 Inflammatory Lesions That Simulate Neoplasms
Inflammatory Lesions That Simulate NeoplasmsThyroid-Related Ophthalmopathy
General Considerations
There are a number of orbital inflammatory processes that are important in the differential diagnosis of orbital tumors. The most important of these are thyroid-related ophthalmopathy (TRO) and idiopathic nongranulomatous orbital inflammation (INOI) (“inflammatory pseudotumor”). In addition, there are a number of bacterial, fungal, and idiopathic granulomatous disorders that can occur in the orbit.
TRO is a common autoimmune disorder usually associated with hyperthyroidism. The diverse nomenclature for this intriguing entity has been confusing and inconsistent and includes Graves disease, Graves orbitopathy, Graves ophthalmopathy, thyroid eye disease, thyroid orbitopathy, thyroid ophthalmopathy, dysthyroid orbitopathy, dysthyroid ophthalmopathy, thyroid-related orbitopathy, thyroid-associated orbitopathy, and thyroid-associated ophthalmopathy. We have chosen to use the term “thyroid-related ophthalmopathy” because the condition is related to thyroid dysfunction and affects other ocular structures (eyelids, conjunctiva) in addition to the orbit.
TRO typically affects the extraocular muscles and has rather characteristic features. In some cases, it can simulate a neoplasm clinically and radiographically. Numerous chapters and articles have addressed the clinical features, pathogenesis, pathology, and management of TRO; only some of the more recent or more important ones are cited here (1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17).
Clinical Features
Clinically, the patient with TRO usually shows characteristic unilateral or bilateral proptosis, eyelid retraction, eyelid lag on downgaze, eyelid edema, and conjunctival hyperemia and edema. In more advanced cases, the patient can develop diplopia, exposure keratopathy, and optic disc edema owing to compression of the optic nerve in the orbit (1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17).
Diagnostic Approaches
Although there are many laboratory tests to assess thyroid gland function and diagnose TRO, the main ones used by most clinicians are tri-iodothyronine, total and free thyroxin, and thyroid-stimulating hormone. Orbital imaging studies have become highly reliable methods of diagnosing TRO. Computed tomography (CT) and magnetic resonance imaging (MRI) show characteristic enlargement of rectus muscles without appreciable involvement of the muscle tendons and orbital fat. Ultrasonography has been largely replaced by these superior imaging techniques (1).
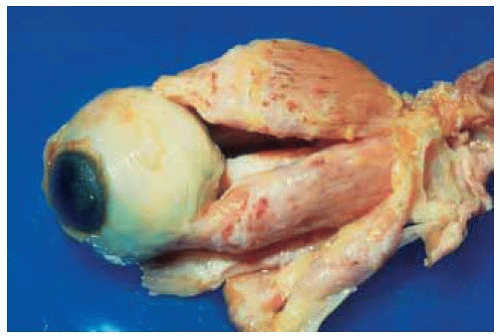
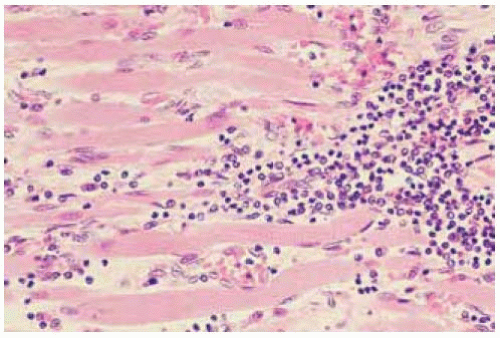
Pathology
Management
Selected References
1. Rootman J, Dolman PJ. Thyroid orbitopathy. In: Rootman J, ed. Diseases of the Orbit. 2nd ed. Philadelphia: Lippincott, Williams & Wilkins; 2003: 169-212.
2. Shields JA. Inflammatory conditions that can simulate neoplasms. Dysthyroid orbitopathy. In: Shields JA, ed. Diagnosis and Management of Orbital Tumors. Philadelphia: WB Saunders; 1989:69-72.
3. Bartalena L, Wiersinga WM, Pinchera A. Graves’ ophthalmopathy: state of the art and perspectives. J Endocrinol Invest 2004;27:295-301.
4. Hufnagel TJ, Hickey WF, Cobbs WH, et al. Immunohistochemical and ultrastructural studies on the exenterated orbital tissues of a patient with Grave’s disease. Ophthalmology 1984;91:1411-1419.
5. Larsen DA, Ehlers N, Bek T. Thyroid-associated orbitopathy (TAO) treated by lateral orbital decompression. Acta Ophthalmol Scand 2004;82:108-109.
6. Prabhakar BS, Bahn RS, Smith TJ. Current perspective on the pathogenesis of Graves’ disease and ophthalmopathy. Endocr Rev 2003;24:802-835.
7. Chavis PS. Thyroid and the eye. Curr Opin Ophthalmol 2002;13:352-356.
8. Gorman CA, Garrity JA, Fatourechi V, et al. The aftermath of orbital radiotherapy for Graves’ ophthalmopathy. Ophthalmology 2002;109:2100-2107.
9. Konuk O, Atasever T, Unal M, et al. Orbital gallium-67 scintigraphy in Graves’ ophthalmopathy. Thyroid 2002;12:603-608.
10. Hatton MP, Rubin PA. The pathophysiology of thyroid-associated ophthalmopathy. Ophthalmol Clin North Am 2002;15:113-119.
11. Inoue Y, Tsuboi T, Kouzaki A, et al. Ophthalmic surgery in dysthyroid ophthalmopathy. Thyroid 2002;12:257-263.
12. Chu YK, Kim SJ, Lee SY. Surgical treatment modalities of thyroid ophthalmopathy. Korean J Ophthalmol 2001;15:128-132.
13. Clauser L, Galie M, Sarti E, et al. Rationale of treatment in Graves ophthalmopathy. Plast Reconstr Surg 2001;108:1880-1894.
14. Bradley EA. Graves ophthalmopathy. Curr Opin Ophthalmol 2001;12: 347-351.
15. Bahn RS. Understanding the immunology of Graves’ ophthalmopathy. Is it an autoimmune disease? Endocrinol Metab Clin North Am 2000;29: 287-296.
16. Bartalena L, Pinchera A, Marcocci C. Management of Graves’ ophthalmopathy: reality and perspectives. Endocr Rev 2000;21:168-199.
17. Gorman CA, Garrity JA, Fatourechi V, et al. A prospective, randomized, double-blind, placebo-controlled study of orbital radiotherapy for Graves’ ophthalmopathy. Ophthalmology 2001;108:1523-1534.
Thyroid-Related Ophthalmopathy (TRO)
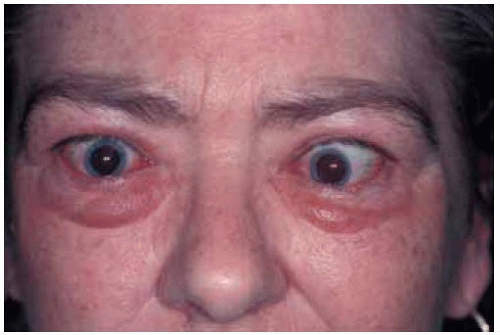
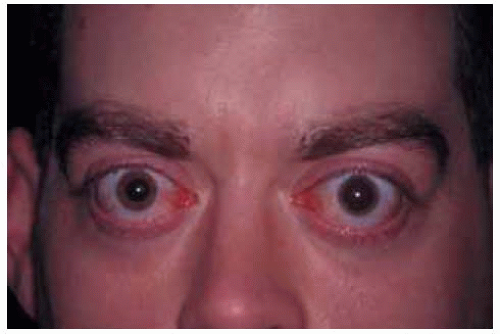
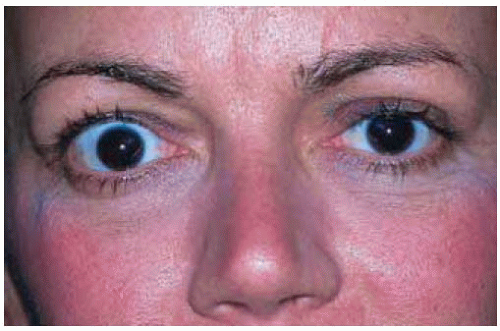 Figure 26.1. Thyroid-related ophthalmopathy with characteristic proptosis and eyelid retraction affecting mainly the right eye in a 35-year-old woman. |
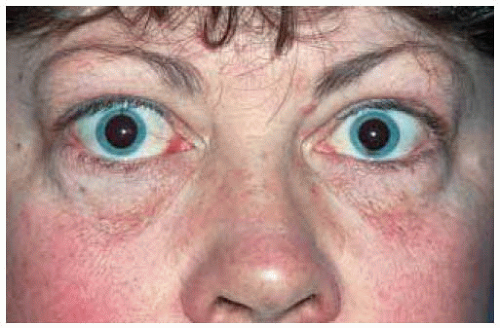 Figure 26.2. Thyroid-related ophthalm opathy with bilateral symmetric proptosis and eyelid retraction in a 43-year-old woman. |
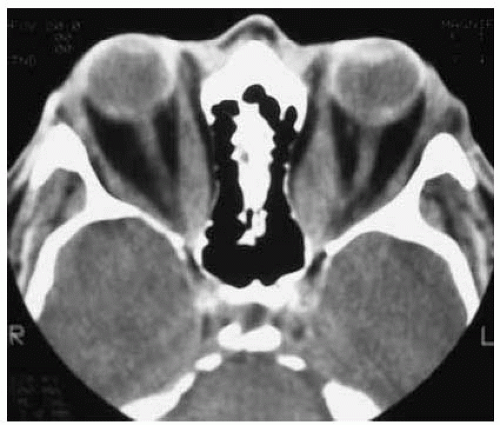 Figure 26.3. Axial computed tomography showing typical enlargement of extraocular muscles with sparing of the tendons. In this case, the medial rectus muscles are mainly involved. |
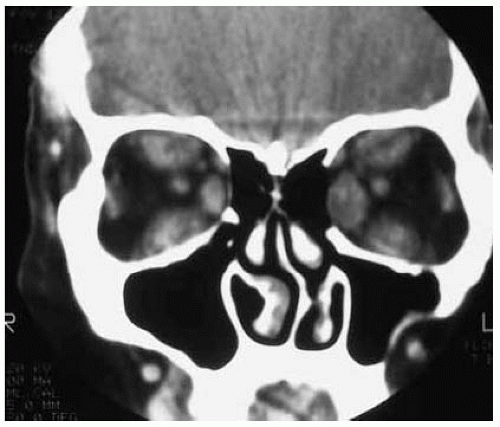 Figure 26.4. Coronal computed tomography through the midportion of the orbit, showing enlargement of several rectus muscles. |
