/HIGH DIOPTER INTRAOCULAR LENSES
Many companies today have low and high power IOLs. The STAAR IOL is good as a piggyback. Another good option is the Rayner Sulcoflex.
STAAR Surgical created low dioptric power IOLs with an overall optic diameter of 6.3 mm and overall length of 14.0 mm of 10-degree angled polyamide modified C-loop haptics (model AQ501OV). Powers available are +1.0 to +4 D in a convex/plano optic, 0 D in a plano/plano optic, and -1 to -4D in a plano/concave optic. These can be used primarily in the capsular bag or in the ciliary sulcus as a piggyback IOL in situations in which IOL power calculation results have resulted in residual postoperative refractive error. Its diameter and edge characteristics have made it the lens of choice for piggyback sulcus placement in cases of negative pseudophakic dysphotopsia.86 They also have been used to physically push back the Crystalens optic if the optic or haptic became involved in asymmetric anterior capsular fibrosis and the fibrosis had to be physically disrupted as a secondary procedure.
Similar products are available from other manufacturers, for example, the AMO DL60 L with a positive dioptric power range from 1 to 5 D and the DL60 N with a minus dioptric power range from -10 to -1 D. Both of these are 13-mm overall length one-piece PMMAs. The Alcon AcrySof Expand Series MA60 MA is available with a diopter range from +5.0 to -5.0 in whole diopter steps and 13.0-mm overall length. The Bausch & Lomb P574UV and BVR165XL are available in -18 D to +3 D in whole diopter steps. Models P359UV and P389UV are available in plano to +45 D in half-diopter steps. These are all single-piece PMMA lenses of 13.0 mm overall length. The three-piece silicone LI61 AO is available in positive dioptric powers of 0 to 4.0 D in whole diopters and 5.0 to 30.0 D in 0.5 diopters.
Light-Normalizing Intraocular Lenses
Background
Concerns about human eye retinal damage by UV radiation emerged because of the results of animal light toxicity studies in the late 1970s.87,88,89 This awareness stimulated the IOL industry to develop ultraviolet radiation (UVR)-filtering (215–400 nm) IOLs, especially because there should be no downside, that is, UVR did not contribute to vision. But, when these IOLs were introduced in the early 1980s, questions were raised regarding toxicity of the UVR-filtering chromophore90 and its potential for long-term leaching and possible release during Nd:YAG laser posterior capsulotomy. Unproven stability, safety, and lack of clinically demonstrated benefit of safety for these IOLs added to initial skepticism. The U.S. FDA testing for of the UVR-filtering material addressed these concerns, as did demonstration of protection by such filtering in animal experiments.91 Human studies92,93 demonstrated a need for protection from UVR. As a result, by the mid-1980s, virtually all manufacturers decided to offer only IOLs with an incorporated UVR-filtering chromophore. The Crystalens is a notable exception in offering very little UV protection.
In 1978, Mainster88 discussed retinal damage from intense near-UVR light sources after providing measurements of a non–UVR-filtering PMMA IOL and comparing it with that of human crystalline lens data from the literature.94 He recognized the need for tinting IOL material. But because of the excellent visual results of nonblue light-filtering IOLs at that time, and to avoid new materials, he suggested continued formulation of permissible exposure levels of UV radiation and continued counseling pseudophakic patients about possible occupational or environmental hazards to excessive visible light. Zigman89 supported yellow tinting of IOLs, citing his own earlier experiments in animals, which showed retinal damage even with nonintense near-UVR sources. He cited references95,96 on the yellow color of the crystalline lens of several species including human. He gave other circumstances such as light sensitivity, exposure to light-sensitizing drugs, and excessive glare, for which he suggested tinting of IOLs. He even created light-normalizing PMMA by a degradation process with UV radiation on an existing IOL material and IOLs made from it.97 In 1983, Sliney98 reported that removing wavelengths of light less than 500 nm would filter out only 10% of useful visible light but would remove 97% of harmful UV and blue radiation. In 1985, Marshall reviewed the issues of radiation and the aging eye99 and suggested tinted IOLs so that they would transmit light in similar fashion as the natural human lens. Filtering of UVR and blue light for pseudophakic eyes was also suggested in 1986.100,101 Similarly in 1987 Mainster102 reviewed the possible link between light and macular degeneration and recommended UVR and deep blue protective sunglasses for aphakic and pseudophakic eyes without any blue light-filtering IOL as precaution until the relationship between photic retinopathy and age-related macular degeneration (AMD) might be better understood. In addition to these studies, animal electroretinography (ERG) research,103,104 laboratory lipofuscin fluorophore A2E assessments,105 and human vitreous autofluorescence and fluorophotometry106 all suggest a potential benefit by reducing the amount of blue light overexposure experienced in UV-filtering IOLs.
In 1987, Young107 wrote a review on pathophysiology of AMD and followed up next year with a review108 on the hypothesis that solar radiation plays a key role in AMD and suggested both antioxidants and protective radiation filters be intrinsic components of a program of preventive medicine. As of 1988, it was recognized that although the need for sunglasses in very bright sunlight could not be met by an IOL, at least filtering as much as possible damaging UV and blue light for all light conditions in pseudophakic eyes might be reasonable.
Anil Patel, Ph.D., was the chief scientist at CooperVision in the early 1980s and contributed significantly to the development of UVR blocking and blue light–attenuating IOLs. At CooperVision he also worked on phototoxicity safety issues for its argon laser delivery system for retinal photocoagulation. In a historically interesting parallel, he had become part of the CooperVision organization through its acquisition in 1979 of the dental company Cavitron (New York). Before the acquisition, he made presentations to the FDA about phototoxicity considerations of a new UVR light source (Quicklite), which was used in the curing of dental material.109 Of course, CooperVision acquired Cavitron because it was Cavitron that manufactured the ultrasonic teeth-cleaning machine on which Dr. Charles Kelman based his first phacoemulsifier.110
Before the introduction of the foldable AcrySof Natural IOL, Hoya Corporation of Japan had created a similar yellow IOL in PMMA with chemically unbound yellow chromophores, in the early 1990s with a Japanese patent application date of early 1988.111 It had yellow dye but was not foldable; thus it was not significantly used inside or outside of Japan. It is interesting to note that Hoya created it primarily to reduce the frequency of clinical cyanopsia noted in pseudophakic patients. There exist several publications112,113,114,115 reporting that patients with pseudophakic vision with clear IOLs have color distortion because of excessive blue light, a characteristic called cyanopsia or dyschromatopsia. It is interesting to mention that the French artist Claude Monet chose yellow cataract spectacles (on one side) as he continued painting in old age after his cataract removal circa 1920 (Fig. 11-60). There are several publications on clinical studies of the Hoya PMMA IOL, which showed safety and normal color vision,116 improved contrast sensitivity,117 and long-term efficacy in decreasing blood-retinal barrier disruption.106 A subsequent publication found no clinically significant change in color contrast sensitivity between the AF-1 (UV) (Hoya) in one eye and the AF-1 (UY) (Hoya) in the other.118 An additional study compared two other IOLs by the same manufacturer (Hoya yellow-tinted YA60BB vs non-tinted VA60BB) and found no significant differences in photopic or mesopic contrast acuities. No difference in color perception was reported 3 months after surgery.119
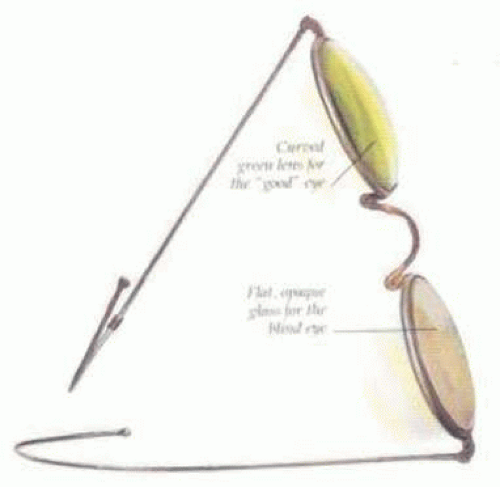 Figure 11-60. It is interesting to mention that the French artist, Claude Monet, chose yellow cataract spectacles (on one side) as he continued painting in old age after his cataract removal Ca. 1920 |
Menicon Corporation introduced a yellow-tinted IOL in Japan in 1994. It too was nonfoldable and thus not used in any significant numbers inside or outside Japan. Hoya Healthcare Corporation introduced a foldable Hoya Acryfold-1 (UY) IOL120 which is a UVR- and blue light-filtering IOL composed of Hoya’s hydrophobic acrylic material. At the 2004 ASCRS meeting121 a color vision expert from Japan presented a foldable light-normalizing silicone IOL from Canon.
Light-normalizing IOLs were also used in Sweden, and although ultimately no product came from Pharmacia, it patented122 a yellow PMMA material. ERG studies in rabbits103,104 in Sweden in the late 1980s showed that yellow filters protected the rabbit retina from light damage. Light-normalizing IOLs have been used in Russia as well. Since the mid-1980s, approximately 500,000 yellow-tinted PMMA IOLs have been implanted in patients at the S.N. Fyodorov Eye Microsurgery State Institute in Moscow, Russia (Fig. 11-61) (personal communication, Boris E. Malyugin, M.D., Ph.D., September 2004).
 Figure 11-61. Early light-normalizing IOL invented by Professor Fyodorov and used at his institute in the mid 1980s. |
In the fall of 2003, Alcon Surgical introduced the AcrySof Natural IOL, the first foldable light-normalizing IOL. The AcrySof Natural IOL with Alcon’s proprietary violet and blue light-filtering chromophore filters light in a manner that approximates the human crystalline lens in the 400- to 475-nm blue light wavelength. In addition to standard UV-light filtering, the AcrySof Natural IOL reduces transmission of blue light wavelengths by 71% at 400 nm and by 22% at 475 nm. The details of the creation of the bondable nonleaching, nontoxic, inert biocompatible patented dye for the foldable light-normalizing AcrySof Natural IOL material are proprietary to Alcon with several patents for the dye and material.123 The transmission characteristic for the light-normalizing single-piece AcrySof Natural IOL is on average similar to that of a 25-year-old natural crystalline lens, and provides comparative transmission spectra for the current AcrySof Natural IOL, the UVR-filtering single-piece AcrySof IOL, and the range of a 4- to 53-year-old human crystalline lens (Fig. 11-62).
This lens is manufactured by using the platform of the single-piece AcrySof and has been shown to be safe and effective in terms of biocompatibility and visual performance124,125,126 but the protective effect of this lens is still controversial and needs to be further investigated.
Questions about the accuracy of color vision perception and the quality of vision in photopic, mesopic, or scotopic circumstances seem to have been raised for three reasons.
First, while there are multiple blue light-filtering IOLs available outside of the United States, each with their own transmission characteristics,127 the proprietary nature of the chromophore bonding chemistry gave a marketing exclusivity to the Alcon Company, which is the only manufacturer able to provide a foldable light-normalizing IOL in the United States.
Second, although there are some studies that suggest that there may be an increase in the progression of macular degeneration after cataract surgery,128,129,130,131 there are no studies demonstrating a reduction in the incidence of new macular degeneration or its progression in patients receiving the AcrySof Natural IOL versus those receiving conventional UVR-filtering clear IOLs. Existing population studies are difficult to interpret because of vastly different study methods of phakic eyes; a lack of uniformity in what defines macular degeneration; inherent differences in genetics, diet, and general health standards; and environmental experiences of the subjects. Because of these problems many of the current large epidemiologic studies are in fact contradictory. Epidemiologic studies are difficult to interpret because some studies have shown a correlation between macular degeneration and light exposure, whereas others have not.132,133,134,135,136,137,138,139,140 Studies of the retina and macular degeneration after cataract surgeries have also produced conflicting results.128,129,130,141,142,143,144,145,146 Several reports,129,130,131,147,148 a postmortem study,128 and data from the Beaver Dam Eye Study149 have raised concerns that cataract surgery could increase the development of late age-related maculopathy (ARM) (neovascular ARM or central geographic atrophy) or progression to late ARM in eyes with early late-stage ARM lesions (soft drusen and retinal pigmentary abnormalities). Other studies have demonstrated a nonrelationship between cataract surgery and AMD.145,150,151
Third, there has been a value theorized for the relatively excessive short-wavelength blue light transmitted by non–light-normalizing IOLs for actually improving vision in scotopic conditions.152 There is a concern that patients might not function as well in scotopic conditions if this short-wavelength blue light is removed.88,152,153 Older adults have difficulty seeing in dim environments especially if they have early AMD,154,155,156,157 but the practical utility of scotopic vision is interesting to consider. It is colorless, pure rod night vision ranging in luminance from 0.000,001 cd/m2 to 0.001 cd/m2 (a moonless overcast night). Scotopic sensitivity is available when light energy is insufficient to stimulate cone photoreceptors. At these levels of illumination, which would be exemplified by a moonless overcast night, only rod receptors are active.
However, mesopic vision is much more valuable and practical for humans as it is experienced within intermediate luminance levels.158 It starts as a moonlit night extending to the levels of light experienced at early twilight or dusk. It is a common misconception that cones function only in the day and rods only at night. Both rods and cones function simultaneously within mesopic limits and when color is just beginning to become visible. From a practical standpoint, the levels of illumination encountered during night driving represent mesopic conditions.159
As to vision in mesopic and photopic ranges of luminance, a well-controlled comparative study with control of pupillary aperture has been reported.117 This study demonstrated improvement in contrast sensitivity for the midspatial frequencies of 6°C and 12°C and a reduced effect of central glare on the contrast sensitivity for light-normalizing IOLs.
Considering non–light-normalizing IOLs, there may be a tradeoff for any relative potential increase in scotopic sensitivity. Photochemical damage potential increases as wavelength shortens from green to higher-energy blue light. The potential of a few more photons to pass through a UVR-filtering IOL for scotopic night vision as a benefit in the aging pseudophakic eye needs to be balanced with the consideration of allowing the potential of exposure to 1,000,000,000 times more blue-light photons during bright daylight vision.160
There have been measurements made in several categories of scientific study that support the use of spectrum-normalizing IOLs. In addition to studies already mentioned, animal ERG research,103,104 laboratory lipofuscin fluorophore A2E assessments,105,161 and human vitreous autofluorescence and fluorophotometry106 all suggest a potential benefit by reducing the amount of blue-light overexposure experienced in UV-filtering IOLs. Human retinal pigment epithelium (RPE) cells have been exposed to blue light and were found to have a more significant reduction in apoptosis when an AcrySof blue-light filter was used than when an AcrySof UV filter was used.161,162 Vascular endothelial growth factor (VEGF) production has been used as an indictor of cell damage in A2E-laden RPE cells exposed to white light. Cells exposed to the light through a blue light-filtering lens material had significantly less VEGF up-regulation than those exposed to light through a UV-blocking lens material.163
Currently, scientific literature does not directly or indirectly prove the effectiveness of light-normalizing IOLs in preventing AMD or maintaining functional scotopic vision.164 A provisional argument has been made to acknowledge the general principle that the body’s repair processes become increasingly less efficient as exposure to environmental stimuli accumulates and that protection against those stimuli may be beneficial. But in the eye, it is difficult to discriminate between problems produced by a lifetime of light stimulation and those that may be seen as a result of a normal reduction in the effectiveness of repair mechanisms over time. They may be actually inseparable viewpoints of the senescent effects of the passage of time moderated by variations in the predispositions of genetics, nutrition, and lifetime radiation exposure.
Even internationally known experts do not agree as to the value of light-normalizing IOLs. Mainster and Sparrow152 reached different but somewhat similar conclusions about light protection in the bright outdoor sunshine. Mainster preferred a UV-filtering IOL plus sunglasses for use outdoors, whereas Sparrow preferred a UV- and blue light-filtering IOL and sunglasses.
At this point it is unknown whether blue light-normalizing IOLs will have any effect on the incidence or progression of AMD. In trying to consider this question, scientists and surgeons have begun to consider not only the concept of cumulative exposures but also the cumulative risks and benefits. The benefits of light-normalizing IOLs might be (a) elimination of cyanopsia, (b) improvement of contrast sensitivity and reduced glare effect126 in mesopic and photopic conditions, and (c) a potential protective effect of foveal function. The risk might be the loss of photons, which may have the potential for creating improved scotopic vision and interference with circadian rhythm.
The circadian system coordinates the timing of multiple biologic events to a specific phase of the 24-hour environmental cycle. Entrainment is the proper synchronization of a biologic entity to the daily light-dark cycle. This photoentrainment has been thought to occur because of visible light perceived by rods and cones. In 2002, it was discovered that the primary contribution for entrainment may actually come from intrinsically photoreceptive retinal ganglion cells (ipRGCs) with rods and cones playing a secondary role.165,166 These cells contain a photopigment called melanopsin that has maximum sensitivity to light in the blue part of the spectrum (around 480 nm). Since blue light-filtering IOLs reduce transmission in this range, arguments can be made regarding the potential interference with the amount of light available for proper entrainment. Seven different “action spectra” ranging from 480 to 492 nm have been discovered and labeled by their identifying research effort (usually their main author) since identification of the original peaked sensitivity of 480 nm in 2002.167 Two action spectra were identified (both peaked at 460 nm) prior to the ipRGC discovery. Because of the longer wavelengths, the newer action spectra support a significantly higher comparative effectiveness for entrainment for blue light-filtering IOLs than the earlier action spectra of shorter wavelength. The results of photoentrainment computations expect the blue light-filtering IOL in 65- to 80- year-olds to be within +6% to -13% effectiveness of the normal adult lens of 30- to 40-year-olds. Although these many discussions are extraordinarily interesting, the contribution to blue light blocking literally pales in comparison to the vast environmental variation of individual living circumstances.167
The validity of the potential long-term benefit of light-normalizing IOLs as a preventive measure for the preservation of macular vision needs long-term comparative clinical trials in pseudophakic eyes to be carried out similar to the AREDS study168 for establishing the role of UVR- and blue light-filtering IOLs as a potential preventive measure against known loss of vision with further aging for all eyes and AMD for susceptible eyes. It may be discovered that the current blue light-normalizing IOLs may not have a preventive effect on the occurrence or progression of AMD. If no preventive effect is found, it is possible that an increased concentration of dye could be used to possibly render an effect. If an effect is found, it might be possible that a lesser concentration of dye might still be salutary. Long-term trials should yield answers as to the potential benefits of light-normalizing IOLs.
AcrySof Natural
This study of the AcrySof Natural SN30AL was carried out, presented, and accepted by the U.S. FDA as part of the process to gain premarket approval.169 Study results showed no significant differences in performance between SN30AL and SA30AL models with regard to visual acuity, contrast sensitivity, or color perception as measured by the Farnsworth-Munsell D-15 test. The material has passed all requisite biologic testing for degradation, leaching, mutagenesis, and carcinogenesis. This IOL was approved for commercial use in the United States by the FDA in 2003. Since then, more millions of cases have been performed with only one report of IOL exchange because of color vision misperception with the Natural IOL.170 Contrast sensitivity and color discrimination with the Farnsworth-Munsell 100-Hue test have been confirmed comparing AcrySof Natural versus nontinted AcrySof SA60AT.124 It has also been corroborated that the AcrySof Natural does not increase anterior chamber inflammation.171
As an aside, in J.A.D.’s clinical experience, color misperception after cataract surgery has been rarely observed in people in whom we thought might have the potential for ultrafine color perception (i.e., artists and engineers). Sometimes after surgery with conventional UVR-filtering IOLs, a rare number of those patients had difficulty with color-matching components of their wardrobes or work materials. They would report that things had a greater blue tint than they had before surgery. Likewise, in 200 cases of implanting a conventional IOL in one eye and an AcrySof Natural in the other (a unique situation that occurred during the introduction period of the Natural in which patients’ first eyes were implanted with a conventional IOL and their second with the pigmented version), J.A.D. has seen only two patients who noticed that colors seemed “whitish blue” with the conventional IOL versus “skin toned” with the AcrySof Natural IOL. Neither perception was favored, nor was the combination bothersome to those patients.169
Toric Intraocular Lenses: 2010
With corneoscleral incisions of old, there was almost always some eventual against-the-rule astigmatism induced over the long term, this despite suture patterns that created some with-the-rule astigmatism immediately after surgery. These variations would have made the use of modern toric IOL technology unreliable. Now with modern toric IOLs available, the use of three-piece IOLs, which tend to rotate more than currently engineered toric IOLs,175 it has been estimated that 15% to 20% of cataract patients have more than 1.5 D of pre-existing keratometric astigmatism and could use the technology to reduce their postoperative refractive cylinder.176
Two styles of toric intraocular lenses are available in the United States, and they can both be used with ease correcting appropriate amounts of anticipated postoperative refractive cylinder. Whereas the patient candidacy requirements are stringent for multifocal IOLs, i.e., no significant imperfections in the visual system or ocular anatomy, there are very few restrictions for use of toric IOL technology. Since toric IOLs work best for correcting regular keratometric astigmatism in eyes with normal ocular surfaces, they have even been used in refractive lens exchage.177 They can also help improve refractive cylinder requirements in irregular astigmatism and in cases of form fruste keratoconus178 and pellucid marginal degeneration.179 Care must be taken to discover lid lesions or characteristics that may induce keratometric astigmatism and to make sure that astigmatism analysis is true and consistent in patients with corneal anterior basement dystrophy. There are various methods for alignment of toric IOLs,180 but a basic routine involves making a small mark with a pen at the 6 o’clock corneal limbus with the patient seated so as to avoid the occasional cyclotorsion effects of recumbency, marking the desired orientation of the toric IOL, and then achieving that orientation after all viscoelastic has been removed and normal intraocular pressure has been restored. Although correct placement is relatively easy, there are multiple assumptions that need to be made prior to surgery to get a good optical result.
It is probable that even the 1.8-mm incisions that have been enlarged for IOL insertion from the 1.0-mm incisions created during bimanual microincision cataract surgery (MICS) have some minimal astigmatic effect,181 but certainly the keratometric consequences of the 2.2- to 3.0-mm micro-coaxial cataract surgery incisions need to be included in the surgeon’s plan for each patient. Monarch cartridges have evolved so that they are now available in sizes to go through 3.0-mm 2.8-mm, and 2.4-mm incisions (Fig. 11-63). Called the incision-assisted insertion, the AcrySof IOL can be inserted through a 2.2-mm incision with the D cartridge pressed against but not through it. Some surgeons believe that these small incisions induce no consistent astigmatism, but Masket et al have shown that a 2.2-mm clear corneal incision induces an average 0.35 D of surgically induced keratometric flattening and a 3.0-mm incision induces 0.67 D.182 Surgeons may get some idea of their own characteristic incision-induced astigmatism after entering preoperative and postoperative data from a minimum of 10 cases courtesy of a free program on line provided by Warren Hill, M.D. (http://www.doctor-hill.com, accessed August 4, 2010)
The astigmatism-changing power of incisions can be seen when examining the consequences of creating a pair of incisions across the steep axis. They can have a magnitude of astigmatism-correcting effect similar to that of toric IOLs.183 In that study paired clear corneal incisions of 2.75 and 3.0 mm lengths (one used for surgery and the other just created for astigmatic effect) were created across the steep keratometric axis. AcrySof toric IOLs were compared to incision effect. In the toric IOL group, 95% of eyes achieved 20/40 or better uncorrected visual acuity (UCVA) and 70%, 20/25 or better with an average cylinder reduction of 1.75 D. In the paired incision group, 80% of eyes achieved 20/40 or better UCVA and 50%, 20/25 or better with an average cylinder reduction of 1.61 D.
There are other methods available today for correcting corneal astigmatism such as corneal or limbal relaxing incisions and excimer laser surgery. However, even though limbal relaxation incisions are accurate in some hands,184 astigmatic keratotomy is limited in its range and may be somewhat less predictable185 and excimer surgery can be expensive and is contraindicated in abnormal corneas.
The toric IOL is a potentially predictable and safe way to neutralize preoperative corneal astigmatism. Proper patient rotation selection and preoperative evaluation, as well as accurate implantation with an IOL that will present stability, are important in attaining the best possible results.
It is important to briefly mention some of the contraindications to the toric IOL. These include irregular corneal astigmatism, suspicion of zonular instability, and use of surgical techniques with unpredictable effects on postoperative astigmatism such as ECCE.
According to the 2009 Market Scope Survey of Cataract Surgeons, slightly more than 40% of surgeons reported making no attempt to surgically correct astigmatism. Toric IOLs were used by about a quarter of the surgeons who responded to this survey.
Preoperative Evaluation
It is critically important for the surgeon to develop an efficient routine to effectively evaluate patients’ candidacy for astigmatic correction—to develop the plan and then execute the plan. A disciplined routine of obtaining, evaluating, analyzing, and calculating preoperative and postoperative measurements needs to be established and followed. Hard or soft toric contact lenses should be removed for at least 3 weeks and soft at least 1 week before evaluations can occur. Testing should include manual and automated refraction, lensometry of current glasses, IOL Master (Carl Zeiss Meditec, Inc., Dublin, CA) axial lengths and keratometry length measurement, manual and automated keratometry, and computerized videokeratography. All test results should be in basic agreement or re-analysis and re-testing will need to be accomplished. Special care should be used in evaluation of the corneal surface and corneal videokeratography (CVK) images. Patients with corneal anterior basement membrane (ABM) dystrophy may exhibit some improvement in vision with various amounts of cylinder, but these refractions can change depending on the state of the ABM dystrophy (Fig. 11-64).
It must be remembered that all measurements, even the very reliable and repeatable ones obtained from sophisticated machines like the IOLMaster,186 are subject to the device’s inherent limits of absolute accuracy; the numbers used for calculations should be thought of as the mean numbers that could be obtained from the device. Then it must also be remembered that the devices are being operated by humans, usually in a private practice setting, who have varying levels of training and abilities. This human factor also introduces a compounding range of accuracy for each number obtained from each device. There then must be a consideration for the cumulative error that can occur in an individual patient because of the results of theoretically derived formulae, calculations, rounding of the IOL power selection up or down to the nearest 0.5-D availability, axis marking, actual individual surgery incision location and effects, IOL placement accuracy, and actual IOL (A-P) final placement versus anticipated effective lens position, and rotational stability, all subject to a similar range of means and standard deviations. For each individual case, if the numbers are average, then we should get results that are average, which would be good clinical results. If some numbers come from one side of an extreme and others from the other side, then the results can still be average and good clinically. But if a preponderance of data and calculation errors come from one side of the extreme, the cumulative effect of the errors can combine to provide an extreme result from that side, viz, a result that is not average and does not seem to be clinically correct for spherical equivalent or cylinder. Small variations in individual ingredient performances can produce relatively large final result variations. The effects of cyclotorsion are somewhat variable,187 but perhaps most important, theoretical estimations have shown that approximately 1 degree of off-axis rotation can result in a loss of up to 3.3% of the IOL’s cylinder power, so a 10-degree variation from perfect can result in a 33% loss of effect.188 After all of these considerations, it seems like somewhat of an intellectual contradiction to depend on a marking pen dot at the 6 o’clock limbus that may be 5 degrees wide even if perfectly applied.
A clinical case example is the left eye of a 70-year-old male operated by J.A.D. who had a preoperative manifest refraction of -3.00 × 086 degrees, keratometry 43.27 × 43.89 at 116. We had populated the Alcon Toric IOL calculator with an incision location of 10 degrees and a surgically induced astigmatism incision factor of 0.32 D. With that input, the calculator predicted that a T3 toric IOL placed at 111 degrees would leave a refractive cylinder residual of 0.12 D at 021 degrees. However, the patient’s final refraction was + 0.75 to 1.25 × 085 for 20/20. He expected to see well without glasses but did not, and he was unhappy with his investment in the toric IOL option, which should have delivered better uncorrected distance vision. He was presented with options, and he selected a refund of his upgrade fee and was happy just to wear the glasses that he had already purchased.
This second example demonstrates the difficulty in the possible effective use of an IOL, which might correct 0.50 to 0.75 D of astigmatism. There would be a lot of prospective patients, since J.A.D.’s current use distribution of AcrySof toric IOLs is 50% T3, 25% T4, and 25% T5. But, the currently available measurement and formula validities and accuracies might not provide enough reliability to deliver good results to each individual patient with that small amount of preoperative keratometric astigmatism.
Surgical Technique
There are various methods to align the toric IOLs. Some are complex photographic iris anatomy registration techniques180,189 and some involve limbal marking at the slitlamp. Carefully applied slitlamp markings have been found to have similar accuracy to photographic iris registration.190 One more basic routine involves making a small mark with a pen at the 6 o’clock corneal limbus (3 and 9 o’clock marks are also popular) with the patient seated (but not using a slitlamp) so as to avoid the occasional cyclotorsion effects of recumbency, and marking the desired orientation of the toric IOL (Figs. 11-65–11-70). Another way to have a long-lasting mark is by creating a small scratch of the corneal epithelium on the periphery of the cornea. During IOL insertion, the IOL is rotated to a position about 20 degrees short of its final desired axis position. It is important to remove all viscoelastic in front of and behind the IOL before final positioning because even a small amount of residual viscoelastic may create postoperative rotation in larger eyes and even the process of removal of viscoelastic can change final IOL orientation.191 It is important to have seated the IOL haptics and optic so that they are in a neutral position. The aim of the surgeon is to align the mark on the IOL with the desired axis mark on the cornea that was prepared by the surgeon at the beginning of the surgery (based on the orientation mark that was made while the patient was sitting). The AcrySof toric IOL may even be used without problem in cases where an anterior radial capsular tear has occurred during surgery. The 0.4-mm square haptics are long enough so that they can bridge an anterior radial tear, which extends to the capsular bag equator and will also provide enough friction so that the IOL will not rotate after placement (Fig. 11-71). Although correct placement is relatively easy, there are multiple assumptions that need to be made prior to surgery to end up with a good optical result.
 Figure 11-65. The six o’clock limbal mark made by pen with the patient in the seated position is seen to the right while the marking pen inks the axis markers. (Courtesy J.A.D.) |
 Figure 11-66. The axis identification ring is aligned with the 6 o’clock mark and placed on the conjunctiva so that the pupil and limbus are centered. (Courtesy J.A.D.) |
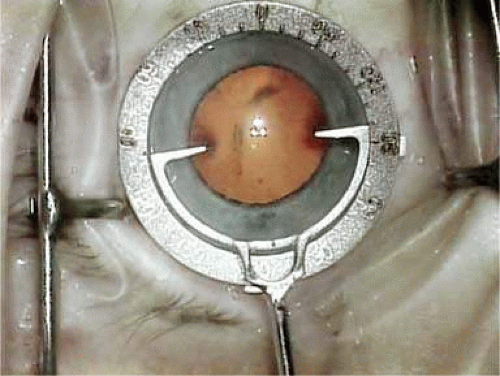 Figure 11-67. The inked axis marker has been oriented at 100 degrees, placed within the ring marker with that axis orientation, and pressed against the corneal surface. Courtesy J.A.D.) |
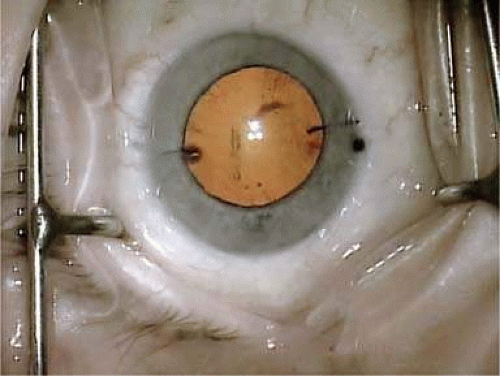 Figure 11-68. The inked axis marker has left its imprint on the cornea at 100 degrees. (Courtesy J.A.D.) |
Available Toric IOL Models
In the United States, only two models, the STAAR and AcrySof IOLs, are available. The other models mentioned in the following paragraphs are available in Europe and elsewhere.
STAAR Toric Lenses
The classic STAAR Surgical silicone posterior chamber plate-style IOL was modified to create an optic with toric capability (Fig. 72). Examples are models AA-4203TF (10.8 mm length) and AA-4203TL (11.2 mm length) approved by the FDA in 1998. STAAR’s single-piece plate design includes a silicone lens, with a biconvex optic, 1.15-mm fixation fenestrations, and textured haptics, designed to be implanted within the capsular bag. The cylindrical powers of the IOLs are of 2 and 3.5 D in the long axis of the lens.
 Figure 11-72. STAAR toric silicone IOL axis is marked with two lines to approximate its position under the steep keratometric meridian. (Courtesy STAAR Surgical.) |
The initial 10.8-mm-long version of that IOL (AA4203 TF) in the 1998 FDA study had a reported overall postoperative wrong orientation of greater than 20 degrees in 12% of cases.192 Performance of that style lens was improved by lengthening it to 11.2 mm in 1999 (AA4203 TL). David Chang,193 M.D., reported a surgical reorientation rate with that longer lens of 2.5% in his series in which alignment in 4% of cases were greater than 15 degrees off target alignment.
AcrySof Toric Intraocular Lens
There are three models that were approved by the FDA in September of 2005: the T3, T4, and T5, which respectively feature positive cylinder correction in the IOL plane of 1.5, 2.25, and 3.5 D, which translates to positive cylinder corrections of 1.03, 1.55, and 2.06 D in the spectacle plane. Models T6 through T9 are designed to correct higher amounts of astigmatism and are awaiting FDA approval. The IOLs feature three alignment marks on each side of the lens to assist with axis orientation. Implantation is also performed by injection with the Monarch III system (Fig. 11-73).
 Figure 11-73. Three dots at each central optic-haptic junction of the IOL identify the axis, which should placed to coincide with the patient’s steep keratometric axis. (Courtesy Alcon Surgical.) |
The AcrySof Toric IOL Calculator is an on-line tool that is used to calculate the IOL spherical power and toric model (Fig. 11-74) (http://www.acrysoftoriccalculator.com, accessed August 4, 2010). It takes into account the surgeon’s characteristic incision location for right and left eyes and induction of average amount of corneal flattening. It has been recommended by Alcon that preoperative manual keratometry readings or automated keratometry readings are entered to calculate the toric model while IOL Master keratometry and axial length measurements are used for spherical IOL power determination.
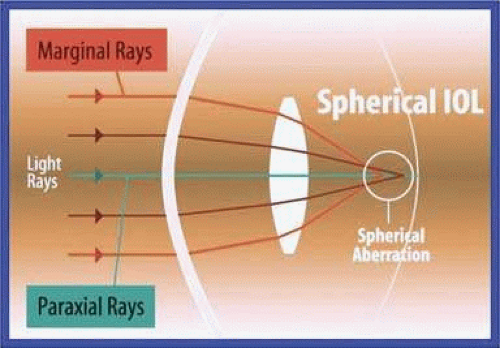 Figure 11-74. The Toric IOL Calculator gives a recommendation for IOL model, axis orientation, and anticipated residual astigmatism. (Courtesy Alcon Surgical.) |
The 2005 FDA clinical study was conducted to demonstrate the safety and effectiveness of the AcrySof Toric Posterior Chamber Lens Model SA60TT (Models SA60T3, SA60T4, and SA60T5, collectively as SA60TT).192 This was a randomized clinical study that included the AcrySof Model SA60AT as a control lens. More patients implanted with a Model SA60TT (n = 211) achieved better (all p-values <0.0001) uncorrected distance visual acuities as compared to those subjects implanted with the control lens Model SA60AT (n = 210): 20/20 or better, 38.4% (SA60TT) versus 19.0% (SA60AT); 20/25 or better, 66.4% (SA60TT) versus 40.9% (SA60AT); 20/40 or better 93.8% (SA60TT) versus 77.1% (SA60AT).
Residual refractive cylinder values were statistically significantly lower among those subjects implanted with either an AcrySof Toric Model SA60T3, SA60T4, or SA60T5 IOL when compared to the corresponding subjects implanted with the control Model SA60AT: 62.4%, 54.8%, and 67.8% mean reduction in refractive cylinder from the preoperative visit (keratometric cylinder) of SA60T3, SA60T4, or SA60T5, respectively, versus 10.8%, 22.1%, and 27.7% of SA60AT. Each of the AcrySof Toric Lens Models SA60T3, SA60T4, and SA60T5 had at least a threefold increase in the likelihood of achieving residual refractive cylinder of 0.5 D or less as compared to the corresponding control model.
Subjects implanted with lens Model SA60TT exhibited stability of cylinder at 3 months with greater than 90% of all subjects changing less than or equal to 1.00 diopter at consecutive visits between 1 month and 12 months. The rotational stability of the AcrySof Toric Model SA60TT is established with most lenses rotating 5 degrees or less, and the amount of rotation seen in each AcrySof Toric IOL model is independent of the amount of cylinder power present on the lens.
Statistically significantly more Model SA60TT subjects reported postoperative distance-vision spectacle independence compared to Model SA60AT subjects (59.9% vs 37.7%, respectively) when unilaterally implanted as specified by the terms of the FDA study.
Another study conducted by Lane et al194 compared clinical and patient-reported outcomes with bilateral AcrySof Toric or spherical control intraocular lenses (toric, n = 40; control, n = 22). The visual acuity, refractive cylinder, spectacle use, and patient satisfaction were assessed at 6 months. A significantly greater proportion of patients with toric IOLs achieved spectacle independence for distance vision (p = 0.019). Patients with toric IOLs had significantly less residual refractive cylinder (p <0.0001) and better binocular distance uncorrected visual acuity (UCVA) (p = 0.0014) than those with spherical control IOLs.194 A similar study found that residual refractive cylinder of less than 0.75 D was achieved in 74% of eyes and of less than 1.00 D in 91% of patients. Mean misalignment was 2.5 ± 2.1 degrees in the T3 group, 3.5 ± 2.3 degrees in the T4 group, and 4.1 ± 3.5 degrees in the T5 group.
Rayner
The Rayner toric IOL model 571 T (Rayner Intraocular, Ltd.), using the C-Flex lens as a platform, is well suited for this purpose because it is manufactured with a specific haptic design that should allow the IOL to stay centered and avoid the effects of capsular contraction or optic rotation. It should be resistant to rotation because the space between the closed-loop” type haptics functions in a fashion similar to the large holes or “foramen” seen with modern plate IOLs, in which fibrous tissue can grow. The toric model 571 T has demonstrated good performance in European clinical trials.
T-flex (573 T and 623 T)
The main technical features of these acrylic lenses are their one-piece design, with C-haptics and squared optic edges. Customizable toric power is added on the anterior side with radial markings along the steepest axis. The 623 T is available for lower spherical powers and the 573 T for higher-powered correction.
M-flex T
This lens is a multifocal aspheric IOL with customizable astigmatic correction between 1.0 and 6.0 D in 0.5-D increments. The lens uses refractive multifocal technology with four to five annular zones and provides 2.25 or 3.0 D of reading add at the spectacle plane. The C-haptic, squared edge optic, and single-piece acrylic design are similar to the C-flex.
Sulcoflex Toric
This lens is designed to correct postsurgical ametropia as a piggyback lens (adds on) implanted in the sulcus. It has a one-piece foldable design with undulated C-haptics. The 653 T model spherical correction is ranged between -3.0 and +3.0 D in 0.5-D increments with a cylindrical correction of 1.0, 2.0, and 3.0 D. Premium range of the IOL includes SE of -6.0 D to +6.0 D in 0.5-D increments, and cylinder power of +1.0 D to +6.0 D in 0.5-D increments
MicroSil Lens
The MicroSil MS 6116 toric IOL (Dr. Schmidt, formerly of HumanOptics, Erlangen, Germany) is a three-piece silicone-optic, PMMA-haptic lens. It has truncated edges, a spherical front surface, and a toric back surface. The overall diameter of the lens is 11.6 mm, with an optical diameter of 6 mm. Added blue light protection is optional. The haptics of the lens have a special Z-shaped design, which is purported to influence the rotational stability of the lens within the capsular bag and to balance any mechanical forces during postoperative capsular bag shrinkage. Because of the extended Z-haptics of this toric IOL, care must be taken during implantation as insertion into the capsular bag is more difficult than that of a conventional IOL, and an oval capsulorrhexis has been recommended.195 This lens is available in a wide power range of cylindrical powers from 2.0 to 12 D in 1-D increments. The MicroSil line also includes a three-piece lens with C-haptics for primary implantation into the sulcus (MS 614 T) and another IOL designed for the sulcus using a piggyback technique (MS 714 TPB).
Carl Zeiss
This company offers the Acri.Tec model toric IOLs, specifically the Acri.Comfort 646TLC and the Acri.LISA 466TD. Both 6.0-mm optic diameter lenses are made from an acrylic polymer with a hydrophobic surface and have squared optic and haptic edges. They can both be inserted through incisions smaller than 2.0 mm and are available in cylinder powers between 1.0 and 12 D. The Acri.LISA is a bifocal lens with an anterior toric surface and a posterior diffractive surface with asymmetric light distribution (65% for far and 35% for near) and 3.75 D of near addition. The Acri.Comfort has bitoric optic design.
Oculentis
This company has a line of toric foldable IOLs with squared optic edges made from an acrylic material with 25% water content and a hydrophobic surface. These IOLs can also be inserted through a MICS incision, and the models are available in a wide range of astigmatic corrective power.
Aspheric Intraocular Lenses
The purpose of the aspheric IOL is to lower the total optical higher-order aberration (HOA) level by minimizing a particular fourth-order HOA known as spherical aberration (SA), a symmetrical HOA induced in an optical system when peripheral rays have a different focus than central rays (Figs. 11-75 and 11-76).
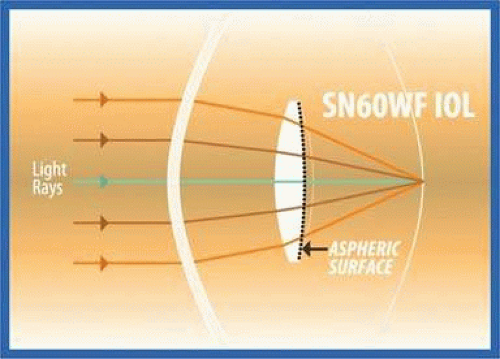 Figure 11-75. Axial and paraxial rays are focused on the retina while peripheral marginal rays are overfocused anterior to the retinal surface in spherical optic designs.(Courtesy Alcon.) |
 Figure 11-76. The aspheric posterior optic surface helps focus the light focused by the central and peripheral optic onto the desired retinal surface. (Courtesy Alcon.) |
The major contributors to ocular SA are the cornea and the lens. The SA of the cornea is positive.196,197 This means when central rays are focused on the retina, peripheral rays are focused in front of the retina. Several large studies196,197,198 have determined that the average SA induced by the cornea for a 6-mm aperture is approximately +0.27 μm, a value that remains relatively unchanged with age.197 The magnitude of corneal SA error is progressively lower for smaller apertures. As pupil diameter increases, more off-axis peripheral rays are focused in front of the retina, increasing the magnitude of the SA. Approximate magnitudes of corneal SA at decreasing aperture diameters are 0.13 μm at 5 mm, 0.051 μm at 4 mm, and 0.016 μm at 3 mm.199 Therefore, the effect of this aberration is sensed most acutely under scotopic conditions when pupils are maximally dilated.
In young people the crystalline lens counteracts positive corneal SA by exhibiting a negative SA, and as a result ocular SA remains low. With age the crystalline lens undergoes changes and the SA induced by the lens becomes progressively more positive. Although there is interpatient variability, on average by ages 40 to 50years the lens SA has risen such that ocular SA is greater than zero, with lenticular SA continuing to progressively higher positive values.196 Typical spherical IOLs act similarly to the aged crystalline lens in that they induce a positive SA by overrefraction at the lens periphery. The SA induced by a given spherical IOL is proportional to the lens power198 and increases with pupil dilation. For this reason, spherical IOLs are expected to reduce visual function below optimum levels postoperatively. Aspheric IOLs are different. Through a modification of one or both of the IOL interfaces, aspheric IOLs do not induce a positive SA. Aspheric IOLs can even be modified to induce a negative SA similar to the role of the crystalline lens in young people, potentially eliminating ocular SA altogether.
The IOLs
The first aspheric IOL to appear commercially was the Tecnis Z9000, a foldable, 3-piece silicone IOL. The Tecnis Z9000 is a CeeOn Edge IOL model 911 with a modified prolate anterior surface. This alteration lowers the refractive power of the IOL at its periphery, inducing a negative SA. This negates the effects of overrefraction at the periphery of the optical zone at the cornea responsible for corneal-induced spherical aberration. The Tecnis Z9000 and the related Tecnis Z9002 and Tecnis ZA9003 were designed to fully negate the average corneal SA by inducing a negative SA at 6 mm of -0.27 μm.
Another aspheric IOL, the AcrySof IQ, was developed by Alcon. It has the same ultraviolet and blue light-filtering chromophores as those found in the single-piece acrylic AcrySof Natural IOL (SN60AT, Alcon). The unique feature of the AcrySof IOL is the posterior aspheric surface that adds -0.20 μm of spherical aberration to the eye at the 6.0-mm optical zone. This only partially corrects corneal SA, leaving the average patient with residual positive SA. A small amount of residual SA could benefit the patient by increasing depth of field, allowing the patient to better tolerate some residual ametropia and be less dependent on spectacle correction for near tasks.
For aspheric IOLs that induce negative SA, such as the Tecnis or AcrySof IQ, to be maximally beneficial they need to be well centered and without tilt.200 If the aspheric IOL is not well centered, the induced HOAs from the malpositioned aspheric can cause the IOL to perform worse than a spherical IOL decentered to the same degree.199,200 Decentered spherical IOLS are expected to increase ocular coma and other aberrations as well201 but not to the same degree.
In light of this problem, Bausch & Lomb introduced the SofPort Advanced Optics model LI61 AO (Fig. 11-77). The SofPort IOL is an equiconvex silicone lens with prolate anterior and posterior surfaces. The aim of the IOL is to be perfectly aspheric and not induce HOA with tilt or decentration. Unlike the AcrySof IQ or Tecnis IOLs, which are designed to outperform spherical IOLs only up to a specific limit of tilt and/or decentration,201 the SofPort IOL should outperform a spherical IOL at a much larger range of optic orientations.202 However, because the SofPort does not reduce corneally induced SA, a well-centered Tecnis or AcrySof IQ IOL with minimal decentration or tilt should theoretically outperform the SofPort IOL in the average patient.
In recent years, other aspheric IOLs appeared in the market. These new models tend to imitate the basic strategy of one of the three IOLs mentioned previously; they either fully correct the average corneal SA like the Tecnis, partially correct the average corneal SA like the AcrySof IQ, or aim to be perfectly aspheric like the SofPort IOL. The Softec HD by Lenstec, (Clearwater, Florida) features a proprietary aspheric treatment to both anterior and posterior optic surfaces of its single-piece hydrophilic plastic. It has been presented that the biaspheric IOL is less sensitive to decentration and tilt. Perhaps most important, the company makes its IOLs available in quarter-diopter intervals rather than the industry standard half-diopter interval.
Simulations
Holladay et al198 simulated the effect of IOL tilt and decentration with an IOL designed to fully correct an SA of 0.27 μm at a 6-mm aperture. For a 5-mm pupil, the IOL outperformed a spherical IOL at up to 7 degrees of tilt and 0.4-mm decentration.
A study by Baumeister et al203 compared the level of HOAs and simulated visual function parameters in patients in whom one eye received a Tecnis IOL and the other eye received the Sensar AR40e, a spherical IOL. Scheimpflug imagery determined mean IOL tilt and decentration to be less than 3 degrees and 0.3 mm, respectively. The simulation by Holladay et al198 predicted the Tecnis IOL would improve visual function. However, although the Tecnis IOL did significantly reduce total HOA levels with 6.0-mm pupils, simulations with 6-mm pupils indicated that visual function with best spectacle correction was not significantly improved.
One explanation for this discrepancy is that the Holladay simulation198 overestimated the benefit of the Tecnis IOL by assuming it would completely eliminate ocular SA through its negation of the average corneal SA. In reality, negating the average SA will leave many pseudophakic patients with significant residual SA if their preoperative SA is significantly different from the population average. The corneal SA in cataract patients ranges between 0.055 μm and 0.544 μm at 6 mm.197
Also in doubt is the utility of correcting SA when many simulations have shown little to no expected benefit with 3-mm pupils. The 70-year-old pupil is approximately 2 mm smaller than the 20-year-old pupil.204 This helps reduce deterioration of visual quality with increased age and increased corneal and lenticular HOA. It also limits the benefit of SA correction with aspheric IOLs. One simulation204 estimated that for a patient with a 4-mm pupil, a centered spherical IOL, and full spectacle correction, fully neutralizing SA adds less benefit than would be gained by correcting 0.25 D of defocus. This potential benefit increases with increased pupil size, but with senile miosis it is unclear whether these effects are clinically significant.
Clinical Studies Comparing Aspheric and Spherical Intraocular Lenses
Ocular Aberrations
All reviewed studies comparing spherical and aspheric IOLs found significantly lower ocular SA in eyes with an aspheric IOL. In studies comparing different aspheric IOLs205,206,207,208 the trend was for the residual SA at 6 mm with a Tecnis IOL to be approximately 0 μm, with the AcrySof IQ 0.1 μm and with the SofPort AO about 0.2 μm. Considering that the average corneal SA at 6 mm in the population is 0.27 μm196,197,198 and the SA correction at 6 mm in the Tecnis (-0.27 μm), AcrySof IQ (-0.20 um), and SofPort AO (0) IOLs, these results are in line with preoperative expectations. It can be concluded that all three aspheric IOL strategies affect postoperative SA in the pseudophakic eye to the degree intended and significantly reduce SA in comparison with spherical IOLs.
Aspheric IOLs have also been shown to significantly reduce the magnitude of ocular HOAs compared with spherical IOLs but only at wider pupil diameters. This result is in line with expectations because the IOLs are designed to reduce only SA, and the magnitude of the SA correction is greater at larger aperture diameters where SA is larger and forms a greater proportion of total ocular HOA. In contrast, spherical IOLs increase ocular SA, especially at larger aperture diameters. This magnifies the difference between aspheric and spherical IOLs with larger pupils. Studies that compared the Tecnis, AcrySof IQ, and SofPort IOLs to each other207,209 found no difference in HOA magnitude at any aperture diameter between 4 and 6 mm. Therefore, it is possible that the reduced HOA levels with aspheric IOLs are not due to the specific magnitude of SA correction in the aspheric IOL and rather caused by not implanting an HOA-inducing spherical IOL. Still the question remains, even if aspheric IOLs effectively reduce HOAs compared with spherical IOLs, do they provide any clinical benefit?
Visual Acuity
Nearly all reports in the literature have concluded that there is no evidence that aspheric IOLs improve visual acuity postoperatively.205,206,207,209,210,211,212,213,214,215,216,217,218,219,220,221,222,223,224,225,226,227,228,229,230,231,232,233,234,235 Two papers did find a small but statistically significant best corrected visual acuity (BCVA) advantage in eyes implanted with the Tecnis 9000.236,237 One of these compared the Tecnis 9000 to the Alcon SA60AT,237 a result not corroborated by another similar study that compared the same two IOLs.216 Given the weight of evidence in the literature, it can be concluded that high-contrast photopic BCVA measurements are not significantly improved by implanting an aspheric IOL in the place of a standard spherical IOL.
Contrast Sensitivity
Contrast sensitivity measurements, more than visual acuity, have been shown to predict functional vision and visual performance for a range of object scales. A significant improvement in contrast sensitivity might support the widespread implantation of aspheric IOLs despite the lack of evidence demonstrating improved visual acuity.
There is some evidence that aspheric IOLs slightly improve photopic contrast sensitivity, especially at lower spatial frequencies.216,225,229,234,236,237,238,239,240 Even if true, it is unclear if this small benefit would provide the average patient with any clinically significant improvement in quality of vision. Another point to consider is that at a luminance of 85 cd/m2, where most of these photopic measurements were performed, the average pupil diameter was approximately 3 mm.205,213,219,222,225,232,237 At this level, aspheric IOLs have not been shown to significantly reduce ocular HOAs. If an improved HOA level is associated with improved visual function, it is more likely that improved contrast sensitivity will be conclusively found to occur at the larger pupil diameters induced by reduced lighting conditions.
Mesopic contrast sensitivity measurements performed at a median luminance of 3 to 6 cd/m2 where the average pupil diameter in elderly pseudophakic patients is about 4 mm.205,219,225,226,228,232,236,237,241 At this larger pupil diameter there is more potential benefit in correcting SA as it induces a larger proportion of total HOAs. As expected by simulations, contrast sensitivity improved significantly under these conditions, with a large majority of studies212,218,224,225,226,227,229,231,232,234,236,237,238,239,240 showing a benefit for aspheric IOLs over spherical counterparts at one or more mainly lower spatial frequency.
Subjective Visual Function
Two studies compared patient satisfaction with aspheric and spherical IOLs. One228 compared subjective visual function in patients with bilaterally implanted Tecnis 9000 or AcrySof MA60AC IOLs and found no difference in Visual Function-14 scores. Another study230 compared the AcrySof IQ to the AcrySof SA60AT and found no statistically significant differences in vision-related quality of life with the National Eye Institute Visual Functioning Questionnaire (NEI VFQ-25), despite significantly lower HOAs and SA levels in eyes with the aspheric IOL. The authors explained this discrepancy by stating that many in their patient population performed few tasks requiring highly functioning nighttime contrast sensitivity like night driving. More careful patient selection may have improved the subjective benefit enjoyed by trial subjects.
Optimal Spherical Aberration Correction
A fundamental flaw in many of the comparative studies between spherical and aspheric IOLs is the assumption that any single aspheric IOLs should be a one-size-fits-all solution. In one study the corneal SA in patients presenting for cataract surgery ranged from 0.055 μm and 0.544 μm at 6 mm.197 Implanting a Tecnis IOL into each patient may leave a large group with an average postoperative ocular near zero, but individual patients with nearly no preoperative SA would end up with a highly negative final ocular SA. As a consequence, the group would experience suboptimal benefit from the aspheric IOL. Selecting an IOL based on the patient’s specific corneal SA error and aiming for a particular value of postoperative asphericity may provide more predictable results.
A clinical trial by Packer et al242 took a group of cataract patients and targeted zero postoperative SA in each patient. Preoperative corneal SA was measured and a Tecnis, AcrySof IQ, or SofPort IOL was chosen so the arithmetic sum of the IOL and corneal SA came closest to zero. Total postoperative ocular SA in this group measured -0.013 ± 0.072 μm. The standard deviation in this population was lower than in other trials of similar size with aspheric IOLs, indicating that it is possible to effectively target a specific postoperative ocular SA.
No Residual Spherical Aberration
Peirs et al243 compared letter acuity and contrast sensitivity for two different values of SA in four young phakic patients with 4.8-mm pupils. The first condition was 0.154 μm of SA at a 4.8-mm pupil, the average amount of SA in pseudophakic patients with a spherical IOL, and the second condition was complete correction of the subject’s SA. Average contrast sensitivity improved 32% with complete negation of ocular SA. It should be noted that pupil size in these subjects was greater than that found in the average pseudophakic patient under all but scotopic conditions.211,235 Therefore, the effect of negating SA in elderly pseudophakic patients is likely overestimated. However, the study does suggest that achieving a postoperative SA of 0 could have a clinically significant effect on contrast sensitivity under conditions where pupil size is larger.
Residual Positive Spherical Aberration
It has also been suggested that targeting a postoperative SA of +0.10 at 6 mm is a better choice244 than targeting zero residual SA. This line of thinking is supported by Levy et al,245 who found a mean total SA of +0.110 ± 0.077 μm at 6 mm in 35 young patients with ‘supernormal’ vision (uncorrected visual acuity μ20/15). However, there is no logical basis for the implication that the patient’s SA was responsible for supernormal visual acuity. In fact, the authors found that the SA level in this population was comparable to a group of preoperative myopic patients considering refractive surgery (+0.128 ± 0.074 μm).246
Nevertheless, a trial by Beiko244 tested the theory of a postoperative SA of +0.10. He selected a group of patients with corneal SA greater than +0.33 um (average +0.370 ± 0.024 μm, range +0.337 to +0.411 μm) and implanted a Tecnis IOL with -0.27 μm of SA correction. The result in this group was compared to an unselected group of patients with an average preoperative corneal SA of +0.291 ± 0.081 μm at 6 mm (range +0.149 to +0.419 μm) who also received a Tecnis IOL. The first group demonstrated significantly better mesopic and photopic contrast sensitivity at multiple spatial frequencies. This interesting finding would need to be confirmed in a larger trial that compared visual function between groups targeting +0.10 or 0 residual ocular SA. This study does further support the notion that it is possible to target final ocular SA in a small group of patients.
No Single Spherical Aberration Is Best
It can also be argued that there is no single ideal target SA value, and that the optimal SA value depends on patient-specific factors. A study by Applegate et al247 examined the effect on visual acuity of combining various pairs of aberrations. They found that certain combinations of aberrations could improve letter acuity above the levels induced by either aberration alone. A report by Wang and Koch248 carried out a simulation examining the effect of varying ocular SA levels while leaving all other HOAs intact in 154 emmetropic eyes. Best image quality was obtained in more than 90% of eyes with a residual SA of between -0.05 and +0.05 μm. However, the specific pseudophakic ocular SA that optimized visual function depended on the interaction of many different corneal HOAs as well as pupil size.
Another point to consider is the potential benefits of residual SA. Increased ocular SA is associated with increased depth of focus249,250 and improved distance corrected near visual acuity. With higher SA, fewer incoming rays converge at the focal point of the eye. Although this decreases optimum image quality at best focus, the off-center rays provide improved visual function at other points along the visual axis. When SA is corrected, fewer rays are focused at other points along the visual axis reducing depth of focus and distance corrected near acuity. The trade-off between optimized contrast sensitivity when SA is minimized and increased tolerance to defocus with higher residual SA should be considered when determining a patient’s ideal postoperative HOA profile.
Which Patients Should Receive Aspheric IOLs?
Although aspheric IOLs can improve visual function in comparison with spherical IOLs and the available technology allows for the targeting of a specific pseudophakic ocular SA, we do not yet know how to optimally correct ocular SA. Gaining maximal benefit from the technology will likely require individualizing patient asphericity,242,244 a change that would necessitate preoperative calculations of corneal SA and other HOAs. Even if this information is collected, it is unclear how it should be used to determine the optimal aspheric IOL correction. Still, collecting these data may be a necessary first step before the role of aspheric IOL technology can be established.
There is another concern with aspheric IOLs. Given senile miosis, their purported advantage would be clinically significant only in a subgroup of elderly cataract patients who perform a significant amount of contrast-dependent tasks under mesopic or scotopic conditions, like night driving. The benefits might be more apparent in younger patients with larger pupil sizes who undergo refractive lens exchange or surgery for a congenital cataract. The role of an aberration-free SofPort IOL might be to reduce induced HOAs if an increased risk of optic tilt or decentration exists, for example with significant pseudoexfoliation. However, it is likely that all these indications add up to only a small proportion of cataract patients.
A third factor to consider is that while cataract surgery technology has improved, exact emmetropia is not achieved in all eyes. Studies have shown that aspheric IOLs have the potential to improve visual function with best spectacle correction in place, but many pseudophakic patients do not wear spectacle correction and find their uncorrected vision adequate for most if not all tasks of daily living. As the popularity of multifocal IOLs indicates, many cataract patients are willing to accept less than optimal visual function to remain spectacle free. Patients with uncorrected defocus and astigmatism who have accepted reduced acuity from best corrected values are less likely to derive clinically significant benefit from targeting a specific postoperative ocular SA. Blur from the uncorrected lower-order aberrations should negate the benefit of reducing ocular SA, which can optimally improve only contrast sensitivity, but not letter acuity, with best spectacle correction in place.
Summary
It is clear that reducing HOAs has the potential to improve quality of vision and that aspheric IOLs can play a role in achieving this goal. However, the available information today suggests that only a few cataract patients would derive clinical benefit from aspheric IOL technology. Even among these patients it is unclear how the available IOLs should be used. Future developments, including new multifocal aspheric IOLs, may expand the applications of this technology.
Presbyopia-Correcting Pseudophakic Intraocular Lenses: 2010
Some lucky patients amaze us by seeming to achieve good uncorrected distance and near vision without the aid of spectacle correction. These patients benefit from factors that collectively can be called pseudoaccommodation and/or be able to recruit relative multifocus from astigmatism and/or higher-order aberration to create an accommodative-like effect. Pseudoaccommodation refers to that accommodative-like near vision performance achieved by means other than classic accommodation, which is caused by lens deformity or anterior lens shift or the use of a multifocal IOL.251 Factors that contribute to pseudoaccommodation, and which can improve apparent subjective and objective accommodative performance, include posterior placement of the IOL,252 residual myopic astigmatism253 and small pupil size, and pupillary constriction during accommodation.254 Higher-powered IOLs will have a greater effect and lower-powered IOLs a lesser effect. Additionally, there may be some ability to shift their IOL optic forward in response to ciliary body contraction. Without those pseudoaccommodative elements, good levels of uncorrected distance with some level of intermediate or near vision can currently be reliably obtained by three basic strategies.
Monovision in which the focus for usually the dominant eye is set for distance and the nondominant eye for near or intermediate focus. The work is primarily done by the brain.
Multifocal IOLs, which split light into distance and near components either by multizonal refractive or refractive-diffractive means. The work is primarily done by the light.
Accommodating IOLs, which require some level of anterior displacement of the IOL optic or change in its surface to increase its power in response to ciliary muscle contraction. The work is primarily done by the ciliary muscle.
The annoyance of presbyopia can measurably reduce quality of life versus compared with the improved quality experience of life that can be experienced after successful presbyopia IOL implantation.255 Some have argued for an economic justification for using presbyopia-correcting IOLs.256 It is important to remember that a trade-off exists when considering these devices. Patients who do not like wearing spectacles for reading and are sufficiently motivated toward complete spectacle independence can take an increased chance of having to endure glare and halos. And in the future, the correction of presbyopia may not be just an IOL option as a LASIK procedure, presbyLASIK has yielded similar near vision results to the Acri.Smart 36A (Acri.Tec, Munich, Germany) but better near vision performance than that achieved with the Crystalens AT-45 (AMO).
Monovision
The gold standard for cataract surgery patients has been to calculate for the intraocular lens power to emmetropia and leave the patients with a plano refraction so that they just need to wear reading glasses after surgery. This has been and still is a tremendously effective strategy that provides consistently excellent quality vision and stereopsis. But some patients do not want to wear reading glasses or bifocals even if the distance portion is close to plano. Monofocal IOLs, even in using a monovision strategy, are generally thought to provide better quality of vision and with fewer visual side effects than multifocal IOLs.257 Some clinicians use a minimonovision technique leaving the nondominant eye with a residual approximately -0.75 sphere to help with intermediate and near tasks. Properly selected and motivated patients can be happy and have good functional vision with a slightly greater difference. In one study258 with a mean anisometropia of 1.16 D, 96% of patients had better than 20/30 uncorrected distance visual vision acuity, and 92% had uncorrected near vision of J4 while maintaining good stereopsis s and contrast sensitivity. Another study found that for effective reading ability, monovision with the dominant eye left at plano and the nondominant eye left at -2.00 sphere provided satisfaction in 80% to 90% of properly selected cataract patients.259
A kind of monovision can also be achieved by implanting a multifocal IOL in one eye and a monofocal IOL in the other. One study found that unilateral implantation of a multifocal IOL (ReSTOR SN60D3) in patients who were previously pseudophakic with a monofocal IOL in the other eye, provided high levels of patient satisfaction (75%), spectacle freedom (65%), and good visual acuity without compromising contrast sensitivity.260 However, patients who received the multifocal IOLs bilaterally were more satisfied (92%) and had a greater level of spectacle freedom (77%).
Multifocal Intraocular Lenses
History
Early attempts to manufacture a multifocal IOL were made by 3M Corporation in the 1980s through using a design similar to the current Tecnis (AMO) diffractive IOL based on diffractive lens technology across the entire anterior optic surface, similar to the current Tecnis (AMO) diffractive IOL. IOLAB Corporation, also in the early 1980s, fabricated a two-zone refractive bifocal disc IOL with a central near zone based in part on a design of Dr. John Pearce. Storz evaluated a three-zone refractive IOL in the mid-80s, and in 1989, Pharmacia introduced a multizonal IOL as well (Fig. 11-78). For these IOLs, the near zones of both designs were central and the distance portions peripheral.
 Figure 11-78. An early multifocal lens from Pharmacia 1989. The large central zone was dedicated for near vision while the peripheral zone is dedicated for distance. |
In the 1980s, most IOLs were rigid and large incisions were used. Monofocal IOLs were giving good clinical results, and multifocal IOLs were evaluated on rigid IOL platforms in clinical studies. Similar designs evaluated as contact lenses generally had poor acceptance, and it was not clear at the time how good the multifocal IOL performance needed to be. There was no clear path to approval by either the U.S. FDA or the European regulatory agencies. In 1992, an FDA advisory panel approved the 3M diffractive multifocal IOL, but the FDA then requested that additional studies be conducted to evaluate vision while driving. Additional testing was done successfully, but the move to foldable IOLs in the mid-90s was then well under way, and the lens was never approved in the United States, though it was available for a while overseas. In 1997 Allergan Corporation’s Array lens was the first multifocal IOL to be approved by the FDA, following both conventional visual testing and a driving simulation evaluation.
Because of the superior performance of monofocal IOLs with regard to acuity performance, contrast sensitivity, and visual aberrations, multifocal technology was withdrawn from the U.S. market until 1997 when Allergan Corporation’s Array lens was approved by the FDA. This was the first multifocal design to be placed on a foldable IOL platform and was available until it was replaced by a similar multizonal refractive optic design created using a foldable hydrophobic material, which was designated the ReZoom IOL (AMO).
The Array lens by AMO was the first multifocal IOL to be FDA approved and marketed in the United States. It was a three-piece multifocal multizonal IOL manufactured from a second-generation silicone material (Fig. 11-79). The optic material had a refractive index of 1.46. It had angulated C-haptics made of extruded PMMA. The optical design of this lens was a zonal-progressive type with five concentric zones. It was a distance-dominant, zonal-progressive optic. The center of the lens was primarily for distance, with the other zones having distance and near vision in different proportions: 50% of the available light is devoted to distance vision, 37% to near vision, and it was suggested that 13% be directed to intermediate vision because of aspects of the design, and 37% to near vision. The addition for near vision is +3.5 D.
There were concerns related to a higher incidence of decreased contrast sensitivity, glare, and suboptimal near vision performance with the Array IOL. Due to the accommodative pupil constriction reflex, there was a concern that patients with smaller pupils might not benefit from the peripheral portions of the IOL devoted to near vision. However, Javitt and Steinert261 compared bilateral implantation of the Array lens to a monofocal lens with respect to visual function, patient satisfaction, and quality of life. They found that those patients who had bilateral implantation with the Array obtained better uncorrected and distance-corrected near visual acuities and reported better overall vision, less limitation in visual function, and less spectacle dependency than patients with bilateral monofocal lenses. In a study by Schmitz et al262 reporting on contrast sensitivity and glare disability with halogen light after monofocal and multifocal lens implantation, reduced contrast sensitivity was found in the multifocal group only at the lowest spatial frequency without halogen glare. The monofocal and multifocal groups of patients studied by Schmitz et al had no statistically significant differences in contrast sensitivity with moderate and strong glare.
The Array IOL reached a maximum popularity in 2000, with use in 35% to 50% of some ophthalmologists’ practice.
ReZoom Multizonal refractive intraocular lens
ReZoom was approved by the FDA in March 2005. It is similar to the Array in its five-zone design light-splitting features and three-piece PMMA haptics but is created using a foldable hydrophobic acrylic optic. Zones 1, 3, and 5 are for distance and zones 2 and 4 for near vision. The optic features the OptiEdge (rounded anterior edge and square posterior edge), designed to reduce both edge reflections and PCO. The ReZoom had better intermediate vision performance than the original ReSTOR D3, which had better near vision performance. Because of this, there was a period when mixing these IOLs, usually with a ReSTOR in the dominant eye and a ReZoom in the nondominant eye, was popular with some surgeons. It has generally generated more visual disturbances and aberrations than the ReSTOR or Tecnis263
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree