Innate Immune System
The innate immune system is the first line of defense against foreign substances. It comprises both physical barriers and an array of receptors and antimicrobial compounds. The sinonasal barrier comprises ciliated epithelial cells, subepithelial glands, goblet cells, and the mucus blanket. This epithelium provides a barrier via tight junctions that prevent passage of pathogens across the mucosal surfaces and mucociliary transport to eliminate pathogens trapped in secreted mucus. Nasal glandular products such as lactoferrin, defensins, and lysozyme are the initial defense against infection and function irrespective of prior exposure to the pathogens. Impairment of the innate immune system provides one theory as to how inflammation leads to the development of chronic rhinosinusitis (CRS) (
26).
The cells associated with innate immunity include neutrophils, monocytes, mast cells, eosinophils, basophils, and dendritic cells (DCs). These cells are activated in the presence of microbes and work to rid the body of infection. They use pattern recognition receptors (PRRs) found on their surfaces, intracellular compartments and secreted in the bloodstream to opsonize bacteria, activate coagulation and complement cascades, induce phagocytosis and apoptosis, and implement proinflammatory signaling pathways (
27).
There are several classes of PRRs: Toll-like receptors (TLRs), RIG-I-like receptors, Nod-like receptors, and C-type lectin receptors (
27). PRRs recognize highly conserved DNA sequences that are necessary for the survival of many microorganisms. These highly conserved sequences, known as pathogen-associated molecular patterns (PAMPs), include peptidoglycan and lipoteichoic acid from gram-positive bacteria, lipopolysaccharide (LPS) from gram-negative bacteria, and RNA from viruses. The responses of the innate immune system against the PAMPs signal the adaptive immune system to develop memory and subsequent longer-lasting immune responses. TLRs, first described in 1994, are the most studied of the various PRR classes. They are transmembrane glycoproteins with an extracellular N-terminal leucine-rich domain and an intracellular C-terminal domain, which is itself known as Toll/interleukin (IL-1) receptor (TIR) because of its homology to IL-1 (
28).
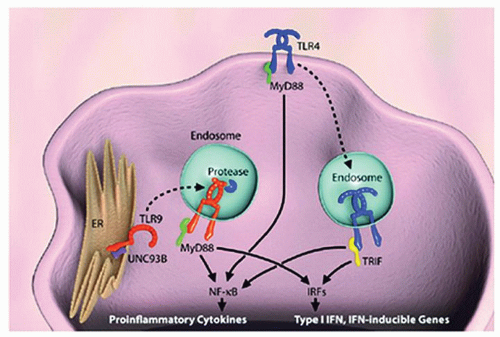
Recognition of the various PAMPs by TLRs on the surface of monocytes, macrophages, DCs, and mast cells initiates inflammatory responses, which induce cytokine signaling that can be Th1 or Th2. This signaling can be categorized as either myeloid differentiation primary response (MyD88)-dependent (
Fig. 25.1), which is used by all TLRs except for TLR3, or TIR containing adaptor-inducing interferon (IFN)-γ-dependent pathways (
29). TLRs respond to specific conserved sequences or to molecules present on invading microbes; for example, TLR4 responds to lipid A, a component of LPS, whereas TLR3 recognizes double-stranded RNA. Inflammatory cells express different classes of TLRs, depending on their lineage and maturity (
30). The spatial distribution of TLRs ultimately determines the microbes they encounter and whether these elements are recognized as being self or nonself (
31,
32). Some TLRs are expressed on the cell surface, whereas others are expressed intracellularly within endosomes (
Table 25.1).
Natural killer (NK) cells originate in the bone marrow and participate in the innate immune system. NK cells simultaneously express both T-cell markers (CD3, T-cell receptor [TCR]-αβ) and NK cell markers (CD56, CD16, CD95, and CD178) (
33). A large fraction of NK cells are referred to as invariant NK cells, and they are characterized by the expression of a single unique TCR-α rearrangement. Upon activation of NK cells, they are capable of rapid and substantial production of cytokines, including IL-4, which is an important cytokine in allergic pathogenesis (
34).
NK cells were first known for their capability to lyse tumors without any prior priming or immunization. They have been shown to have diverse functions ranging from secreting cytokines such as INF-γ, TNF-α, and granulocyte monocyte-colony stimulating factor (GM-CSF) and chemokines (lymphotactin, RANTES (Regulated upon Activation, Normal T-cell Expressed, and Secreted), and macrophage inflammatory protein [MIP]-1α/MIP-1β) after activation (
35) to serving as intermediaries between the innate and adaptive immune systems.
NK cell precursors express IL-2 receptor once they commit to the NK lineage. As they mature, further receptor diversity is procured, and the mature NK cell exhibits specific major histocompatibility complex (MHC) receptors, including 2B4, CD38, CD7, and killer-cell immunoglobulin (Ig)-like receptor. Upon completion of maturation, NK cells migrate to the peripheral blood and congregate in the spleen, lymphoid organs, lungs, and liver. There are two different types of NK cells—those that participate in killing and those that focus on cytokine production (
36).
Another feature of innate immunity is the complement system. The complement system is the primary humeral mediator of antigen-antibody reactions. Initially, complement was thought to play a major role only in innate immunity against invading pathogens. It was discovered subsequently that the complement system plays an important role in the adaptive immune system by being involved with both T and B cells (
37). It has also been shown to play a role in tumor growth (
38), tissue regeneration, and in diseases like hemolytic uremic syndrome and age-related macular degeneration (
39).
The complement consists of at least 30 chemically and immunologically distinct proteins, which can interact with each other, with antibodies, and with cell membranes. The proteins exist either as soluble in the blood or as membraneassociated. There are three routes by which the complement system is activated: the classical pathways, the lectin pathway, and the alternative pathway. All of the pathways converge at C3 (which is the most abundant complement protein found in the blood), resulting in the formation of activation products and the membrane attack complex (C5b-9) (
40).
The biologic activity of complement is manifested in three ways. First, certain complement proteins bind to or opsonize foreign particles. Specific cellular receptors for these complement proteins then mediate the binding and uptake of the opsonized particles by polymorphonuclear leukocytes and monocytes. Second, the small fragments of proteolytic cleavage from complement proteins diffuse readily, and they can bind to neutrophils and macrophages, causing chemotaxis and cell activation. Similar receptors on lymphocytes and antigen-presenting cells bind antigen-antibody complexes and enhance specific immune responses such as phagocytosis and clearance of apoptotic cells. Third, complement causes lysis by the
insertion of a hydrophobic “plug” into lipid-membrane bilayers, allowing osmotic disruption of the target cell. Deficiencies in complement lead to severe infectious or autoimmune diseases.
Complement may play a role, in the nasal mucosa, in an immediate-type hypersensitivity reaction by stimulating mast cells. Studies of asthma have implicated increases of anaphylatoxins C3a and C5a at sites of inflammation after segmental allergen provocation of the bronchus in asthma patients (
41), which can contribute to cellular recruitment of inflammatory cells.
Another important player in the innate immune response is the numerous antimicrobial peptides that are expressed in the sinonasal and lower airway epithelium (
26). These include lactoferrin (
42), lysozyme (
43), cathelicidin (
44), defensins (
45), SP-A and -D (surfactant protein) (
46,
47), acid mammalian chitinase (
47), collectins (
48), serum amyloid A (
49), and secretory leukocyte proteinase inhibitor (
50). The most abundant of the antimicrobial peptides in nasal secretions are the lysozyme, lactoferrin, and secretory leukocyte proteinase inhibitor (
51). In addition to being referred to as endogenous antibiotics by being secreted in response to microbes, these peptides also play a role in activating or suppressing effector cells of the innate and adaptive immune system. Human cathelicidin, LL-37, is chemotactic for neutrophils, monocytes, and T cells (
52). It has also been shown that SP-D can cause a shift from a Th1 to a Th2 cytokine response, inhibit lymphocyte proliferation (
53), and regulate DC responses (
54).
The defensins, composed of α-defensins and β-defensins, are expressed on cells such as neutrophils, monocytes, epithelial cells, and paneth cells (
55). Paneth cells, which are located at the base of intestinal glands, secrete α-defensins when exposed to bacteria and thus contribute to the gastrointestinal barrier. Degranulation of cells that express defensins occur in a dose-dependent manner as part of the host defense in response to infection at epithelial surfaces (
55). It is well known that phagocytes and leukocytes are recruited to sites of infection via chemotactic factors (
56). Defensins induce chemotaxis of the immature DCs to the sites of infections (
56).
The mRNA of HD5, an α-defensin, is expressed in variable amounts in nasal epithelial cells (
57). β-Defensins in mice induce maturation of DCs via TLR4 (
58). Because DCs are involved in both antigen (Ag)-specific immune responses and Th1/Th2 polarization, defensins may contribute to interactions between the innate and the adaptive immune system via their influence on DCs. Cathelicidin is an antimicrobial protein expressed by epithelial cells, monocytes, NK cells, mast cells, and γδT cells. LL-37, a cathelicidin peptide, participates in host antimicrobial immunity by recruiting phagocytes, immature DCs, and T cells to sites of inflammation (
52). Both LL-37 and human β-defensin-2 cause mast cell degranulation, with consequent histamine release (
59). Eosinophil-derived neurotoxin is implicated in antiviral roles and has been shown to play a role in DC development, and like defensin, it may also play a role in bridging of the innate system with the adaptive immune system (
60).
Other roles of the antimicrobial peptides have been postulated. Biofilms, which are implicated as a possible cause of CRS (
61,
62), are organized communities of microbial cells that are attached to living surfaces and are composed of exopolysaccharide matrix encased in a self-produced exopolysaccharide matrix and attached to an inert or living surface (
63).
In vitro, synthetic Cathelicidin LL-37 shows antibiofilm properties at subbactericidal concentrations when cultured with Pseudomonas aeruginosa (
64). In animal models, synthetic LL-37 successfully eradicated biofilm of Pseudomonas sinusitis (
65).
Adaptive Immune System
Innate immunity controls and eliminates infection with the help of the complement system, NK cell activation, and TLR activation. If the innate immune system is unable to mount an inflammatory response sufficient to remove pathogens, the adaptive immune response is activated. The adaptive immune system, in contrast to the innate immune system, is highly specific in nature and benefits from gene rearrangement. Developing B and T lymphocytes obtain randomly assembled Ig receptors and TCR from specific gene segments. The variable, diversity, and joining (VDJ) segments that are flanked by recombination signal sequences are recombined with either addition or subtraction of nucleotides, creating immense variability in the receptor (
66). Whereas the remodeling process that regulates chromatin accessibility of target gene segments remains unclear, it is evident that these processes tremendously expand the diversity of antigen recognition.
The adaptive immune system utilizes many of the same cells as the innate immune system. One major difference between the two responses is the mode of recognition of antigens. Whereas the innate immunity recognizes microorganisms through components present on their cell walls via broad receptor recognition, the adaptive immune system recognizes specific antigens that are processed and presented by antigen-presenting cells in the context of MHC receptors. In cell-mediated adaptive immunity, priming occurs during the first exposure to antigen by MHC II on antigen presenting cells (APCs) with the help of costimulatory molecules. TCR receptors on the T cell associate with the CD3 receptor and, in turn, activate a series of phosphorylation steps within the cell, resulting in the formation of either CD4+ or CD8+ T cells. Activation of T-helper (Th) cells allows the secretion of many cytokines and mediators which, in turn, activate effector cells such as macrophages. Also, in humeral adaptive immunity, the primed Th cell comes into contact with B cells expressing the same antigen. Interaction between these two cell types and linking of CD40 and CD40L on B and activated Th cells, respectively,
causes B-cell activation and differentiation into antibodysecreting plasma cells (
67).
A major difference between the adaptive and the innate immune system is the capability of memory in the adaptive system. Most activated T cells function as effector cells and later undergo apoptosis. Some of them, however, differentiate and persist in the host for many years to provide rapid protection on subsequent antigen exposure. The exact mechanism by which certain subsets of T cells become memory cells, and under what signaling conditions, is under investigation.