Thyroid-Related Ophthalmopathy: Clinical and Radiologic Variations
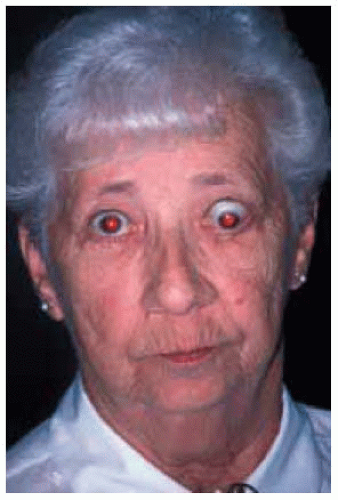 Figure 26.8. Woman with retraction of right upper eyelid and more severe involvement of the left eye with downward displacement of the globe. |
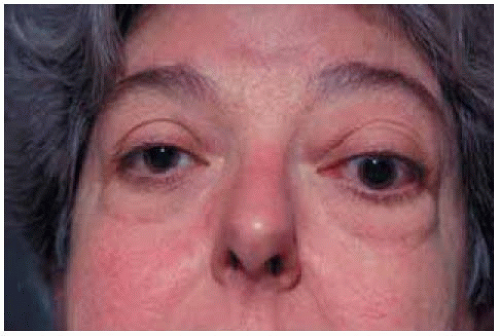 Figure 26.10. Unilateral TRO involving left orbit in a 57-year-old woman. In this instance, there is proptosis of left eye with less edema and eyelid retraction. |
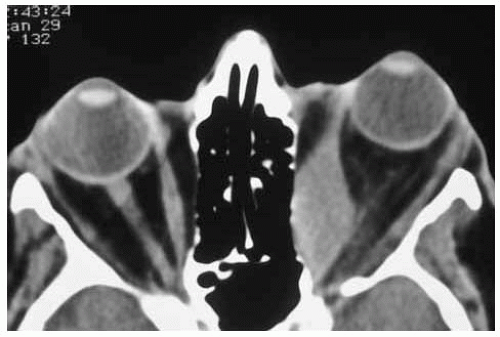 Figure 26.11. Axial computed tomography of patient shown in Figure 26.10, demonstrating marked enlargement of left medial rectus muscle but no evident enlargement of other extraocular muscles. Note the sparing of the tendon of the rectus muscle. Myositis from non-TRO causes would be more likely to affect the tendon. |
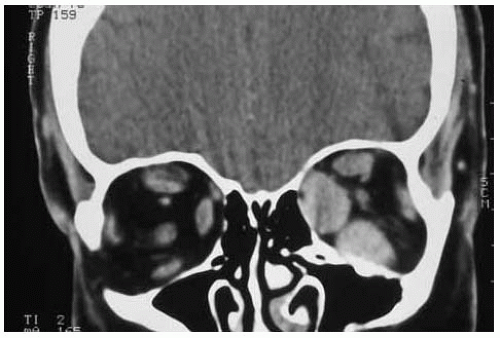 Figure 26.12. Coronal computed tomography of patient shown in Figure 26.10, demonstrating marked enlargement of left medial and inferior rectus muscles in left eye. |
Orbital Cellulitis
General Considerations
Acute orbital cellulitis is an infection of the soft tissues of the orbit and adjacent structures. It can affect children or adults. It most commonly occurs as a result of infectious extension from the ethmoid or frontal sinuses (1,2,3,4,5,6,7,8). However, it can also develop by contiguous spread from sites of trauma or infection, including dacryocystitis and conjunctivitis. Mucocele and mucopyocele, which can also extend into the orbit from the sinuses, are discussed in Chapter 27. Infectious orbital inflammation has been classified into five categories (1):
Group 1: Preseptal cellulitis
Group 2: Orbital cellulitis
Group 3: Subperiosteal abscess
Group 4: Orbital abscess
Group 5: Cavernous sinus thrombosis
Based on traditional terminology, we use the umbrella term “orbital cellulitis” to include all of these categories. Although the classification suggests an orderly progression, that is not necessarily the case and clinical onset can vary considerably.
Clinical Features
