Chapter 5 Hyperthyroidism
Toxic Nodular Goiter and Graves’ Disease
Introduction
Goiter is enlargement of the thyroid gland, commonly encountered in clinical endocrine practice. It may be classified as diffuse or nodular and may be either nontoxic or toxic. Nodular goiter may include multiple nodules (i.e., multinodular goiter [MNG]) or a single nodule. Regardless of different possible mechanisms of development, diffuse thyroid enlargement eventually evolves into a nodular stage.1
Graves’ disease is the most common type of thyrotoxicosis encountered in the industrialized world. Graves’, disease owes its name to an Irish doctor, Robert James Graves’ who described the first case of goiter with exophthalmos in 1835.2 Patients with Graves’ disease usually have diffuse, nontender, symmetric enlargement of the thyroid gland. Other features of Graves’ disease affecting the eyes and skin will be described in ensuing sections in this chapter.
Epidemiology
Toxic Nodular Goiter
TNG is the most frequent cause of thyrotoxicosis in the elderly. It accounts for about 5% to 15% of patients with endogenous hyperthyroidism, but the proportion is higher in iodine-deficient geographic regions.2,3 Changes in the iodine content of salt and in the iodine supplementation of water have been linked to changes in the incidence of TNG. In Switzerland in 1980 and in Spain in 1994, the iodine content of salt was increased, and this was associated with a transient increased incidence of thyrotoxicosis followed by decreased incidence, mainly the result of reduced TNG incidence.4,5
Graves’ Disease
Being the most common cause of thyrotoxicosis in all age groups, Graves’ disease accounts for 70% to 80% of endogenous hyperthyroidism. The incidence of Graves’ disease is five times higher in females than in males, occurring generally during women’s reproductive years, although it may occur at any age.2
Pathogenesis
Toxic Nodular Goiter
The natural history of a nontoxic MNG involves variable growth of individual nodules; this may progress to hemorrhage and degeneration, followed by healing and fibrosis. Calcification may be found in areas of previous hemorrhage. Some nodules may develop autonomous function. Autonomous hyperactivity is conferred by somatic mutations of thyrotropin or thyroid-stimulating hormone receptor (TSHR) in 20% to 80% of toxic adenomas and some nodules of MNGs.6 Autonomously functioning nodules may become toxic in 10% of patients. Hyperthyroidism predominantly occurs when single autonomous nodules are larger than 2.5 cm in diameter. However, in geographic areas with iodine deficiency, smaller autonomous nodules may produce systemic, clinical manifestations of hyperthyroidism.7
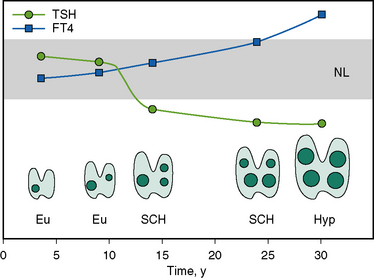
The development of hyperthyroidism in MNG takes many years. The process evolves from a small gland with one small nodule or more to nodules increasing progressively in number, size, and function. Initially, most patients are euthyroid, but with enlarging goiters, autonomy develops, illustrated by low or suppressed serum thyroid-stimulating hormone (TSH) with normal serum levels of thyroid hormones. As shown in Figure 5-1, a state of subclinical hyperthyroidism with low TSH but normal free T4 and free T3 levels may exist for years before progression to clinical hyperthyroidism.
Graves’ Disease
The histology of the thyroid gland in patients with Graves’ hyperthyroidism is characterized by follicular hyperplasia, a patchy (multifocal) lymphocytic infiltration, and rare lymphoid germinal centers. The majority of intrathyroidal lymphocytes are T cells, and germinal centers (B cells) are much less common than in chronic autoimmune thyroiditis (Hashimoto’s disease). Thyroid epithelial cell size correlates with the intensity of the lymphocytic infiltrate, suggesting thyroid cell stimulation by local B cells secreting TSHR-Ab.8 The presence of these antibodies is positively correlated with active disease and with relapse of the disease. There is an underlying genetic predisposition, because of an increased frequency of haplotypes human leukocyte antigen (HLA)-B8 and HLA-DRw3 in white patients, HLA-Bw36 in Japanese patients, and HLA-Bw46 in Chinese patients with the disease. However, it is not clear what triggers the acute episodes. Some factors that may incite the immune response are pregnancy, particularly the postpartum period; iodine excess, particularly in geographic areas of iodine deficiency; lithium therapy; viral or bacterial infections; and glucocorticoid withdrawal.
The etiology and pathogenesis of Graves’ ophthalmopathy are not known. It may involve cytotoxic lymphocytes and cytotoxic antibodies sensitized to a common antigen in orbital fibroblasts, orbital muscle, and thyroid tissue, which may cause inflammation, resulting in proptosis of the globes. It has been suggested recently that TSHR-bearing circulating fibroblasts and fibrocytes may be activated directly by the TSHR-Ab.9 The pathogenesis of dermopathy may also involve this mechanism. Patients with exophthalmos and particularly those with dermopathy almost always have high titers of circulating TSHR autoantibodies, suggesting that these two clinical manifestations represent the most severe form of this disease.
Diagnostic Evaluation
History and Physical Examination
The clinical presentation ranges from no symptoms and a suppressed sensitive serum TSH level to overt or obvious clinical hyperthyroidism. The latter includes symptoms associated with increased adrenergic tone and resting energy expenditure and other hormonal effects. Features related to increased adrenergic tone include nervousness, tremor, increased frequency of defecation, palpitations, diaphoresis, irritability, insomnia, headaches, lid retraction and lid lag, muscle weakness, tachycardia, hyperreflexia, and widened pulse pressure. Those related to increased energy expenditure include heat intolerance, unintentional weight loss without anorexia, and warm, moist skin. In elderly subjects, the presentation may be subtle, with atrial fibrillation, weight loss, weakness, and depression.10
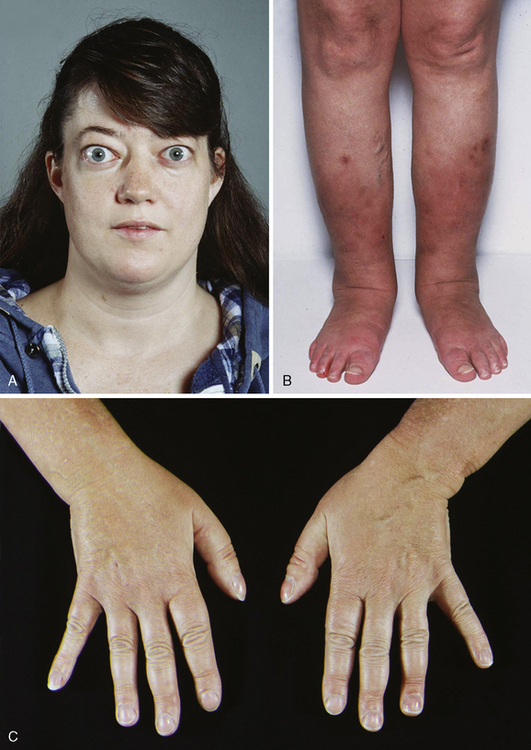
Other features of Graves’ disease include onycholysis, acropachy, and pretibial myxedema (Figure 5-2). Pretibial myxedema is a rare, reddish lumpy thickening of the skin of the shins. This skin condition is usually painless and is not serious. Like the eye disorders of Graves’ disease, the skin manifestation does not necessarily begin precisely when hyperthyroidism starts. Its severity is not related to the level of thyroid hormones. It is not known why this problem is usually limited to the lower leg or why so few people have it. Occasionally symptoms related to the mass effect of a large goiter may occur. Very large goiter may extend retrosternally or substernally, resulting in symptoms and signs of tracheoesophageal pressure. These may include dysphagia, cough, and choking sensation or stridor, particularly if severe tracheal narrowing exists. The development of facial plethora, cyanosis, and distention of neck veins with raising both arms simultaneously may result from deep goiter compression of the structures located within the bony confines of the thoracic inlet (Pemberton sign).
Common and important clinical differences between Graves’ disease and TNG are summarized in Table 5-1.
Table 5-1 Clinical Differences between Graves’ Disease and Toxic Nodular Goiter
| Characteristic | Graves’ Disease | Toxic Nodular Goiter |
|---|---|---|
| Goiter | Diffuse | Multinodular |
| —Size | Small | Large |
| —Growth | Rapid | Slow |
| Patient age, y | < 45 | > 50 |
| Hyperthyroid onset | Rapid | Slow |
| Histologic features | Follicles similar, intense iodine metabolism | Variable follicular size, shape, and intensity of iodine metabolism |
Adapted from Hurley DL, Gharib H: Thyroid nodular disease: is it toxic or nontoxic, malignant or benign? Geriatrics 50:24-26, 29-31, 1995.
Laboratory Evaluation
Diagnosis is always established by the measurement of sensitive TSH and thyroid hormone levels (free T3 and free T4). Thyrotoxicosis caused by TNG or Graves’ disease is usually characterized by a suppressed TSH level with either normal (subclinical) or elevated (overt) free thyroid hormone levels. It is insufficient to rely on the measurement of TSH or free thyroid hormones alone to diagnose TNG or Graves’ disease, because suppression of TSH or elevation of thyroid hormones can be associated with clinical conditions other than TNG and Graves’ disease (Table 5-2). Other serologic findings, such as antithyroid antibodies (antithyroid peroxidase and antithyroglobulin), that support the diagnosis of autoimmune thyroid disease may be detected in patients with Graves’ disease. However, serum TSHR-Ab is occasionally helpful in the diagnosis of Graves’ disease, though there is no consensus regarding its routine measurement in Graves’ disease.
Table 5-2 Clinical Conditions Associated with Abnormal TSH and High Free Thyroid Hormone without Toxic Nodular Goiter or Graves’ Disease
| Low TSH | High TSH |
|---|---|
| Secondary hypothyroidism | TSH-secreting pituitary tumor |
| Nonthyroidal illness | Thyroid hormone resistance |
| Glucocorticoid therapy | |
| Amiodarone use | |
| Excessive thyroid hormone therapy |
TSH, thyroid-stimulating hormone.
Imaging
Radionuclide Imaging
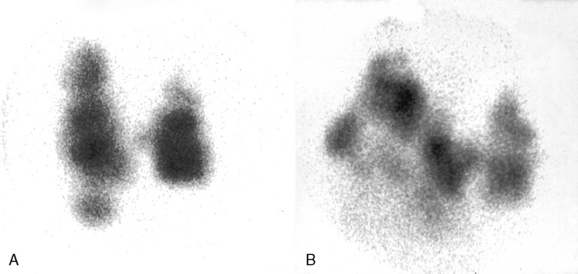
Radionuclide scanning and radioactive iodine uptake (RAIU) are useful tests to elucidate the cause of hyperthyroidism. In TNG, the radioactive iodine (RAI) concentration is in the nodule(s), and uptake is inhibited in the surrounding tissue, giving the appearance of “patchy uptake” (Figure 5-3). Consequently, the total RAIU may be either slightly raised or at the upper limit of normal. In Graves’ disease, because of diffuse thyroid involvement, the RAIU is always intense and increased (Figure 5-4). Thyroid radionuclide imaging may not be necessary in every case when the diagnosis is obvious, but it is helpful in the differentiation of other clinical conditions associated with hyperthyroidism but with low RAIU (Table 5-3).
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree