3 Inflammation
INTRODUCTION
During the evolutionary struggle for survival, a complex series of defense mechanisms (that we in its totality call inflammation) has evolved. The inflammatory response involves a variety of specialized effector cells and a bewilderingly complex interplay of cells, mediators, and biochemical reactions that serves to protect the body against microorganisms and cancer. In addition, the inflammatory process includes mechanisms to repair and restore tissues that have been damaged by foreign invaders, trauma, or chemical and physical agents. The wide spectrum of opportunistic infections and tumors that afflict patients who have the acquired immunodeficiency syndrome (AIDS) underscores generally how effective the intact immune system, the cornerstone of the inflammatory process, is in protecting us from potential invaders.
Fantone and Ward defined inflammation as “a reaction of the microcirculation characterized by movement of fluid and white blood cells from the blood into the extravascular tissues. This is frequently an expression of the host’s attempt to localize and eliminate metabolically altered cells, foreign particles, microorganisms, or antigens.” The inflammatory cells and the biochemical mediators of inflammation largely reside within the lumina of blood vessels. In contrast, the majority of the body’s cells are located in the extravascular compartment, where invasion by microorganisms also generally begins. Hence, inflammatory cells and macromolecules such as antibodies and components of the complement system must leave the vessels and enter the tissues if they are to combat microbial invaders or dispose of dead or damaged cells or other materials. Vasoactive inflammatory mediators such as histamine, serotonin, kinins, prostaglandins, and platelet activating factor cause vasodilation and increase vascular permeability, allowing cells and antibodies access to the tissues. A second heterogenous group of nonimmunoglobulin protein mediators called cytokines recruit and stimulate inflammatory cells. Cytokines are synthesized and secreted by inflammatory cells and include the interleukins, interferons, and colony stimulating factors. Other cell adhesion molecules including selectins, integrins, and cadherins are involved in cellular homing, adhesion, and cell-to-cell interactions. The details of this complex series of interactions are beyond the scope of this introductory chapter.
Vasodilation and increased vascular permeability are responsible for several of the cardinal manifestations of inflammation including tumor or swelling, calor or heat, and rubor or redness (Fig. 3-1). Heat and redness reflect increased blood flow and swelling, the collection of serum and other blood components in the extracellular space. Some inflammatory mediators (e.g., some of the prostaglandins) cause pain and stimulate the contraction of smooth muscle. Spasm of the ciliary muscle and sphincter muscle of the iris contributes to the pain of anterior uveitis, which usually is ameliorated by strong cycloplegic drugs such as atropine. Pain, pupillary miosis, and photophobia are helpful clinical markers that serve to differentiate iridocyclitis from conjunctivitis.
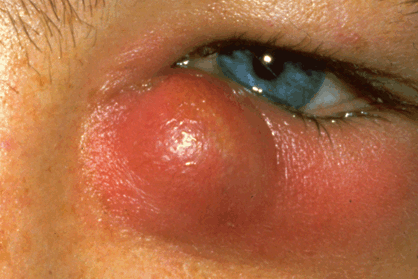
Fig. 3-1. Acute dacryocystitis. Signs of acute inflammation including swelling and erythema are evident in the region of infected lacrimal sac and lower lid.
The increased vascular permeability that occurs in inflammation is readily evident to the ophthalmologist during slit lamp biomicroscopy. Aqueous humor normally is almost totally devoid of protein. The protein content rises when inflammation disrupts the blood–ocular barrier. A focused beam of light illuminating the anterior chamber becomes visible, just as a projector beam is visible in a smoky room. This phenomenon, which is termed aqueous “flare” or “ray,” is caused by the Tyndall effect. The intensity of the aqueous flare correlates fairly well with the severity of the inflammation and may be roughly quantified and noted in the clinical record. Individual inflammatory cells also are evident on slit lamp examination as motes of light. The quantity of cells is also roughly estimated and recorded. The cells normally move with the convection currents in the aqueous. (They sink anteriorly where the aqueous is cooled by the cornea and rise posteriorly where the aqueous is heated by the iris.) Absence of cellular convection currents may indicate clotting of fibrin-rich aqueous in a case with severe vascular permeability. Adhesions readily form in the fibrin-rich milieu of ocular inflammation. Adhesions between the iris and the lens called posterior synechiae can block the flow of aqueous humor through the pupil from the posterior chamber. The pupil is said to be secluded if its entire circumference is bound down by posterior synechiae (seclusio pupillae). Secondary closed-angle glaucoma can develop if the synechiae are not broken. Cycloplegic/mydriatic drugs help to prevent these adhesions by dilating the pupil. Aggregates of inflammatory cells called keratic precipitates or KPs form on the posterior surface of the cornea. The KPs may be small or large and lardaceous. The latter, which are often called “mutton-fat” keratin precipitates, typically occur in eyes that have chronic granulomatous inflammation.
Functio laesa or loss of function is the final cardinal manifestation of inflammation. Although many common ocular inflammations such as conjunctivitis or chalazion are short-lived incapacitations or annoyances, severe inflammations or infections, particularly those that affect the interior of the eye, can cause blindness. Stereotyped inflammatory responses that are designed to protect the body against external invaders can totally destroy the delicate tissues of the eye. A small bacterial infection may be inconsequential in the skin, but it can totally destroy an eye. Even if an intraocular bacterial infection is expediently sterilized, the normal processes of regeneration and repair often cause blindness. A delicate membrane of connective tissue ˂1 mm long can profoundly affect visual acuity if it forms in an inopportune location. Likewise, minor alterations in the structure of the transparent ocular media (cornea, lens, or vitreous) can markedly degrade their optical properties. One must also remember that the eye’s neurosensory components are incapable of regeneration or repair like central nervous system tissue. Blindness caused by retinal damage or destruction is irrevocable and untreatable.
THE CLASSIFICATION OF INFLAMMATION
Histopathologically, inflammation is categorized into acute and chronic categories based on the type of inflammatory cells that are found in the tissue or exudate. Acute inflammation usually is characterized by the presence of polymorphonuclear leukocytes or “polys.” Lymphocytes and plasma cells are found in chronic nongranulomatous inflammation, and their presence generally denotes involvement of the immune system. Activated macrophages or epithelioid histiocytes and inflammatory giant cells characterize chronic granulomatous inflammation.
POLYMORPHONUCLEAR LEUKOCYTES
The polymorphonuclear leukocyte, neutrophil, or poly is the primary cell found in acute inflammation (Fig. 3-2A). The polymorphonuclear leukocyte is the body’s first line of cellular defense. These cells phagocytize bacteria and other foreign material, and their cytoplasm contains many primary and secondary granules that harbor a wide variety of digestive enzymes that they use to kill and digest microorganisms. Polys have pink cytoplasm and a multilobed (typically trilobed) nucleus in routine sections stained with hematoxylin and eosin (H&E). Degenerated polys with round karyorrhectic nuclei frequently are observed in focal collection of polys called abscesses. The term suppurative inflammation refers to the presence of an exudate called pus, which is composed of numerous polys and tissue destruction. Polymorphonuclear leukocytes do not proliferate at the site of inflammation. They are produced in the bone marrow and delivered via the blood stream to the site of inflammation, where they die. They are attracted to the site of injury by chemotactic gradients, adhere to receptors or adhesion molecules on the vascular endothelial cells (margination), and pass through the capillary wall into the tissue (diapedesis). The cell walls of polys have receptors for the Fc component of immunoglobulin. These Fc receptors aid in the phagocytosis of bacteria that have been bound to antibodies in a process called opsonization.
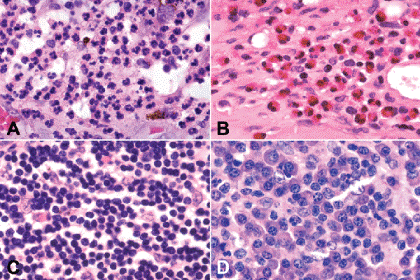
Fig. 3-2. Inflammatory cells. A. Polymorphonuclear leukocytes. Polys have multilobed nuclei and eosinophilic cytoplasm. The polys in this field are well preserved. A few mononuclear histiocytes also are present in the acute inflammatory infiltrate. B. Eosinophils. The cytoplasm of eosinophils contains intensely eosinophilic granules. The nuclei are bilobed. The presence of eosinophils usually suggests the presence of allergy or a parasite. C. Lymphocytes. Lymphocytes have round, intensely basophilic nuclei and scanty cytoplasm that usually is inapparent in routine light microscopic sections. Many subtypes of lymphocytes can be identified with special immunohistochemical stains. D. Plasma cells. Plasma cells have round, eccentrically located nuclei with a cartwheel or clock-face pattern of chromatin clumping. The cytoplasm is basophilic because it contains large quantities of ribosomal RNA used in antibody synthesis. The Golgi apparatus is evident light microscopically as a perinuclear “hof.” (A–D. H&E ×250)
Clinically, acute inflammation is characterized by the presence of pus. Copious quantities of purulent exudate occur in patients who have hyperacute conjunctivitis such as that caused by gonococcus. A layered collection of polys called a hypopyon accumulates in the inferior part of the anterior chamber in eyes with acute keratitis or endophthalmitis. Vitreous abscesses form in acute purulent endophthalmitis. The polys in the vitreous abscess occasionally are arranged in a linear fashion, reflecting the orientation of the type II collagen fibrils that constitute the framework of the vitreous humor.
EOSINOPHILS
Eosinophilic leukocytes or eosinophils are recognized by their intensely eosinophilic, orange, granular cytoplasm and their bilobed nuclei (Fig. 3-2B). Eosinophils are about the same size as polys. Their cytoplasmic granules have a characteristic rhomboid crystalloid configuration disclosed by electron microscopy. The eosinophil granules are rich in acid phosphatase and other lysosomal enzymes and also contain a unique eosinophilic major basic protein that is toxic to certain parasites and normal host cells. Eosinophils are involved in the phagocytosis of antigen antibody complexes and are known to modulate inflammatory reactions mediated by mast cells. The presence of numerous eosinophils in tissue sections is highly suggestive of either an allergic reaction or a parasitic infestation.
LYMPHOCYTES
Lymphocytes are mononuclear cells that are 7 to 8 μm in diameter. Lymphocytes appear as blue spheres in smears and tissue sections; their nuclei are round and intensely basophilic, and the cytoplasm is so scanty that it is often inapparent (Fig. 3-2C). Lymphocytes play a dominant role in chronic inflammation and in both humoral and cell-mediated immunity. Multiple subtypes of lymphocytes have been characterized. B lymphocytes are formed in the bone marrow and are involved in humoral immunity. B lymphocytes differentiate into plasma cells, the chief antibody-producing cells of the body. T lymphocytes, which originate in the thymus, include effector and regulatory subtypes. Effector T cells participate in delayed hypersensitivity and mixed lymphocyte reactions and are a prominent constituent of benign reactive lymphoid infiltrates. Regulator T cells (T-helper/amplifier [T4] and suppressor/cytotoxic [T8] cells) modulate the immune response. T4 cells, which are responsible for initiating the immune response, are preferentially infected and killed by human immunodeficiency virus (HIV), which binds to the CD4 receptor. Other lymphocyte subtypes include killer, natural killer, and null cells.
PLASMA CELLS
Plasma cells are activated B lymphocytes. Plasma cells are the body’s primary source of circulating antibodies. These antibody factories have a characteristic appearance (Fig. 3-2D). Round and eccentrically located, the nucleus has dense clumps of chromatin that adhere to the inner surface of its membrane in a pattern that has been likened to a cart wheel or clock face. Unlike lymphocytes, plasma cells have an abundant quantity of cytoplasm, which is largely occupied by rough endoplasmic reticulum (RER) used to synthesize immunoglobulin. In routine sections stained with H&E, the cytoplasm of plasma cells has a distinctly basophilic or purple hue caused by the affinity of the basic dye hematoxylin for ribosomal RNA in the RER. The Golgi apparatus of plasma cell is apparent light microscopically as a lighter staining crescent next to the nucleus called the perinuclear “hof” (German, “courtyard”). The cytoplasm of plasma cells may become eosinophilic as the cells produce large quantities of immunoglobulin. Eosinophilic crystals of immunoglobulin called Russell bodies occasionally form in the cytoplasm of plasma cells (Fig. 3-3). Usually round, but occasionally square or even hexagonal, Russell bodies reflect the “terminal constipation” plasma cells by immunoglobulin. They generally denote an inflammatory process of some chronicity and may be found intracellularly or free in the tissue. Dutcher bodies are similar smaller crystalline inclusions of antibody molecules that appear to be intranuclear but actually reside in an intranuclear cytoplasmic inclusion. Positive staining with the periodic acid-Schiff (PAS) stain indicates that Russell or Dutcher bodies are composed either of IgA or IgM molecules. A cell that contains multiple small intracytoplasmic Russell bodies is called a morula cell of Mott.
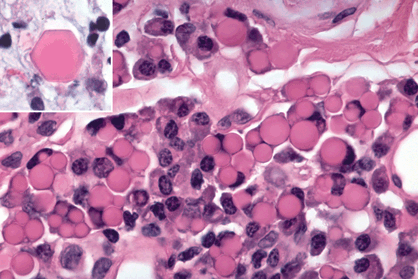
Fig. 3-3. Russell bodies. The uniformly eosinophilic spherules in this chronic inflammatory infiltrate are crystalloids of immunoglobulin called Russell bodies. A rare hexagonal Russell body is seen in the inset. (Both figures, H&E ×250)
An inflammatory infiltrate composed of lymphocytes and varying numbers of plasma cells characterizes chronic nongranulomatous inflammation. Although lymphocytes and plasma cells occasionally constitute an acute inflammatory response to certain viral infections, the presence of these cells usually indicates that the immune system has been activated (Fig. 3-4). Special stains for microorganism generally are nonrevealing in chronic nongranulomatous inflammation.
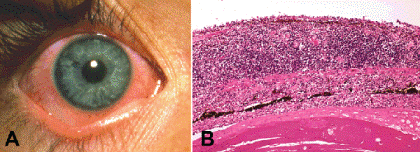
Fig. 3-4. Iritis. A. The inflamed eye had pupillary miosis and an anterior chamber reaction. No exudate is seen. The patient had pain and photophobia. B. Histopathology exam of another case shows massive thickening of the iris stroma by an infiltrate of lymphocytes and plasma cells. The disrupted iris pigment epithelium adheres to the anterior surface of the cataractous lens. No microorganisms were detected. (B. H&E ×50)
MAST CELLS
Mast cells are often called tissue basophils, although there is evidence that they are derived from different precursor cells in the bone marrow. Mast cells play an extremely important role in acute anaphylaxis (Type I hypersensitivity reaction). IgE antibody molecules made by allergic individuals bind to Fc receptors on the plasma membrane of mast cells. Subsequent interaction between the appropriate antigen and two IgE molecules causes mast cell degranulation and the release of potent vasoactive substances including histamine, serotonin, and heparin, which have an immediate effect on vascular permeability. Severe itching and the acute onset of conjunctival edema or chemosis are clinical symptoms and signs of acute allergic conjunctivitis of the anaphylactic type (Fig. 3-5B).
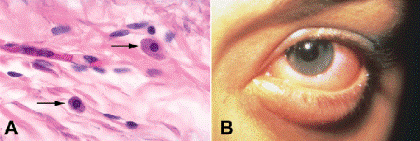
Fig. 3-5. A. Mast cells. Mast cells (arrows) in tissue sections have round, centrally located nuclei. The cytoplasm is mildly basophilic in routine sections stained with H&E and stains intensely with PAS. B. Acute allergic conjunctivitis. Conjunctival chemosis is evident in the photo of a laboratory worker with acute anaphylactic conjunctivitis. The patient was allergic to animal dander and experienced the sudden onset of severe itching.(A. H&E ×250)
Mast cells vaguely resemble plasma cells in tissue sections but have a centrally placed nucleus (Fig. 3-5A). In routine H&E sections, they lack the prominent array of basophilic granules disclosed by Wright stain. The cytoplasm of mast cells is PAS-positive, and its constituent granules are stained intensely blue by the acid-fast stain. Mast cells are found in the conjunctival substantia propria in allergic disorders such as vernal conjunctivitis and giant papillary conjunctivitis. They are also common in some neoplasms such as neurofibromas.
MACROPHAGES
Macrophages or histiocytes are the body’s second line of cellular defense and its chief phagocytic cell. Macrophages are derived from circulating monocytes. They are relatively large mononuclear cells (larger than polys) that have an eccentric reniform or kidney-shaped nucleus. Macrophages have a great capacity to phagocytize material, but unlike polys they cause little tissue damage. Prior to phagocytosis, newly formed macrophages have a modest amount of eosinophilic cytoplasm. In ophthalmic pathology, macrophages generally are characterized by the substances that they have phagocytized. They include macrophages that have ingested blood breakdown products such as hemosiderin and erythrocyte ghost cells, lipid material seen as foamy vacuoles, and other materials such as degenerated lens protein or melanin (Fig. 3-6).
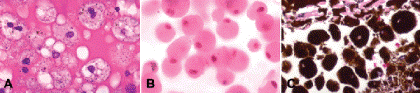
Fig. 3-6. Macrophages. A. Lipid-laden macrophages, subretinal fluid. The frothy vacuolated cytoplasm is filled with lipid vacuoles and scattered elliptical granules of RPE melanin. The surrounding subretinal fluid is protein-rich and intensely eosinophilic. The patient had radiation retinopathy. B. Lens-laden macrophages, phacolytic glaucoma. These macrophages have copious quantities of eosinophilic cytoplasm that reflects the ingestion of degenerated lens protein released from an advanced cortical cataract. C. Melanophages, melanocytomalytic glaucoma. Macrophages have ingested melanin pigment released from necrotic iris melanocytoma. (A–C. H&E, original magnification ×250)
Macrophages play an important role as antigen-presenting cells in the initial stages of the immune response. They phagocytize and process antigenic material and present appropriate epitopes to helper T cells in conjunction with class II major histocompatibility molecules (called HLA-DR in humans), which are located in their cell membranes. During activation, macrophages produce a lymphokine called interleukin-1 (IL-1) that produces fever and is thought to induce the production of a second cellular messenger interleukin-2 by the T cells. They also secrete a wide variety of powerful biologic molecules called monokines that are important, even pivotal, participants in the inflammatory response.
EPITHELIOID HISTIOCYTES AND INFLAMMATORY GIANT CELLS
Under certain circumstances macrophages or histiocytes transform into more metabolically active forms called epithelioid cells or epithelioid histiocytes (see Fig. 3-9A, later in this chapter). Activation typically occurs when the histiocytes encounter large quantities of antigenic material that is relatively insoluble or indigestible. Some microorganisms, particularly those that proliferate intracellularly, stimulate the formation of epithelioid histiocytes. Classic examples include the mycobacteria that cause tuberculosis and leprosy, fungi, and parasites such as schistosomes.
Epithelioid histiocytes are termed “epithelioid” (-oid, resembling; epithelioid, like epithelium) because these cells have abundant eosinophilic cytoplasm and superficially resemble simple epithelial cells. If epithelioid histiocytes are observed histopathologically in a chronic inflammatory infiltrate, the inflammation is termed granulomatous. Epithelioid histiocytes are required for the diagnosis of chronic granulomatous inflammation. Inflammatory giant cells are another characteristic feature of chronic granulomatous inflammation (Figs. 3-7 and 3-8). Inflammatory giant cells are a multinucleated syncytium formed by the fusion of epithelioid histiocytes.
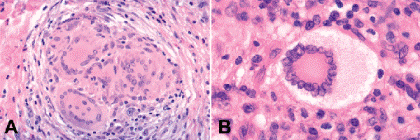
Fig. 3-7. A. Inflammatory giant cells, Langhans type. The nuclei are located peripherally. The cytoplasm is uniformly eosinophilic. Langhans giant cells are found in tuberculosis and other granulomatous diseases. B. Touton giant cells, juvenile xanthogranuloma. Fully developed Touton giant cells have a target configuration. A peripheral rim of frothy lipidized cytoplasm surrounds a ring of nuclei which in turn encompasses a central bull’s eye of eosinophilic cytoplasm. Touton giant cells are found in JXG and other xanthogranulomatous diseases. (A. H&E ×100, B. H&E ×250)
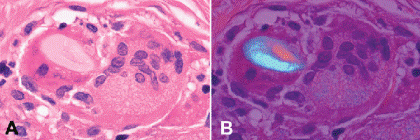
Fig. 3-8. Foreign body giant cell. A. The cytoplasm of the giant cell at left contains an oval cellulose fiber. B. The foreign body shows vivid birefringence during polarization microscopy. The nuclei of foreign body giant cells are arranged haphazardly. (A. H&E ×250, B. H&E with crossed polarizers ×250)
Several kinds of inflammatory giant cells are recognized histopathologically. The Langhans giant cell, which typically is seen in tuberculosis, has a peripheral rim of nuclei and homogenous cytoplasm (Fig. 3-7A). Foreign body giant cells have nuclei, which are randomly dispersed or are centrally located, and their cytoplasm contains particulates of foreign material in large vacuoles (Fig. 3-8). When an extremely large foreign body is encountered, numerous foreign body giant cells adhere to its outer surface, forming an encompassing cytoplasmic barrier that “insulates” the foreign material from the rest of the body. The Touton giant cell occurs in chronic xanthogranulomatous inflammation (Fig. 3-7B). The classic Touton giant cell is shaped like a target. The “bulls-eye” is a central zone of eosinophilic cytoplasm, which is encircled by a ring of nuclei, which in turn is surrounded by a peripheral wreath of foamy lipidized cytoplasm. Although Touton giant cells classically are associated with juvenile xanthogranuloma (JXG), they also are found in other xanthogranulomatous disorders that affect the ocular adnexa including Erdheim-Chester disease, necrobiotic xanthogranuloma with paraproteinemia, and orbital xanthogranuloma with adult-onset asthma.
GRANULOMATOUS INFLAMMATION
By definition, to be classified as chronic granulomatous, an inflammatory infiltrate must contain epithelioid histiocytes and/or inflammatory giant cells. Clinically, the term granulomatous is applied to ocular inflammation when large lardaceous “mutton-fat” KPs are observed. Mutton-fat KPs are miniature granulomas composed of aggregated epithelioid histiocytes. If granulomatous inflammation is noted clinically or histopathologically, a causative organism or specific etiologic agent should be sought. If granulomatous inflammation is encountered in tissue sections, it is imperative that stains for acid-fast organisms, fungi, and bacteria (and occasionally silver stains for spirochetes) be performed. The specimen should also be examined with polarization microscopy to rule out the presence of foreign material that can be inconspicuous. Sarcoidosis is a relatively common cause of granulomatous ocular inflammation (Figs. 3-9 and 3-10A). A diagnosis of exclusion, sarcoidosis should be suspected when a characteristic pattern of discrete noncaseating granulomas is found and special stains for microorganisms are negative. Granulomatous inflammation occasionally is a response to endogenous material. Examples include the response to lipid in chalazia and the thick layer of giant cells bordering keratin in the lumen of dermoid cysts with discontinuous epithelial linings.
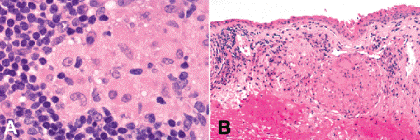
Fig. 3-9. A.Epithelioid histiocytes, sarcoidosis. The epithelioid histiocytes comprising the discrete granuloma have abundant quantities of eosinophilic cytoplasm and vesicular nuclei with nucleoli. B. Positive conjunctival biopsy, sarcoidosis. Substantia propria contains several discrete granulomas consistent with sarcoidosis. (A. H&E ×250, B. H&E ×100)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


