Implantable hearing devices have now become standard of care for the rehabilitation of many hearing loss conditions. Devices include cochlear implants (CIs), bone conduction systems, and also active middle ear implants (AMEIs). These are suitable for a variety of types of hearing loss, ranging from severe to profound sensorineural hearing loss, mixed conductive and sensorineural loss, or purely conductive loss.
Chronic otitis media (COM) may be a predisposing cause for the hearing impairment or it may be coincidental. Either way, the presence of active or inactive COM presents significant challenges in terms of management of the ear to ensure suitability for, and safety of, the implanted hearing device. The most common conditions to manage prior to implantation include tympanic membrane perforations, a radical mastoid cavity, and chronic mucosal disease of the middle ear.
Cochlear implants and AMEIs consist of a postaural receiver stimulator (RS) package and electronic cable with either an electrode in the inner ear (for CIs) or an actuator that drives the ossicular chain, round window, or stapes footplate (for AMEIs). Some AMEIs, like CIs, are partially implantable with a combination of internally implanted and externally worn components, such as the Vibrant Soundbridge (Med-El Corporation, Innsbruck, Austria) and Maxum (Ototronix, Houston, Texas). Totally implantable varieties of AMEIs have also been developed, such as the Esteem (Envoy Medical, Saint Paul, Minnesota), which has an implanted subcutaneous microphone; however, chronic ear disease has been set apart as a contraindication for implantation of the Esteem device by the manufacturer.
Bone conduction hearing aids, such as the Baha system (Cochlear Corporation, Sydney, Australia), use a postauricular osseointegrated screw fixture with a percutaneous connector. Recently, however, transcutaneous implantable bone conduction systems such as the Baha Attract (Cochlear Corporation, Sydney, Australia), BONEBRIDGE (Med-El Corporation, Innsbruck, Austria) and Sophono Alpha 2 MPO (Sophono Corporation, Boulder, Colorado), have become available. These are also positioned posterior to, or within, the mastoid and do not involve the middle ear cleft. Bone conduction implants offer the advantage of potential placement away from the infected tympanomastoid compartment while maintaining the ability to directly stimulate the cochlea and overcome any underlying conductive hearing loss.
The aim of management of an ear with COM in relation to an implantable device is first to eradicate any active disease, such as a chronic infection or cholesteatoma, or inactive disease, such as a dry perforation or retraction, and, second, to create a safe, stable ear with either an aerated tympanomastoid middle ear cleft or an obliterated ear with blind sac closure of the external auditory canal (EAC), with or without Eustachian tube plugging and soft tissue obliteration of the mastoid cavity.
Important principles are to achieve secure soft tissue cover of the postauricular RS package, without any incision crossing the package and no contact between the tympanic membrane or the skin of the EAC and the prosthesis, electrode cable, or actuator. At any point where there is contact, the soft tissue cover should be a thick vascularized layer. This will prevent extrusion of the postauricular RS package, the cable, or the actuator and will also prevent reactivation of infection; hence, eradication of biofilm is essential.
This chapter will provide an overview of the aims and surgical techniques utilized in patients with COM for the eradication of disease and creation of safe, stable ears with the various types of implanted devices described above.
43.2 Patient Selection
Most commonly, patients present with a sequela of long-standing COM, such as severe to profound sensorineural hearing loss, no longer helped by hearing aids, in which case cochlear implantation is indicated. Alternatively, they may present with conductive or mixed hearing loss, which may be suitable for an AMEI or a bone-anchored hearing solution. The type of COM may vary from a central perforation that is dry to one with active discharge. There may be gross atelectasis or frank cholesteatoma, or an open mastoid cavity or old fenestration cavity that may have active granulation and infection, or there may be a radical cavity.
One important factor relating to preimplantation COM surgery is that this may correct a conductive hearing loss and will optimize the use of conventional hearing aids and give a better assessment of the hearing level. The audiologic indications for cochlear implantation continue to evolve as the outcomes achieved with implants progressively improve. Indeed, there is now some overlap with the indications for AMEIs.
Bone-anchored hearing solutions, whether passive percutaneous, transcutaneous, or active (such as the BONEBRIDGE) are usually an option for patients with a conductive or mixed hearing loss with a mild sensorineural component, where the use of conventional hearing aids is not beneficial due to previous ear surgery, canal stenosis, chronic discharge, or congenital ear malformations such as atresia of the EAC. They may also be an option in single-sided deafness. For these devices to provide adequate benefit, the bone conduction thresholds need to be reasonably good, with best results achievable if normal.
An AMEI, such as the Vibrant Soundbridge (▶ Fig. 43.1), may be an option for patients with COM, as conventional hearing aids are sometimes insufficient after several surgical procedures and with poorly functioning middle ears.1 Typically, patients present with a radical cavity and a mixed hearing loss, an acquired stenosis of the EAC/lateralization of the TM, or after failed tympanoplasties and chronic otitis externa from hearing aid use. The Vibrant Soundbridge offers the option of a direct stimulus via the round window in cases where a normal, or at least adequate, middle ear is lacking. Coupling options to the stapes or footplate are also possible. Vibroplasty, a term used to describe tympano-ossicular reconstruction that incorporates the floating mass transducer (FMT) of the Vibrant Soundbridge, is potentially indicated for children older than 3 years or adult patients with an otherwise non-reconstructable conductive hearing loss or mixed hearing loss with a stable bone-conduction threshold above approximately 50 dB at 500–4000 Hz. Especially in patients with a mild to severe sensorineural hearing loss, monosyllabic word scores >50% are essential for a benefit with this device. The option of using the Vibrant Soundbridge for a round window application, or by prosthesis-coupling options, has widened the initial indications. However, in patients with a history of COM, an AMEI is indicated only after previous failed attempts of tympanoplasty and use of conventional hearing aids.
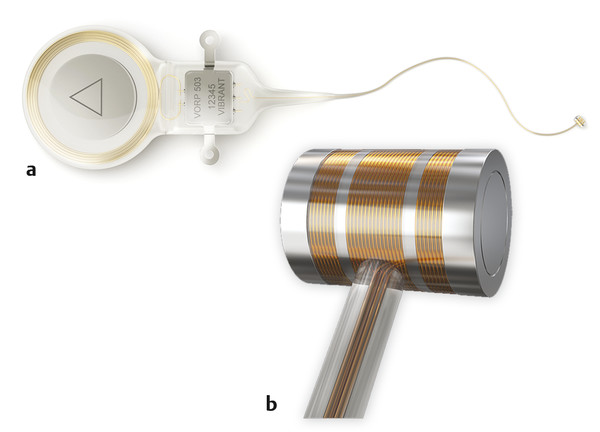
Fig. 43.1 (a) The implantable part of the Vibrant Soundbridge, the VORP, consists of a coil and a magnet to hold the audio processor, a demodulator, and a conductor link to (b) the FMT, a vibrating device converting the electrical signals into mechanical energy. It can be positioned on the ossicular chain or the round window.
(Used with permission from Med-El Corporation, Innsbruck, Austria.)
In terms of preoperative investigation, both a high-resolution computed tomography (CT) scan and magnetic resonance imaging (MRI) are valuable in patients with COM. Both modalities help demonstrate the extent of previous surgery and remaining bony and soft tissue anatomy as well as the presence of active infectious disease. Particularly for cochlear implantation, T2-weighted MRI is important to exclude fibrosis or ossification in the scala tympani basal turn. Preoperative cone-beam CT is an alternative to conventional CT for preoperative imaging. As the field of view is limited, it is particularly suitable for evaluations prior to implanting AMEIs and postoperative evaluation in cases of functional problems.2
Although MRI-compatible (in general to 1.5 tesla), all types of implants with an integrated magnet will give a shadow effect on postoperative MRI. This must be considered in patient selection as it might make future evaluation of the internal auditory canal or structures in the posterior fossa difficult. In reality, it is rare that this excludes implant candidates, but it may apply to side selection. The older Vibrant Soundbridge systems are not MRI-compatible as the FMT contains a magnet and, thus, is at risk of movement. The latest Vibrant Soundbridge 503 is MRI compatible at 1.5 tesla.
In the majority of cases with COM, a two-stage surgical approach is utilized to ensure the eradication of infection or cholesteatoma and stable healing. If the aim is an aerated middle ear cleft, an initial tympanomastoidectomy is performed using the surgeon’s preferred technique that would be predicted to have the greatest chance of long-term stability. Alternatively if the aim is obliteration of the middle ear cleft, a radical mastoidectomy and blind sac closure are performed, with or without fat or muscle packing of the cavity. The CI or AMEI is then placed at a second-stage procedure. A single-stage procedure can sometimes be safely performed if there is no active infection or cholesteatoma. Most often this would involve the implant being placed in combination with a blind sac closure and obliteration, for example, when there is a dry, clean mastoid cavity. The choice of technique may influence the passive hearing and should, therefore, be discussed with the patient.
43.3 Eradication of COM Prior to Implantation
43.3.1 Tympanic Membrane Perforation
A chronic tympanic membrane perforation needs to be repaired prior to implantation to establish a protected middle ear cleft. Cartilage grafting may help stabilize the tympanic membrane in the case of poor underlying Eustachian tube function. Alternatively, removal of the perforated tympanic membrane remnants together with the canal skin, annulus, and the lateral ossicles may be considered, with closure of the EAC in a blind sac as described below. Even if adding complexity to the procedure, this usually heals without complications and the result is independent of Eustachian tube function and healing properties of the tympanic membrane. Unless the perforation is small and the middle ear mucosa clearly healthy, it is recommended that a cortical mastoidectomy and a posterior tympanotomy are performed at the time as the myringoplasty. This is straightforward when a postaural approach is being used and may improve healing, especially if the middle ear is actively infected (▶ Fig. 43.2).
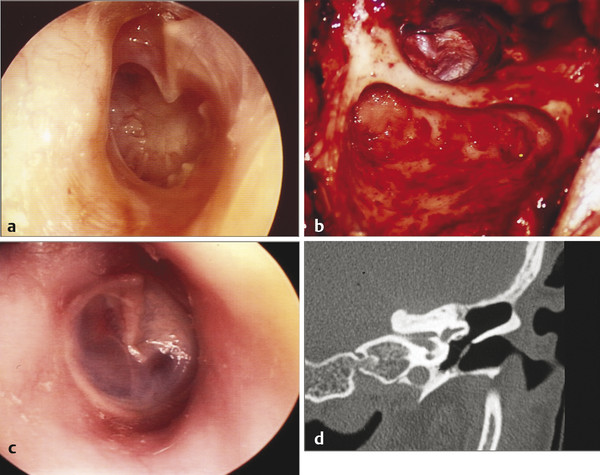
Fig. 43.2 (a) Subtotal tympanic membrane perforation. (b) On-lay myringoplasty and canal wall-up mastoidectomy. (c) Healed tympanic membrane. (d) Coronal view CT scan showing an aerated middle ear cleft.
Although performing a staged procedure should be the general rule in an infected ear, to ensure that the middle ear stays aerated with a healed tympanic membrane and eradication of infection or bacterial colonization, implantation at the same stage as repairing the perforation may be an option in the case of a long-standing, stable non-infected ear. There may be situations where two surgeries are contraindicated, such as in a patient where additional anesthesia should be avoided or where postponing the implantation of the hearing device creates a considerable handicap. If a single-stage procedure is considered, a blind sac closure rather than a tympanoplasty may be preferable as the more conservative option.
It is a common situation that a child has a ventilation tube in place through the tympanic membrane at the time of cochlear implantation. The tube can be removed at the time of implantation or alternatively left in situ to extrude spontaneously. Either approach seems to work well, with very little risk of developing a chronic perforation or infection of the implant. A well-aerated middle ear has a healthy mucosa, and there seems to be little risk of developing either a new episode of otitis media with effusion when the perforation heals or episodes of acute otitis media (AOM) with discharge through the ventilation tube where it is not removed at the time of implantation. Kennedy and Shelton3 reviewed the management of ventilation tubes in children receiving CIs and concluded that various approaches were safe.
43.3.2 Tympanic Membrane Retraction and Atelectasis
“Retraction disease” has a pathophysiology in which recurrent problems are common. Localized retractions in the attic or pars tensa regions may be managed by a limited tympanoplasty prior to implantation, aiming to stabilize the tympanic membrane. This is preferably done as a staged procedure but may be performed with implantation if very limited. For cases with larger retractions, removal of all epithelium and amputating the EAC with a blind sac closure may provide a safer alternative. An atelectatic tympanic membrane may be a substantial challenge. Avoidance of retraction onto the electrode cable or any part of the implant is essential. Cartilage grafting, particularly to the posterior-superior quadrant, may help provide long-term stability.
43.3.3 Cholesteatoma
The presence of cholesteatoma in an ear is clearly incompatible with long-term stability of a CI or an AMEI. It is essential that there is complete eradication of squamous epithelium from the middle ear cleft and reconstruction or obliteration such that recurrence will not occur. In some circumstances (e.g., small attic cholesteatoma) the surgeon may elect to use a canal wall-up technique, aiming to preserve a normal ear canal and aerated middle ear. Given the unpredictable nature of cholesteatoma and its tendency to recur, conversion to a radical cavity with blind sac closure of the meatus is very reasonable. Regardless of which procedure is used, the standard is to use a two-stage approach, with the implant being placed at the second stage after confirmation that there is no residual cholesteatoma. In cases with labyrinthine or cochlear fistulas with imminent cochlear fibrosis, the placement of a dummy electrode should be considered. The replacement of the dummy electrode by an active electrode is performed during the second-stage procedure.
43.3.4 Open Mastoid Cavity
In cases of a pre-existing radical cavity after surgical treatment for cholesteatoma or a fenestration procedure, eradication of all epithelial lining of the tympanomastoid cavity is essential (▶ Fig. 43.3). Any attempt to implant a CI or position a Vibrant Soundbridge in close proximity to skin is very likely to lead to complications of infections or extrusion of the array. Techniques that cover the array or actuator with cartilage or muscle have been described, resulting in a “cavity reduction”; however, complete epithelial removal is likely to be a safer long-term solution.
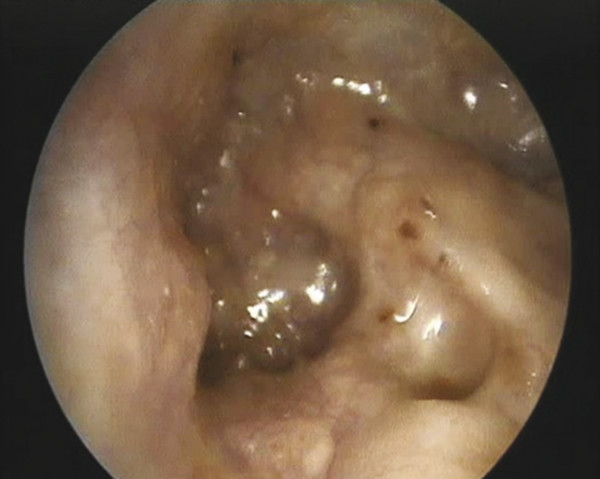
Fig. 43.3 A left ear radical cavity Type IV.
43.4 Surgical Technique
When the extent or recurrent nature of tubotympanic disease indicates that achievement of an aerated middle ear cleft is predicted to fail or has failed, or where there is an existing canal wall-down cavity or cholesteatoma with significant bony canal erosion, then the appropriate procedure is a radical mastoidectomy combined with removal of all tympanic membrane remnants and bony canal skin, with eversion and blind sac closure of the external auditory meatus. It should be noted that this is not a true subtotal petrosectomy, as it is often described. Care is taken not to remove all bone over the mastoid segment of the facial nerve (facial ridge) as a perineurium without a bony cover will adhere to the soft tissues used for obliteration and make a second-stage procedure potentially more complicated.
The blind sac closure is usually combined with obliteration of the cavity, either with a vascularized rotational flap or with an autologous fat graft. The Eustachian tube is plugged. To facilitate secure closure of the Eustachian tube, the tensor tympani muscle can be elevated from its semicanal and rotated into the Eustachian tube after mucosal elevation and the plugging further reinforced with bone paté and muscle or fascia. Bone wax or other foreign material is ideally avoided. For the mastoid cavity obliteration, if the middle ear cleft is actively infected, then it is preferable to use a vascularized soft tissue flap without non-vitalized filler materials; however, if the cavity has been dry and free of infection, then autologous (abdominal) fat may be used.
43.4.1 Blind Sac Closure
Using a postauricular approach, the technique is to divide the skin of the EAC circumferentially at the level of the cartilaginous-bone junction. A subperichondrial plane is developed over the tragal cartilage, and the skin is dissected in a lateral direction creating a “skin tube” that can be everted from the meatus. During the procedure, it is important to avoid tears in the skin as these may heal with epithelium trapped under the skin. The everted skin edges are transfixed with a vertical mattress suture. This maintains eversion of the amputated, closed canal and ensures secure healing, avoiding skin entrapment. Initially, the everted meatus will project slightly; however, with healing it will become cosmetically acceptable and be positioned slightly medial to the conchal bowl (▶ Fig. 43.4).
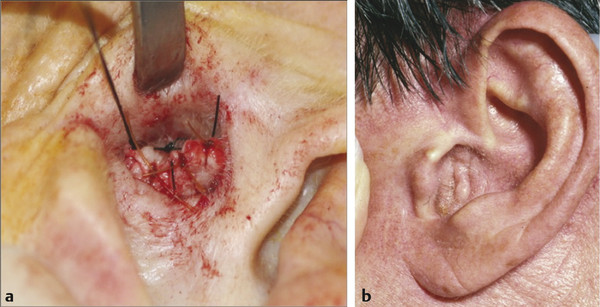
Fig. 43.4 Blind sac closure. (a) The external auditory canal skin is everted out of the meatus and sutured. (b) Once healed, the meatus is closed with a short, or, as in this case, an absent auditory canal.
To secure and stabilize the medial aspect of the everted meatus, a purse string–type resorbable suture can be placed. The medial aspect should be further reinforced with a periosteal and muscle flap, superiorly-anteriorly based or inferiorly-posteriorly based (or a combination of the two), which is rotated into the area under the amputated canal. Care should be taken to position and secure the flap anteriorly toward the temporomandibular joint; the flap may be sutured to the medial aspect of the tragus.
The final result is an absent or short, clean EAC, sometimes healed with a “skin bud” at the bottom. The mastoid cavity and middle ear cleft should be free of squamous epithelium and any pathology associated with it. The everted meatus may be dressed with antibiotic cream during healing.
43.4.2 Mastoid Obliteration
It should be noted that obliteration of a closed mastoid cavity is a different scenario than obliteration of an open mastoid cavity (as is described in Chapter 41); however, techniques involving rotational soft tissue flaps may apply to both situations. The aim of “closed mastoid obliteration” is to create a clean, non-aerated environment, thus reducing the risks of infection associated with introduction of a foreign material. This also achieves secure cover of the electrode cable or actuator, avoiding the risk of exposure and extrusion (▶ Fig. 43.5). Obliteration of the created surgical cavity in the mastoid and the tympanum with abdominal fat or muscle flaps is a traditional way of closing the defect in cases where a large aerated volume would be disadvantageous, such as when a large area of the dura is exposed or when there is risk of cerebrospinal fluid (CSF) leak. In this situation the aim of the obliteration is complete eradication of any aerated space, and this, of course, requires a simultaneous closure of the Eustachian tube.
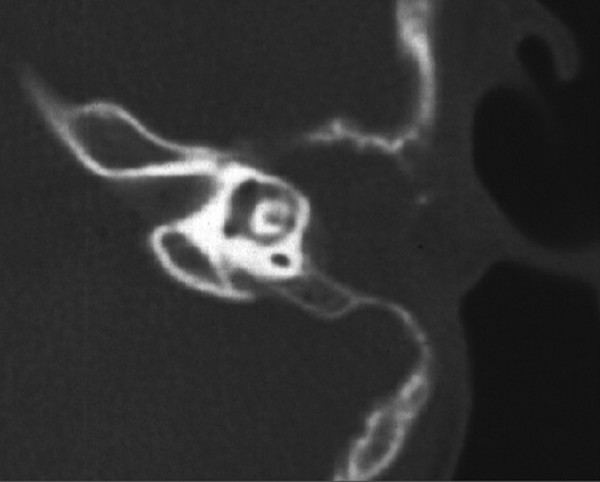
Fig. 43.5 CT scan of an obliterated left mastoid cavity prior to second-stage implantation.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