The clinical findings in orbital cellulitis vary with the inciting cause and the category and severity of the disease. The patient can present with any combination of pain, eyelid edema, conjunctival hyperemia, discharge, proptosis, displacement of the globe, and diplopia. Cavernous sinus thrombosis (group 5) can induce severe symptoms and was often fatal before the advent of antibiotics. In some instances, an orbital abscess can be localized and simulate a cyst or neoplasm. In a child, rhabdomyosarcoma and other malignant tumors can produce inflammatory signs and should be considered in the differential diagnosis (4). It should also be remembered that necrotic retinoblastoma can cause secondary inflammatory signs and simulate orbital cellulitis (7,8).
Diagnostic Approaches
History and physical examination should be directed toward detecting an inciting cause. Imaging studies like computed tomography and magnetic resonance imaging can detect primary infection of the sinuses, determine the extent of the orbital disease, and allow classification of the infectious process.
Pathology
The pathology of orbital cellulitis varies with the cause of the infection and the inciting organism. Histopathologically, the tissues are infiltrated or replaced by acute and chronic inflammatory cells and the inciting organisms can often be identified. Most common organisms include Streptococcus species, Staphylococcus aureus, and Staphylococcus epidermidis. Haemophilus influenza was previously the most common cause, especially among children. However, it has become uncommon since the introduction of specific vaccines.
Management
Treatment generally involves microscopic study and cultures of the discharge and appropriate antibiotic therapy (2). Surgical drainage is often helpful. The prognosis is generally good unless severe cavernous sinus thrombosis develops.
Selected References
1. Chandler JR, Langenbrunner DJ, Stevens ER. The pathogenesis of orbital complications in acute sinusitis. Laryngoscope 1970;80:1414-1428.
2. Rootman J. Inflammatory diseases. In: Rootman J, ed. Diseases of the Orbit. 2nd ed. Philadelphia: Lippincott, Williams & Wilkins; 2003:455-506.
3. Hornblass A, Herschorn BJ, Stern K, et al. Orbital abscess. Surv Ophthalmol 1984;29:169-178.
4. Cota N, Chandna A, Abernethy LJ. Orbital abscess masquerading as a rhabdomyosarcoma. J AAPOS 2000;4:318-320.
5. Shovlin JP. Orbital infections and inflammations. Curr Opin Ophthalmol 1998;9:41-48.
6. Tovilla-Canales JL, Nava A, Tovilla y Pomar JL. Orbital and periorbital infections. Curr Opin Ophthalmol 2001;12:335-341.
7. Shields JA, Shields CL, Suvarnamani C, et al. Retinoblastoma manifesting as orbital cellulitis. Am J Ophthalmol 1991;112:442-449.
8. Mullaney PB, Karcioglu ZA, Huaman AM, et al. Retinoblastoma associated orbital cellulitis. Br J Ophthalmol 1998;82:517-521.
Orbital Cellulitis and Abscess
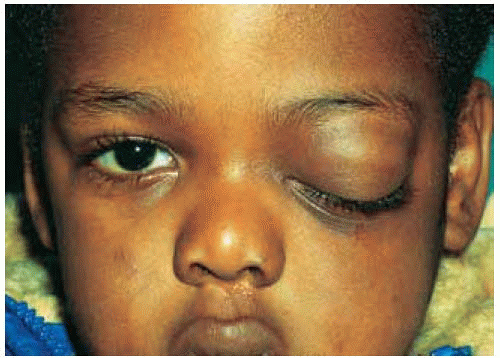 Figure 26.13. Acute orbital cellulitis secondary to ethmoid sinusitis and subperiosteal abscess in a 3-year-old child, showing eyelid swelling and blepharoptosis. |
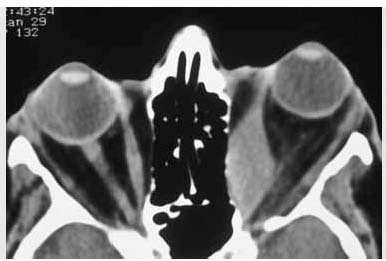
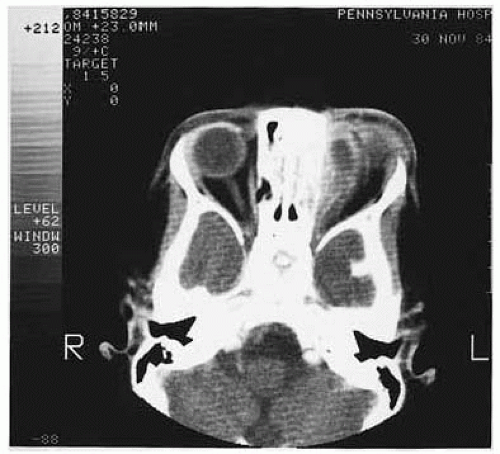 Figure 26.14. Axial computed tomography of patient shown in Figure 26.13, demonstrating ethmoiditis and left subperiosteal abscess. |
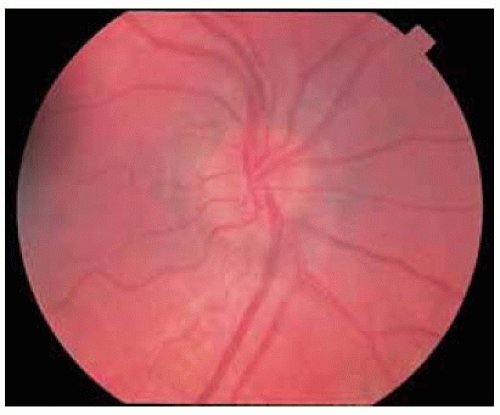
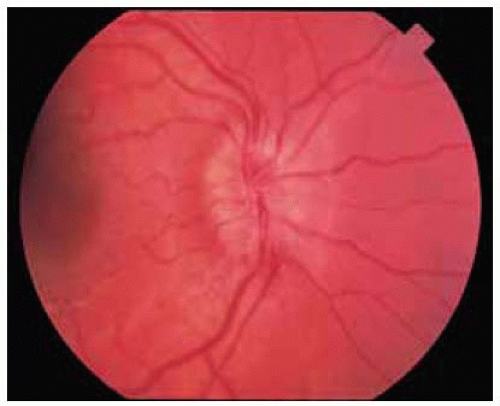 Figure 26.15. Fundus photograph showing edema and hyperem ia of right optic disc in an 8-year-old girl with circumscribed orbital abscess compressing the optic nerve. |
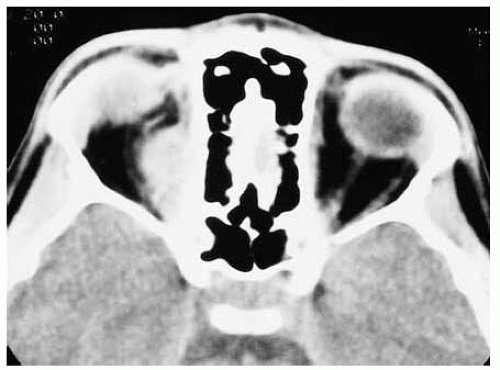 Figure 26.16. Axial computed tomography of patient shown in Figure 26.15 revealing circumscribed lesion in orbit. The peripheral aspects of the lesion demonstrated gadolinium enhancement with magnetic resonance imaging. |
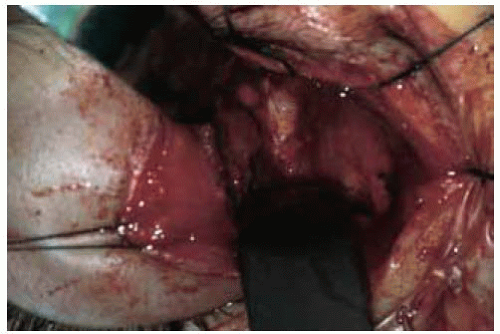 Figure 26.17. Surgical view showing yellow purulent material coming from abscess discovered at the time of surgical exploration. No organisms were found on stains and cultures. |
Orbit: Idiopathic Nongranulomatous Orbital Inflammation (“Inflammatory Pseudotumor”)
General Considerations
“Nonspecific orbital inflammation (NOI)” is traditionally used to describe a nongranulomatous inflammatory process within the orbit that has no demonstrable cause (1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21). The term “pseudotumor” is sometimes applied to this condition. However, that term can apply to any lesion that simulates a neoplasm, including granulomatous inflammation, abscess, and amyloidosis. Therefore, we prefer to avoid “pseudotumor,” and prefer to call this condition idiopathic nongranulomatous orbital inflammation (INOI). There is considerable confusion regarding this condition and the definition, terminology, and classification will most certainly change as it is better understood and more specific etiologies are identified.
In the authors’ series of 1,264 orbital masses, the 98 cases of INOI accounted for 74% of inflammatory-infectious diseases and for 8% of all orbital lesions (1). In a study of orbital tumors in older patients, it accounted for 8% of lesions among patients .60 years of age (17). INOI can affect both adults and children (5). The mean age at diagnosis is about 45 years with an age range of 2 to 92 years. About 25% are bilateral (10). Depending on the tissues involved and the orbital location, INOI has been divided into subcategories, including myositis, dacryoadenitis, anterior, diffuse, and posterior (apical) (3).
Clinical Features
The clinical features of INOI vary with the subcategory of the disease. Myositis affects one or more rectus muscles and produces a rather acute onset of pain that is worse with eye movements, and injection of the muscle insertion. Central vision is usually good. Dacryoadenitis shows superotemporal eyelid and conjunctival swelling, pain, and a tender mass in the lacrimal gland area. The central vision is usually good. The anterior variant is characterized by pain, eyelid edema, conjunctival chemosis, and limitation of ocular motility. Affected patients can have signs of diffuse uveitis and retinal detachment, findings similar to Harada disease. The diffuse variant demonstrates symptoms and signs similar to the anterior variety. However, the findings are more severe and proptosis is more pronounced. The apical variant of INOI causes more severe pain, visual loss, and motility disturbance, but less anterior segment inflammation. The pain is particularly severe with ocular movement. The painful ophthalmoplegia is similar to the Tolosa-Hunt syndrome.
In some instances, INOI can extend beyond the orbit into the paranasal sinuses and brain. Some such patients may have only minimal symptoms and signs to suggest neurologic extension, but it can be recognized on neuroimaging (18). Severe cases can rarely present with central retinal artery and vein obstruction secondary to compression of the globe (19).
Differential Diagnosis
The differential diagnosis of INOI includes a variety of inflammatory and neoplastic conditions. Myositis can closely resemble thyroid-associated orbitopathy or metastatic carcinoma. Any variant must be differentiated from lymphoma, metastatic carcinoma, and other circumscribed or diffuse orbital masses.
Diagnostic Approaches
Findings on computed tomography and magnetic resonance imaging vary with the location and extent of involvement. Myositis shows enlargement of one or more rectus muscles, often with involvement of the muscle tendons, a finding that helps to differentiate it from thyroid-related orbitopathy. Dacryoadenitis shows enlargement of the lacrimal gland and inflammatory signs and may cause findings similar to a ruptured dermoid cyst. Anterior and diffuse involvements show an ill-defined mass and choroidal thickening and retinal detachment. The apical variant shows a diffuse or ill-defined mass at the orbital apex with compression of nerves and muscles. As mentioned, neuroimaging studies can sometimes detect extraorbital extension of INOI (18). In patients with classic symptoms and signs, an orbital biopsy is generally not indicated unless the lesion fails to respond to medical therapy (see Management).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree