CHAPTER 15 Head and Neck Manifestations in the Immunocompromised Host
Spectrum of Immunosuppression
Immunosuppression occurs in the setting of genetic, infectious, and other acquired disorders. The cells of the immune system derive from hematopoietic stem cells in the bone marrow, circulate in the blood and lymph, and are present in nearly every tissue.1 Protection of the host occurs by two mechanisms: innate and adaptive. The innate immune system is widely conserved among many vertebrate species and is the first line of defense. The primary cells involved are neutrophils, eosinophils, basophils, macrophages/monocytes, dendritic cells, and natural killer cells. Adaptive immunity protects the host against pathogens that escape innate immune responses and is a characteristic of higher vertebrates. The cellular components involved in adaptive immunity are T cells and B lymphocytes. Immunodeficiencies can affect any of the components of the innate and adaptive immune systems.
Congenital immunodeficiencies, which are less common than acquired, rarely affect the innate immune system. Severe combined immunodeficiency (SCID) affects both the T and B cells of the adaptive immune system, and children born with this disorder often die of infections in early childhood. Other genetic immunodeficiencies that affect T and B cells include DiGeorge’s syndrome, X-linked agammaglobulinemia, Wiskott-Aldrich syndrome, common variable immunodeficiency, and selective immunoglobulin deficiencies in which serum concentrations of one or more immunoglobulin subclasses are reduced. Clinical characteristics of T-cell dysfunction include onset of symptoms in early infancy (4 to 5 months) with recurrent fungal, viral, and mycobacterial organisms and infections with opportunistic infections such as Pneumocystis.2 B-cell deficiencies are marked by recurrent infections with encapsulated organisms such as sinopulmonary infections, otitis media and sepsis, and increased incidence of atopy, but no increased susceptibility to fungal or viral infections.2
Acquired immunodeficiencies are much more common than genetic immunodeficiencies. Acquired immunodeficiency may result from infection with the human immunodeficiency virus (HIV); hematologic malignancies and myeloproliferative disorders such as multiple myeloma or leukemia; diabetes mellitus; or iatrogenic drug-induced immunosuppression from chemotherapeutic agents, corticosteroids, and other immunosuppressive agents after solid organ and bone marrow transplantation (Table 15-1). Patients with immunosuppression are at higher risk than their immunocompetent counterparts for developing fungal, bacterial, and viral infections, and have higher rates of certain malignancies. Much of this pathology may manifest in the head and neck and thus otolaryngologists should be familiar with the spectrum of disease affecting immunocompromised patients.
Table 15-1 Causes of Immunosuppression
| Genetic |
| Acquired |
Human Immunodeficiency Virus/Acquired Immunodeficiency Syndrome
Biology and Immunology
HIV infects and debilitates lymphocytes and macrophages leading to progressive immune compromise. Progressive decline in immune function in later stages of infection culminates in the acquired immunodeficiency syndrome (AIDS). Anecdotal reports3 of unusual infections and idiopathic immune deficiency, primarily in homosexual men, began in the early 1980s and rapidly grew into the HIV/AIDS epidemic that has affected the world population. Every day, more than 6800 people become infected with HIV and 5700 people die of AIDS.4 Worldwide, approximately 33.2 million people are living with HIV infection, with sub-Saharan Africa being the most severely affected region, where AIDS is the leading cause of death. After many years of increase, the global prevalence of HIV has stabilized and in some countries has even decreased.4 With wider availability of effective treatment, rates of death have also recently begun to decline.
Transmission of HIV occurs through body fluids and tissues. Virus from an infected patient may inoculate the bloodstream of another individual through a breach in skin or mucosa, or via intravenous infusion. Modes of transmission include sexual intercourse, the sharing of needles by intravenous drug users, vertical transmission from mother to child, and, rarely, transfusion of contaminated blood products or accidental exposure in health care workers. More than 50% of new HIV infections in the United States occur in men who have sex with men; infection after heterosexual intercourse and injection drug use lead to 32% and 18% of new infections, respectively.4
HIV is a retrovirus in the Lentivirus subfamily, named for the slow progression of disease in affected individuals. These viruses establish chronic infections with a long incubation time and slow progression of disease. Viruses in the Lentivirus family typically infect cells involved in immune modulation; in the case of HIV, that is primarily CD4 T cells and macrophages, resulting in defects in both humoral and cell-mediated immunity. The virus life cycle begins when the virus binds to the CD4 receptor, a surface protein on the helper T subset of T lymphocytes, which is also expressed on macrophages. Fusion of the viral and cell membranes allows entry of the viral core into the cell. The reverse transcriptase enzyme, a protein carried by the virus, allows transcription of RNA into DNA (a reversal of normal transcription), then mediates transcription of the viral RNA genome into viral DNA. Viral integrase, another viral protein, then facilitates incorporation of the viral DNA into the host genome. The viral DNA is transcribed into multiple RNA copies by the host cell. This newly created RNA may be spliced and translated into viral proteins or it may remain intact as a future viral genome. Translation of some viral RNA sequences results in protein precursors or multiple proteins bound together. These precursors undergo proteolytic processing by a viral protease that liberates the functional viral proteins. These proteases are required for viral infectivity. Following replication of the viral genome and proteins, the new viruses bud from the infected cell and proceed to infect new cells. The viral DNA polymerase is error-prone, incorporating one mismatched nucleotide per genome per round of transcription. This, combined with the amount of replication that occurs, establishes a vast pool of genetic diversity.5,6 The development of vast genetic diversity also gives the virus an advantage in the development of drug resistance and provides a critical barrier to vaccine development.
Diagnosis and Classification
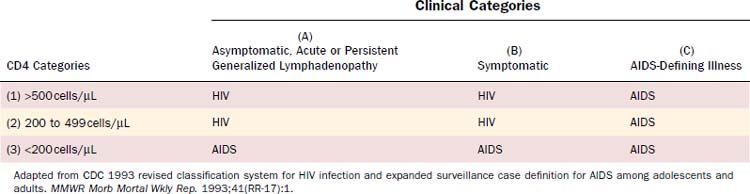
HIV infection is diagnosed when anti-HIV antibodies are detected in the serum by screening enzyme-linked immunosorbent assay (ELISA) and is confirmed with specific antibody detection with Western blot. Persistent antibodies against HIV typically occur within 3 months of infection. The CDC classification system for HIV, last updated in 1993, is based on the clinical manifestations of disease and the patient’s CD4 count (Table 15-2).7 These classifications assist in assessing the level of immune compromise and the corresponding risk for development of opportunistic infection or neoplasm. Three clinical categories have been identified: (A) asymptomatic HIV infection, persistent generalized lymphadenopathy, or acute HIV infection (a mononucleosis-type syndrome); (B) symptomatic conditions such as oral thrush, oral hairy leukoplakia, and fungal sinusitis that are attributed to HIV infection but do not fall into category A or C; (C) AIDS-defining conditions (Table 15-3). Patients may be further stratified according to their CD4 counts: (1) >500 cells/µL; (2) 200 to 499 cells/µL; (3) <200 cells/µL. The lowest accurate CD4 count, not the most recent, is used for classification purposes. AIDS is diagnosed for any patient who develops an AIDS-defining illness (category C) or CD4 less than 200 cells/µL (category 3). The World Health Organization (WHO) classification for HIV infection includes four clinical stages8:
Table 15-3 AIDS-Defining Illnesses
Mycobacterium avium complex, Mycobacterium kansasii, or other species, disseminated or extrapulmonary |
Adapted from CDC 1993 revised classification system for HIV infection and expanded surveillance case definition for AIDS among adolescents and adults. MMWR Morb Mortal Wkly Rep. 1993;41(RR-17):1.
Highly Active Antiretroviral Therapy
When left untreated, HIV infection causes gradual debilitation of the immune system over a period of years, resulting in profound immunocompromise and AIDS. Advances in understanding of HIV biology have allowed for the development of medications targeting the reverse transcriptase and protease enzymes critical to the viral life cycle. Current drugs do not eradicate the virus, thus the major goals of treatment are the complete inhibition of HIV replication, reduction in HIV-related morbidity, and prevention of vertical transmission.9–11 Antiretroviral agents from five classes of drugs are currently available to treat HIV infection. These include nucleoside/nucleotide reverse transcriptase inhibitors (NRTIs), non-nucleoside reverse transcriptase inhibitors (NNRTIs), protease inhibitors (PIs), entry inhibitors (EIs), and integrase inhibitors.11 Combinations of three or more of these agents allow for such effective suppression of viral replication that the emergence of drug-resistant variants is delayed, forming the basis of highly active antiretroviral therapy (HAART).10 The use of HAART is credited for a 45% decline in mortality rates during the late 1990s, although in more recent years, the decline in mortality has been more modest, only 8% between 2000 and 2004.12
The U.S. Department of Health and Human Services recommends initiation of HAART for patients with history of an AIDS-defining illness or with CD4 less than 350 cells/µL.11 The timing of HAART initiation in asymptomatic patients with CD4 greater than 350 cells/µL is not well defined and the decision should be guided by the potential benefits and risks associated with therapy, patient comorbidities, and willingness to adhere to long-term treatment. Drug resistance testing is recommended for all patients with diagnosed HIV, regardless of whether treatment will be initiated. Two types of combination regimens are generally used: NNRTI-based (one NNRTI with two NRTI) and PI-based (one or two PI with two NRTI). The choice of regimen is determined by patient comorbidities (e.g., tuberculosis infection, liver disease, and cardiovascular disease), dosing convenience, potential drug interactions, and results of genotypic drug resistance testing.
Immune Restoration Disease
Recently, infections with opportunistic organisms occurring in HIV-positive patients who initiate antiretroviral therapy have been recognized and are known as immune reconstitution syndrome or immune restoration disease (IRD).13,14 Diagnostic criteria for IRD include a previous diagnosis of AIDS, concurrent antiretroviral therapy with increasing CD4 cell count, and an exacerbation or atypical presentation of opportunistic infections.15 The most common pathogens associated with this condition are Mycobacterium (tuberculous and nontuberculous), Cryptococcus, herpesviruses, and hepatitis B and C viruses.13 Symptoms may occur as soon as a few days following HAART initiation, although most patients develop symptoms 2 to 8 weeks after beginning HAART.15 IRD may occur in 10% to 50% of patients initiating HAART, but opportunistic infections are rare, occurring in 5% of patients.16 It is also seen in 5% of transplant recipients after dose reduction in immunosuppressive medications.17 IRD is thought to be caused by overly exuberant pathogen-specific immune responses as patients recover immune function.13–1517 IRD-associated mycobacterial infections generally present with fever, lymphadenopathy, and worsening respiratory function.18 HIV-positive patients not already receiving HAART who present with mycobacterial infections should not be started on HAART until after the infection is controlled.15 Trials examining the role of steroidal and nonsteroidal anti-inflammatory agents in the treatment of IRD are ongoing.18
Occupational Exposure to Human Immunodeficiency Virus Infection
With more than 1.3 million HIV-positive children and adults in the United States4 and with the high prevalence of otolaryngologic complaints among these patients, most otolaryngologists will treat HIV-positive patients. An understanding of the risks of occupational transmission as well as precautions that may minimize such transmission is essential. There have been 57 documented cases of HIV seroconversion among U.S. health care personnel following occupational exposure, the most recent occurring in 2000.19 Because of the voluntary nature of the reporting system, there may be underreporting of cases. Exposures that carry a risk for HIV transmission are percutaneous injuries (e.g., needlestick or cut with a sharp object) or contact of mucous membrane or nonintact skin with blood, tissue, or body fluids that are potentially infectious. The risk of transmission from fluid other than blood or transmission through nonintact skin is too low to be estimated in prospective studies. Even though cerebrospinal fluid carries a risk of transmission, nasal secretions, saliva, sputum, sweat, tears, urine, and vomitus are not considered infectious unless they are visibly bloody.20 Pooled prospective data suggest an average risk of HIV transmission of 0.3% for needlesticks and 0.09% following mucous membrane exposure.21 Whereas injury from suture needles has been suggested as a possible source of occupational exposure to HIV, such an injury has not been implicated as a source of transmission in prospective studies. Although a lower viral load (<1500 RNA copies/mL) or one that is below the limits of detection probably indicates a lower titer exposure, it does not rule out the possibility of transmission.
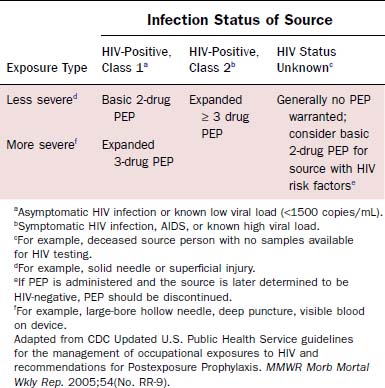
Prevention of blood exposure through the use of safe practices, barrier precautions, safe needle devices, and other innovations is the best way to prevent infection with HIV and other blood-borne pathogens.22 Simple strategies such as using double gloves, eye protection, impermeable gowns, and special techniques for passing sharp objects have all been suggested to minimize exposure risk. Despite such precautions, needlesticks and mucosal exposures will continue. After exposure to blood or other infectious fluids, the following actions should be taken immediately22: Wash needlestick areas and cuts with soap and water; flush nose, mouth, or skin with water; irrigate eyes with clean water, saline, or sterile irrigants. To minimize the risk of disease transmission, postexposure prophylaxis (PEP) should be initiated within hours of exposure and should continue for 4 weeks.20 The number of agents used depends on the type of exposure and risk profile of the source patient (Table 15-4). Treatment may be discontinued if the source patient subsequently tests negative for HIV infection and does not demonstrate any evidence of acute HIV infection. Exposed individuals should be tested for HIV at the time of exposure with follow-up testing at 6 weeks, 12 weeks, and 6 months after exposure.20 Prevention of HIV transmission following PEP is not complete. There have been at least 20 cases of seroconversion despite the use of PEP,21 with some of the patients having received multiple drug regimens. Thus careful adherence to universal precautions with avoidance of exposure remains the most prudent way to prevent infection.
Immunosuppression and Malignancy
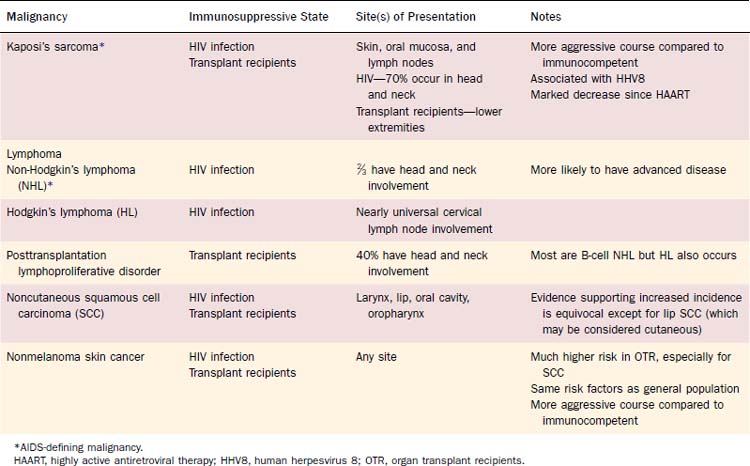
Patients with immune deficiency are at increased risk for developing malignancy (Table 15-5). The mechanism of oncogenesis is unknown, but the striking similarity between increased malignancies in immunosuppressed transplant recipients and patients with HIV suggests that immune function plays a vital role. In HIV, three types of cancer occur at markedly increased rates during later stages of the disease and are thus considered AIDS-defining malignancies. These include KS, non-Hodgkin’s lymphoma (NHL), and invasive cervical cancer. A meta-analysis comparing the incidence of cancer in patients with HIV/AIDS and immunosuppressed transplant recipients demonstrated a striking similarity in the pattern of increased rates of most types of cancer. Both groups had increased rates of AIDS-defining malignancies, Hodgkin’s lymphoma, squamous cell carcinoma of the lip, and nonmelanoma skin cancer.23 Non–AIDS-defining malignancies in patients with HIV also occur later in the disease course, often after the diagnosis of AIDS.24,25 These findings suggest that immune deficiency plays a role in the pathogenesis of cancer, although the mechanism of oncogenesis is unknown. There is no clear evidence linking immune depression with other types of head and neck cancer.
Kaposi’s Sarcoma
Epidemiology and Pathogenesis
KS is an angioproliferative disorder that causes lesions marked by spindle cell proliferation, neoangiogenesis, inflammation, and edema.26,27 There are four clinical variants with identical histologic features but with distinct epidemiologic patterns: classic KS, endemic KS, immunosuppression—or transplant-related KS, and HIV-associated KS (AIDS-KS).28–30 The classic form of KS was first described in older men of Eastern European or Mediterranean descent, with lesions typically occurring on the upper and lower extremities.29 An endemic variety of KS was also recognized in black adults and children in Africa, where it was and continues to be very common.31 In 1981, Friedman-Kien published a report of KS occurring in otherwise healthy young homosexual men32 and eventually the association between HIV infection and the development of KS became well established. By 1989, HIV-KS was reported in 15% of all US patients with AIDS.33 The overall risk of KS in AIDS patients was estimated to be 20,000 times that of the general population and more than 300 times that of other immunosuppressed patients. Since the introduction of effective antiretroviral therapy, there has been a marked decrease in the incidence of AIDS-KS,34,35 with estimated incidence now being approximately 5%.36 As solid organ and hematopoietic transplantation became more common, transplant recipients were also found to be at increased risk for developing KS.37–39 Prevalence reports vary depending on geographic location, ranging from 0.5% in the United States to 5.3% in Saudi Arabia.40 The risk for KS in organ transplant recipients is highest within the first 2 years after transplant and is increased with HLA mismatches.41
During the early AIDS-KS epidemic, a striking relationship was seen between the mode of HIV infection and the risk of developing KS. The risk for acquiring AIDS-KS was reported to be as low as 1% for patients who acquired HIV via blood transfusion, whereas in the population of men who have sex with men (MSM), the risk was 21%.33 KS was seen in only 2% of AIDS cases in women, with most of them reporting sexual contact with MSM.31 These epidemiologic disparities led to speculation that an infectious component, possibly sexually transmitted, played a role in AIDS-KS.33 In 1994, Chang and colleagues identified Kaposi’s sarcoma–associated herpesvirus (KSHV), a new herpesvirus, within KS lesions.42 KSHV is formally designated as human herpesvirus 8 (HHV8) and, along with Epstein-Barr virus (EBV), is a member of the gammaherpesvirus subfamily of herpesviruses.43,44 HHV8 has been identified in more than 95% of all KS lesions regardless of the epidemiologic subtype.29,30 The varying prevalence of HHV8 infection worldwide may explain the wide range of KS prevalence in different countries.45 The mechanism of oncogenesis has not been fully elucidated, in that KSHV is present in both immunosuppressed and immunocompetent individuals.46 It is believed that HHV8 infection coupled with the host’s state of immunosuppression plays a causal role.29,47,48
HHV8 infection itself appears to be a worldwide epidemic whose onset may have predated the HIV epidemic.47,49–51 Despite its widespread prevalence, the modes of transmission of HHV8 are incompletely understood. Some studies suggest that sex between men is an important route of transmission.49,52,53 In populations with high rates of endemic KS, familial clustering of HHV8 seropositivity was seen.54 Data suggesting vertical transmission from mother to child have also been reported,55,56 but do not explain the increased prevalence of HHV8 infection in prepubescent children. Some researchers have concluded there may be a role for child-to-child nonsexual transmission, perhaps via saliva.56–58
Presentation and Diagnosis
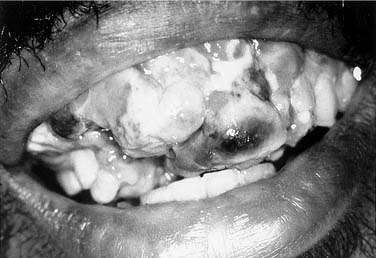
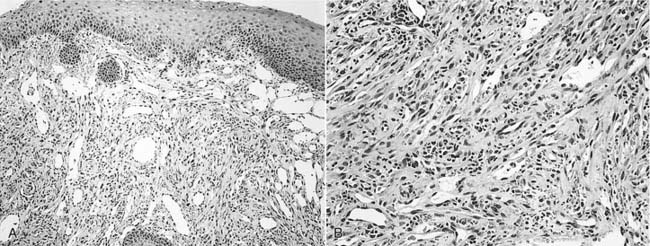
Compared with the classic and endemic forms of KS, immunosuppression-related KS and AIDS-KS are more aggressive. The clinical course of AIDS-KS ranges from an indolent, slowly progressive disease to a rapidly progressive and fatal course.28 AIDS-KS is associated with a shortened life expectancy, although most patients die of opportunistic infection or lymphoma and not KS per se.59 Multiple lesions develop in many patients, who more commonly have lymph node or visceral organ involvement. Frequently involved sites include the skin, oral mucosa, and lymph nodes. AIDS-KS occurs in the head and neck in as many as 70% of cases,60,61 whereas immunosuppression-related KS tends to occur in the lower extremities.30 Oral KS occurs in roughly one third of AIDS-KS,62 whereas in immunosuppression-related KS cutaneous lesions are most common.63 Cutaneous disease presents as multicentric macular and papular lesions that are nontender and nonblanching. These frequently coalesce and progress to violaceous, nodular lesions (Fig. 15-1). They are usually asymptomatic but may become pruritic and aesthetically displeasing. Mucosal KS commonly occurs in the oral cavity, and oral KS, which is associated with lower CD4 counts than cutaneous disease,66 may be a first sign of HIV infection.64,65 Oral KS may resemble cyclosporine-associated gingival hyperplasia in organ transplant recipients but cyclosporine usually causes generalized fibrotic gingival hyperplasia whereas oral KS produces a more localized red-purple enlargement.67 The most frequently affected sites in oral KS are the hard palate, gingiva, and tongue.64 Mucosal KS is more likely to be symptomatic than cutaneous disease. Lesions may cause loose teeth and are associated with pain, ulceration, and bleeding.
Depending on its location, visceral KS may be asymptomatic or rapidly fatal. Postmortem studies suggest that more than 25% of AIDS-KS patients have visceral lesions.30 These most commonly involve the gastrointestinal (GI) tract, liver, spleen, and lungs. GI disease is often asymptomatic, whereas median survival in pulmonary KS without treatment is only a few months.68 The most common presenting symptoms of pulmonary KS are dyspnea and cough, usually without fever unless concomitant infection is present. It may be difficult to distinguish pulmonary KS from other neoplastic and infectious diseases clinically, thus radiologic studies play an important role in its diagnosis. Computed tomography (CT) of the chest is often sufficient to diagnose pulmonary KS69 and may identify lymphatic or extrapulmonary involvement.68
Otolaryngologists may encounter patients with KS of the larynx associated with symptoms ranging from chronic cough or hoarseness to acute upper airway obstruction.70–73 If diagnosis and treatment proceed appropriately, acute airway obstruction may be avoided. Local treatment in isolated disease or systemic treatment in multicentric disease is recommended to avoid progression to airway compromise.71
Once the diagnosis of KS is suspected, biopsy may be performed for pathologic confirmation. The histopathology of KS is characterized by abnormal proliferation of lymphatics. Lesions may occur in three forms: patch, plaque, and nodule.74 Nodular lesions are more common in immunosuppression-related and AIDS-KS, and are marked by proliferation of slitlike vascular channels, extravasated erythrocytes, and spindle-cell proliferation (Fig. 15-2). KS lesions may be mimicked by bacillary angiomatosis, which also causes vascular proliferative lesions. The presence of pleomorphic bacilli on Warthin-Starry silver stain helps distinguish bacillary angiomatosis.75,76 The identification of HHV8 DNA may help distinguish KS from other vascular lesions.77 If KS is identified in a patient without a known history of HIV infection, an HIV test is warranted.
Treatment
Despite multiple therapeutic options, treatment is palliative and there is no cure. The disease course is variable; many patients achieve remission of KS but succumb to other causes of death. The course is complicated by increased susceptibility for developing opportunistic infections related to immunosuppression. Treatment of KS is determined by the extent and location of disease, as well as the severity of symptoms. Specific indications for treatment of KS include cosmetically disfiguring lesions, symptomatic oral lesions, symptomatic visceral lesions, or pain or edema associated with lymphadenopathy or extensive cutaneous disease. Local therapies may be useful in localized lesions or for cosmesis, but they do not prevent new lesions from developing in untreated areas78 and recurrence rates are high.30 Local treatments include alitretinoin topical gel (the only FDA-approved topical treatment for KS), local radiation, intralesional chemotherapy injection, cryotherapy, laser therapy, and surgical excision.
Restoration of immune function when possible should be a primary goal, because lesions often regress with the reversal of immunosuppression. Withdrawal of immunosuppressive agents in transplant recipients may, however, lead to transplant failure in up to half of patients.29 Some data suggest that using sirolimus instead of cyclosporine-based immunosuppression regimens may result in regression of immunosuppression-related KS lesions, but further investigation is needed to substantiate these results.63,79 HAART is now recognized to be a cornerstone of AIDS-KS treatment of all stages.34,78,80 Antiretrovirals have been shown to decrease KS tumor growth, and active investigation in the use of protease inhibitors for non-AIDS patients with KS is ongoing.81,82 The AIDS Clinical Trial Group (ACTG) staging system for KS originally took into account tumor burden, immune status as reflected by CD4 count, and the presence of systemic illness to predict likelihood of survival.83 More recent data have demonstrated that only high tumor burden and systemic illness portend an unfavorable prognosis.84
Four agents are currently FDA-approved for systemic treatment of KS. These include liposomal anthracyclines (doxorubicin and daunorubicin), paclitaxel, and interferon-alpha. Other commonly used agents include vinca alkaloids (vincristine and vinblastine) and bleomycin.34,85 Caution must be exercised when using paclitaxel because serious drug interactions with various components of HAART may occur. Multidrug chemotherapy is associated with increased toxicity, including myelosuppression, and results have been disappointing in widespread disease.85 HIV-positive patients on chemotherapy are also at increased risk of developing opportunistic infections.86 As the molecular basis of KS pathogenesis becomes more clearly defined, therapies targeting specific pathways are being developed. These experimental treatments have mostly focused on angiogenesis, HHV8 replication and life cycle, and cytokine regulation.87 There are ongoing clinical trials investigating antiangiogenic compounds such as imatinib mesylate,88 matrix metalloproteinase inhibitors,89 and interleukin-12.90
Cutaneous Neoplasms
Immunosuppressed organ transplant recipients and HIV-positive patients have a higher risk of developing skin cancer compared to immunocompetent individuals. Risk factors for the development of skin cancer in immunosuppressed patients are the same as those in the general population, with fair skin, sun exposure, and family history of skin cancer being the principal factors.91–94 Even though the rate of nonmelanoma skin cancer (NMSC) is increased in both groups, organ transplant recipients have a markedly higher risk than people with HIV/AIDS.23 The incidence of NMSC in OTR can be as high as 34%.95,96 In the general population, basal cell carcinoma (BCC) is more common than cutaneous squamous cell carcinoma (SCC), whereas organ transplant recipients develop SCC twice as frequently as BCC.97 The risk of developing NMSC increases dramatically with longer posttransplant times.97,98 At 10 years after transplantation, the prevalence of NMSC is 32%.99 Within the HIV-positive population, BCC is second behind KS as the most common cutaneous malignancy, with a prevalence of 1.8%.93 CD4 count does not correlate with the incidence or severity of NMSC.100,101 Both groups have increased rates of lip SCC.23,24 The evidence linking immunosuppression to increased risk of melanoma is less clear than for NMSC, in that increased rates of detection may reflect greater surveillance in immunosuppressed patients.92,101
Compared to NMSC in the general population, NMSC in these patients, particularly SCC, is more often invasive and aggressive, with higher rates of metastasis and recurrence.92,100–102 Melanoma also has more aggressive features in the setting of immune depression, and shorter disease-free intervals and overall survival have been demonstrated in HIV-positive patients.103
Treatment outcomes vary with the type of skin cancer. Although more aggressive types of BCC in HIV-positive patients have been reported,104–106 standard excisional techniques result in cure rates similar to those in the general population.100,101 In contrast, cutaneous SCC is associated with higher mortality in immunosuppressed patients, with mortality around 3% in organ transplant recipients107 and up to 50% in HIV patients who have metastatic SCC.108 As in the management of Kaposi’s sarcoma, reduction of immunosuppression in organ transplant recipients is associated with reduced incidence and improved outcomes for skin cancer.102 There are no clear guidelines for the management of cutaneous SCC or melanoma in HIV patients, but local excision with early consideration for systemic therapy is generally recommended.101 Because of the aggressive nature of skin cancers in immunosuppressed patients, early detection with a low threshold for biopsy of suspicious lesions is recommended.
Squamous Cell Carcinoma
Data regarding the incidence of noncutaneous SCC in immunosuppressed patients are controversial. Several studies have reported increased incidence of noncutaneous SCC in organ transplant recipients, with one large series reporting a 0.5% incidence, which was a 20-fold increase over the control population.109,110 In particular, patients undergoing liver transplantation had an even greater risk.111,112 There have also been reports of increased head and neck SCC in HIV-positive patients.24,113 Confounding factors of pretransplantation tobacco and alcohol use, however, may contribute to the increased incidence seen in both groups.109,112–114 High levels of immunosuppression in organ transplant recipients109 and lower CD4 count in HIV-positive patients115 may confer an increased risk. Immunosuppressed patients, however, do appear to experience a more virulent clinical course than the general population. Organ transplant recipients and HIV-positive patients tend to present at an earlier age and with more advanced disease than their immunocompetent counterparts.109,110,115 As in immunocompetent individuals, the larynx is the most common site of involvement, followed by the oral cavity and oropharynx.114,116 Tobacco cessation and alcohol abuse counseling, as well as early detection through biopsies of suspicious lesions, are warranted.
Higher susceptibility to infection with HPV in organ transplant recipients and HIV-positive patients may play an important role in the development of noncutaneous head and neck SCC.24,116 HPV infection has been associated with head and neck SCC, particularly of the tonsil.117 Organ transplant recipients and patients with HIV/AIDS also have increased incidence of other HPV-related cancers, such as cervical and anogenital cancer.23 It is thought that immune compromise could lead to increased infection rates and failure to resolve the infections.118 As more insight is gained into the pathophysiology and clinical behavior of HPV-related cancers, management strategies and the role of prophylactic HPV vaccination in immunosuppressed patients may be better defined.
Treatment of organ transplant recipients and HIV-positive patients with head and neck SCC should follow established guidelines. Even though surgical intervention remains an important option, patients often require combined therapy for advanced disease. The use of chemotherapy in these patients may put them at significant risk for opportunistic infection. Little data exist regarding the tolerance and efficacy of radiation therapy; however, patients with HIV have been shown to tolerate radiation therapy to the head and neck.119
Lymphoma
HIV-positive patients and organ transplant recipients are at higher risk for developing non-Hodgkin’s lymphoma (NHL) and Hodgkin’s lymphoma (HL) than immunocompetent individuals. EBV-associated NHL is the most common type of lymphoma to develop in both groups. NHL comprises a heterogeneous group of tumors, but the overwhelming majority (>95%) are B-cell derived.78 Lymphomas occurring in immunosuppressed organ transplant recipients are categorized within a disease entity known as posttransplantation lymphoproliferative disorder (PTLD). The WHO classification system divides PTLD into four categories120,121:
The WHO classification for AIDS-related NHL includes three groups122:
Most AIDS-related NHL are EBV-associated B-cell lymphomas such as Burkitt’s lymphoma, diffuse large B-cell lymphoma, and plasmablastic lymphoma (PBL).78,85,123,124
EBV, along with HHV8, is a member of the gammaherpesvirus family. It is estimated that more than 90% of the world adult population is infected.125 It is found in up to 50% of AIDS-related NHL126 and nearly 90% of PTLD.127 EBV alters tumor suppressor p53 gene expression and protein regulation, which is thought to play a role in oncogenesis,44,85 but given the nearly ubiquitous presence of EBV infection, it may be that the intensity of immune suppression determines the risk of developing NHL.126 However, immunosuppressed patients have not been found to have increased rates of EBV-associated nasopharyngeal cancer,128 and a large proportion of AIDS-related NHL have not demonstrated EBV. Thus the role of EBV in NHL oncogenesis remains unclear and is an area of ongoing investigation.
AIDS-Related Non-Hodgkin’s Lymphoma
Epidemiology and Presentation
AIDS-related NHL occurs in up to 19% of HIV-positive patients85 and is the second most common malignancy to develop in HIV-positive patients.129 Patients are much more likely than their immunocompetent counterparts to present with advanced disease, B symptoms (fever, chills, night sweats, and weight loss), and extranodal disease, including bone marrow involvement.78,85,129 Seventy percent to 80% of AIDS-related NHL is diagnosed with initial stage III or IV disease, in stark contrast to only 10% to 15% of high-grade NHL in the HIV-negative population.130–133 Whereas most NHLs develop in the setting of advanced immune suppression, NHL may occur in the setting of relative immune competence and thus cannot be ruled out based on a high CD4 count or low viral load. Burkitt’s lymphoma, in particular, has been noted to occur in patients with a relatively high CD4 count.134,135 Even though the incidence of Kaposi’s sarcoma has clearly declined since the advent of HAART, the effect of HAART on AIDS-related NHL has been less significant. Although some studies suggest a trend toward declining incidence, others have found an increased incidence of NHL in the HAART era.78,136
As with immunocompetent patients with NHL, AIDS-related NHL most commonly presents in the head and neck, with nearly two thirds of patients with head and neck manifestations.129 Extranodal disease is twice as common in AIDS-related NHL compared to non-HIV associated NHL.129,137,138 Extranodal head and neck sites include the oral cavity, sinonasal region, pharynx, nasopharynx, orbit, parotid gland, larynx, mandible, and CNS.139–145 Other sites of extranodal disease include the GI tract, bone marrow, and liver. Nodal disease predominates in the neck with frequent involvement of the submandibular, jugulodigastric, and supraclavicular regions.146
NHL of the head and neck often presents as a growing mass. Constitutional symptoms are frequently present with fever, night sweats, and unintentional weight loss (greater than 10% of body mass) present in 82% of patients.147 Initial symptoms are dependent upon the location of disease. Sinonasal lymphoma usually presents with nasal obstruction or other nonspecific symptoms consistent with chronic rhinosinusitis.148 NHL of the oral cavity most commonly affects the gingiva and palate and may manifest as a persistent sore, an enlarging mass, or loose teeth. Hoarseness, respiratory symptoms, and dysphagia may indicate laryngeal or pharyngeal disease. Nasopharyngeal lymphoma may present with nasal obstruction and serous otitis media. Because of the close association with HIV, patients newly diagnosed with NHL should be screened for HIV infection.
PBL is a unique type of diffuse large B-cell lymphoma that typically involves the oral cavity and mandible but has been reported in other regions including the paranasal sinuses, skin, and lymph nodes.78,149 Although first described as an AIDS-related NHL, there are reports of PBL occurring in HIV-negative patients and OTR.150–152 The gingiva and hard palate mucosa are most commonly involved and tumors have a propensity for adjacent bone invasion.153 These lesions must be distinguished from benign gingival enlargements like pyogenic granuloma and peripheral giant cell granuloma.149 Unlike other NHLs, PBLs are generally HHV8 and EBV-negative,150 although some series report positivity.152,154
Diagnosis
The diagnosis of lymphoma in HIV-positive patients begins with a high index of suspicion. Multiple common manifestations of HIV disease such as peripheral generalized lymphadenopathy and benign oral ulcerations may mimic the findings for patients with lymphoma. The combination of a rapidly enlarging lesion and constitutional symptoms is particularly concerning for NHL. The diagnosis of lymphoma may be made with fine needle aspiration (FNA) biopsy. Even though histologic type may be determined from a cell block following aspiration, this test may not be performed and some pathologists prefer larger tissue specimens for diagnosis and determination of histologic type. Thus open biopsies may be considered. The prognosis and management depend upon the presence of extranodal disease; therefore a thorough investigation of the CNS, mediastinum, and abdomen should be performed with magnetic resonance imaging (MRI), or CT if MRI is not available. Leptomeningeal disease is the most common manifestation of CNS involvement, occurring in up to 10% of patients,155 and is best evaluated with MRI.156 A bone scan and bone marrow biopsy may also be useful in the staging process.
Prognosis and Treatment
The International Prognostic Index (IPI), developed in 1993, is a clinical tool used to predict survival in NHL.157 Factors associated with poor prognosis include age older than 60 years, advanced tumor stage, elevated serum lactate dehydrogenase, poor performance status and more than one extranodal site of disease. It has been validated in patients with AIDS-related lymphoma, in that high-risk IPI scores have been shown to predict poor survival.130,158,159 One large study demonstrated 3-year survival rates for low, low-intermediate, high-intermediate, and high-risk groups to be 66%, 42%, 35%, and 8% respectively in the HAART era.160 Recent data suggest that, in the HAART era, the relationship between CD4 count and survival in NHL may be less significant than was previously thought.158,161 However, other reports show that CD4 count continues to be a predictor of survival.136,162,163 Before advent of HAART, overall survival in NHL was only 10%.78 Numerous studies have demonstrated improved survival when combining HAART with standard combination chemotherapy. Up to 92% of patients achieve complete remission136 and nearly 50% achieve 3-year survival even with high-intermediate risk IPI scores.130,164
AIDS-related NHL is typically treated with multiagent chemotherapy in combination with HAART.165 The therapy must balance the need to eradicate the neoplasm with the risk of further immune suppression. Systemic treatment of NHL may be complicated by marrow suppression, immunosuppression, mucositis, and opportunistic infections, resulting in a high rate of morbidity and mortality.85 Rituximab, an anti-CD20 antibody, has shown promising results in the treatment of NHL in HIV-negative patients, and clinical trials are ongoing to investigate its use in AIDS-related NHL. Radiotherapy has a role for patients with localized disease or for palliation of symptomatic lesions. NHL confined to the cervical nodes has an improved response to therapy with longer associated survival than extranodal disease involving the paranasal sinuses, mandible, and other extranodal sites.145 Primary CNS lymphoma has perhaps the poorest prognosis with its tendency to recur and association with profound immune suppression.133,166 Outcomes for relapsed, aggressive NHL are extremely poor and no effective treatments are available. Whereas the treatment of choice for relapsed NHL in immunocompetent patients is autologous stem cell transplantation (ASCT), opportunistic infections and toxicity have limited its use in AIDS-related NHL.165
Posttransplantation Lymphoproliferative Disorder
PTLD is a complication of solid organ and hematopoietic transplantation marked by EBV-driven abnormal lymphoproliferation. The overall incidence in adults is approximately 2% to 3%,127 while in children the incidence is close to 8%.167 It is the second most common malignancy to develop in organ transplant recipients, after cutaneous malignancy.114 Nearly 40% of patients with PTLD present with findings in the head and neck, especially in Waldeyer’s ring and cervical lymph nodes.168 PTLD involving the head and neck usually presents with mononucleosis-like symptoms and adenotonsillar hypertrophy, and may be associated with upper airway obstruction.114,168,169 There are also reports of PTLD involving the nasal cavity and paranasal sinuses,170,171 as well as facial cutaneous manifestations.140
Most PTLDs are NHLs of the B-cell type.114 Incidence varies significantly with the type of organ transplanted, with the highest rates in multiorgan or intestinal transplants and lowest rates in renal transplants.127 PTLD develops in the setting of severe immunosuppression, which impairs the formation of a cytotoxic T lymphocyte immune response to EBV. The risk of developing PTLD is highest in EBV-naive patients, which may account for the higher incidence in children. It occurs in a bimodal temporal distribution, with most cases occurring within the first 2 years after transplantation and a smaller peak occurring later than 2 years after transplantation.127
Treatment should be directed first toward decreasing immunosuppression regimens to allow an adequate T-cell response. Up to 50% of patients respond to immunosuppression reduction.172,173 Patients who fail to respond to immunosuppression reduction may require systemic therapy such as combination chemotherapy, cytokine therapy, or anti-CD20 therapy (rituximab).114,127 Surgery is reserved for management of local symptoms, and acute upper airway obstruction caused by tonsillar hypertrophy may require tonsillectomy.174
Hodgkin’s Lymphoma
Hodgkin’s lymphoma can be seen in transplant recipients and patients with HIV. In transplant patients, it is a much less common manifestation of PTLD than NHL, comprising only 1.8% to 3.5% of all PTLDs,175 but its incidence is still 15-fold greater than that in the general population.176 Risk factors for the development of HL in transplant recipients include bone marrow transplantation and a history of graft-versus-host disease (GVHD).175,177 HL-like PTLD is clinically similar to classic HL occurring in the posttransplant setting but is distinguished by its pathologic features such as cell markers and background proliferation.175,177 HL occurring in the setting of PTLD has a better prognosis than other PTLD lymphomas176 and should be managed like other PTLD lymphomas with a focus on reduction of immunosuppression and appropriate chemotherapy.175
Hodgkin’s Lymphoma in Human Immunodeficiency Virus
Although not considered an AIDS-defining malignancy, HL is the most common non–AIDS-defining tumor occurring in patients with HIV.123,178 The incidence of HL is increased nearly 5- to 15-fold in patients with HIV (HIV-HL) compared to that in HIV-negative patients.175,179 Data suggest that HIV-HL is more common in homosexual and intravenous drug–using men than in other HIV risk groups.85,180 Several features distinguish HIV-HL from HL occurring in HIV-negative patients. HIV-HL is more likely to present with advanced disease, extranodal involvement including bone marrow in up to 50%, and B symptoms in 40% (compared to 27% in HIV-negative patients).178,180,181 In both groups, cervical lymphatic involvement is common; one series demonstrated cervical node involvement in all cases of HIV-HL.180 Notably, EBV is present in more than 90% of HIV-HL patients compared to less than 50% of non-HIV patients.123,181 The histology of HIV-HL is also distinct from that of the general population. HIV-positive patients more frequently develop the aggressive mixed cellularity and lymphocyte-depleted subtypes in comparison to the predominance of nodular lymphocytic subtypes in the general population.179 The most surprising finding in HIV-HL is that its incidence has unexpectedly increased in the HAART era. Patients with moderate immunosuppression (CD4 between 225 and 249 cells/µL) are more than twice as likely to develop HIV-HL than patients with severe immunosuppression (CD4 < 25 cells/µL).179 It is believed that increased CD4 counts allow for improved survival of Reed Sternberg cells, which are the characteristic pathologic cells seen in HL.182 Upon diagnosis of HIV-HL, staging evaluation should include CT imaging of the brain, chest, and abdomen, as well as a bone marrow biopsy.123 Evaluation of the patient’s immune status should also be performed with CD4 count and viral load testing.
There have been no randomized controlled trials for treatment of HIV-HL,183 but the accepted standard of treatment consists of combination chemotherapy and antiretroviral therapy. Patients treated with HAART in combination with chemotherapy have shown improvement in response to therapy, longer disease-free survival, and longer overall survival when compared with patients treated with combination chemotherapy alone.181,184 Estimated 2-year survival improved from 45% in patients treated with chemotherapy alone to 62% in those treated with combined HAART and chemotherapy.185 Complete response to therapy and overall survival are much worse than in HIV-negative patients, however. Overall survival rates in HIV-HL patients compared with HIV-negative patients were 68% vs. 92% at 1 year and 41% vs. 77% at 5 years.181
Salivary Gland Disease
Parotid Lesions in Human Immunodeficiency Virus
Parotid lesions are common in HIV-positive patients, especially children in whom up to 18% may present with parotid masses.186–188 These lesions may result from AIDS-related malignancies such as NHL143,144 or KS189,190 or from diffuse infiltrative lymphocytosis syndrome (DILS), but the majority of parotid enlargement in HIV-positive patients is the result of a benign process known as benign lymphoepithelial cyst (BLEC). BLEC of the parotid produces persistent, nontender parotid enlargement associated with cervical lymphadenopathy in up to 90% of patients.191 Inspissated secretions can lead to duct obstruction with resulting sialadenitis and pain.192 These lesions have varying proportions of cystic and solid components,193 and even though only unilateral clinical disease may be evident, radiologic evaluation nearly always reveals bilateral changes.194,195 Histologically, BLEC has cyst walls lined by hyperplastic and metaplastic squamous epithelium, and contains aggregates of lymphoid proliferation.191 The differential diagnosis of cystic parotid lesions includes Sjögren’s syndrome, cystic Warthin’s tumor, and branchial cleft cysts. Bilateral cystic Warthin’s tumors may be differentiated radiologically from BLEC based on the presence of focal nodularity in Warthin’s tumors and the associated lymphadenopathy of BLEC.195
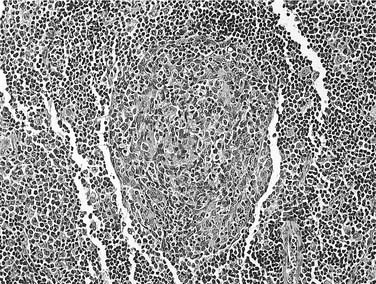
FNA can be useful in the diagnosis of parotid masses. Unilateral masses, or masses suspicious for malignancy, should undergo FNA. A study of 99 parotid FNAs in HIV-positive patients found 75% to be consistent with BLEC, 14% were infectious/inflammatory, and 6% were neoplastic. Of the neoplastic lesions, all were malignant, with three NHLs, one multiple myeloma, one metastatic adenocarcinoma from the lung, and one direct extension from a cutaneous BCC. In 6% of patients, the FNA was nondiagnostic.196 FNA of BLEC reveals a heterogeneous lymphoid population, scattered foamy macrophages, and anucleated squamous cells in a proteinaceous background.191 Germinal centers, myoepithelial islands representing metaplasia of ductal epithelium, and cystic ductal dilatation (Fig. 15-3) differentiate BLEC from lymphoma.197 Aspiration may also be useful for the relief of symptoms in larger cysts, although the lesions may recur. Initial aspirates should be sent for cytologic and microbiologic evaluation.
A variety of treatment options exist for patients with BLEC. Patients not already receiving HAART should be referred for treatment initiation because these lesions may regress with antiviral therapy.198,199 For minimally symptomatic patients without significant cosmetic deformity, observation alone represents the best option. Low-dose radiation treatment results in a greater than 50% reduction in the size of the lesion. This improvement, however, typically lasts less than 10 months.200 Some patients are treated with repeated needle aspirations although the repetitive nature of this treatment is suboptimal. Needle aspiration combined with doxycycline or tetracycline sclerotherapy may result in significant size decrease.201–203 A 1 mg/mL doxycycline solution has been used, injecting 1 to 2 mL through an intravenous catheter following cyst aspiration. Most patients are left with smaller residual fibrotic masses but long-term results of this treatment are unknown.201,202
HIV-associated BLEC typically does not require parotidectomy. Parotidectomy may be considered in those rare cases of BLEC that undergo rapid size change, are disfiguring, or have significant pressure symptoms. Other indications for parotidectomy include FNA cytology suggestive of neoplasm or unilateral masses with a significant solid component or features worrisome for malignancy. There have been no reports of malignant transformation of BLEC.191
Diffuse Infiltrative Lymphocytosis Syndrome
Parotid gland enlargement associated with sicca symptoms was recognized in HIV-positive patients in the 1980s, and Itescu and colleagues first categorized DILS as a discrete entity in 1990.204 DILS occurs in the setting of HIV and is marked by salivary gland enlargement with circulating and visceral CD8 lymphocytosis. It is often associated with cervical lymphadenopathy, and up to 60% of patients also report sicca symptoms such as xerostomia and xerophthalmia.192,205,206 In the pre-HAART era, DILS occurred in approximately 3% to 4% of HIV-positive patients, but in the HAART era its incidence has dropped significantly to less than 1%.205 It occurs twice as frequently in black patients as in whites.207,208 Diagnosis is confirmed with minor salivary gland biopsy demonstrating lymphocytic infiltration or with positive gallium Ga67 scintigraphy when biopsy is not possible.192
DILS is phenotypically similar to Sjögren’s syndrome in terms of salivary gland enlargement, sicca symptoms, and salivary gland histology. However, whereas parotid swelling is universal in DILS, it occurs in less than one third of patients with Sjögren’s syndrome.192 In addition, DILS is characterized by more frequent occurrence of extraglandular lymphocytic infiltration, predominance of CD8 cells in lymphoid aggregates (as opposed to CD4 in Sjögren’s syndrome), and the infrequent presence of serum autoantibodies.192,205 Extraglandular disease in DILS may include lymphocytic interstitial pneumonitis in up to half of patients,209 but this complication is becoming less common in the HAART era.205
Xerostomia in Human Immunodeficiency Virus and Transplant Recipients
HIV-positive patients as well as patients who suffer from chronic graft-versus-host disease (GVHD) are significantly affected by xerostomia. The prevalence of xerostomia is reported to be 2% to 10% in HIV-positive patients.210 Chronic GVHD may develop in up to 40% to 70% of patients receiving transplants from matched, unrelated donors,211 and 80% of patients with extensive GVHD complain of xerostomia.212 In the setting of HIV, some cases are iatrogenically induced by the use of HAART, antidepressants, and other drugs, whereas other cases are caused by chronic mouth breathing secondary to sinonasal disease or adenoidal hypertrophy. Still others are related to DILS as noted earlier. Salivary flow rates are diminished in the setting of HIV infection or GVHD,210,213,214 which leads to increased incidence of dental caries and impaired deglutition. Salivary substitutes, frequent saline rinses, and sialogogues help alleviate these problems. Dental caries can be prevented with fluoride.
Cervical Lymphadenopathy in Immunosuppressed Patients
Although constitutional symptoms by themselves are not a specific indicator of infection or malignancy, the presence of such symptoms without a known cause warrants further investigation. Cervical lymphadenopathy caused by lymphoma in immunosuppressed patients is associated with B symptoms in nearly 50% of patients.78,178,180,215 The distribution, size, and mobility of neck nodes may suggest infectious or malignant etiology. Cervical lymphadenopathy that is greater than 2 cm, unilateral, painful, deep, or asymmetric is suspicious for a pathologic cause, specifically granulomatous disease or lymphoma.216 Tender adenopathy is more likely to be secondary to bacterial infections, including tuberculosis, whereas nontender, enlarging neck nodes may result from malignancy.217 A thorough head and neck examination should search for potential primary sites of infection or malignancy.
Organ transplant recipients are at higher risk for development of cervical involvement, which is seen in 6% of patients, and extrapulmonary tuberculosis, which is seen in 10% to 20%.218 Head and neck sites of tuberculosis infection in organ transplant recipients include the cervical lymph nodes,219 larynx,220,221 and middle ear.222 Nontuberculous mycobacterial infections also occur in immunocompromised patients and frequently cause pulmonary infections, cervical lymphadenitis, and ulcerative skin lesions, but has also been reported to involve the temporal bone.223
Cervical Adenopathy in Human Immunodeficiency Virus
Data regarding the incidence of cervical adenopathy are sparse, but one series demonstrated that of all HIV-positive patients presenting for FNA of an enlarged lymph node, 54% had cervical involvement.224 Approximately 40% of HIV-positive patients with adenopathy have benign reactive lymphadenopathy.225–227 and 20% to 30% have tubercular etiology.226 In this background of hyperplastic adenopathy, however, cases of infectious and neoplastic etiology exist, including Mycobacterium tuberculosis, fungal infection, Pneumocystis jiroveci (formerly Pneumocystis carinii), lymphoma, KS, and other processes that also occur in the general population (Table 15-6).224,225,227 Malignancy may be present in up to 10% of cases. Differentiating among these processes remains challenging, yet critical to the appropriate management of lymphadenopathy in the setting of immunosuppression. The tendencies for multiple pathologic processes to coexist in this population and the poor sensitivity of many clinical findings and tests often make it necessary to perform microbiologic and histologic evaluations of lymph node tissue.
Table 15-6 Differential Diagnosis of Lymphadenopathy in Immunosuppressed Patients
Extrapulmonary tuberculosis is the second most common cause of cervical lymphadenopathy in HIV-positive patients.224,225,227 Tuberculosis is the leading cause of death in HIV-positive patients and effective treatments are available, thus prompt diagnosis and initiation of therapy are essential. The most common site of extrapulmonary tuberculosis involvement in HIV-positive patients is the lymph nodes,224 but other head and neck sites that may be involved include the larynx,220,228,229 oral cavity and lip,230,231 or the parotid gland.232
The patient’s immune status, as indicated by the history of opportunistic infection, CD4 count, and viral load, may help narrow the differential diagnosis of cervical adenopathy. Whereas NHL and mycobacterial infection are more likely to be present in the setting of advanced immunosuppression, the CD4 count alone is not sufficient to exclude malignancy or infection.134,135,179 The purified protein derivative (PPD) or tuberculin skin test may facilitate the diagnosis of mycobacterial lymphadenitis. In the setting of advanced HIV infection and immune compromise, however, the patient may become anergic, resulting in a low sensitivity of the PPD test. The criterion for a positive result in an HIV-infected patient is a skin reaction greater than 5 mm in diameter rather than 10 mm as in the general population.
FNA biopsy should be the initial method of tissue sampling in most cases of suspicious cervical lymphadenopathy in the HIV-infected patient. If possible, a cell block should be created from the sample and the sample should be sent for cytology, flow cytometry, and culture and stains for aerobic and anaerobic bacteria, mycobacteria, and fungi. In the largest series among HIV-positive patients, FNA provided a definitive diagnosis in more than half of patients and directed further clinical investigation in another 30%, while 20% of the initial samples were inadequate for pathologic evaluation.227 A subsequent study in children corroborated these results, with only 8% of samples being inadequate for diagnosis.225
An FNA diagnosis of follicular hyperplasia should be correlated with the clinical picture and should not, by itself, absolve the clinician’s suspicion of lymphoma. The decision to perform a diagnostic open biopsy should be driven by a suspicion of malignancy or infection in the setting of a negative or inconclusive FNA.217,233 Conditions that would favor proceeding with an open biopsy in the setting of an inconclusive finding include nodes greater than 2 cm and growing; onset associated with a low CD4 count; asymmetric, unilateral, or localized lymphadenopathy; constitutional symptoms of unknown origin; mediastinal adenopathy; or hepatosplenomegaly. Open biopsy of suspected metastatic carcinoma should be avoided and, if possible, the diagnosis should made by FNA. When metastatic carcinoma is diagnosed, a thorough examination of the upper aerodigestive tract should be performed under general anesthesia in search of a primary tumor.
Sinonasal Infection
Presentation and Pathogenesis
As in the general population, sinonasal complaints are common among immunosuppressed patients. Reports of prevalence among HIV-positive patients range from 10% to 68%, depending on the criteria used.234–237 Nearly 50% of patients undergoing allogeneic stem cell transplantation (STC) complain of rhinosinusitis, and 12% of solid organ transplant recipients develop acute sinusitis.238,239
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree