This chapter provides an overview of several important general principles that are highly relevant to the safe and efficient surgical management of chronic ear disease. Although a diversity of opinion exists in the otologic surgical community regarding many of the fine points outlined here, it is critical for surgeons at all levels to consider the general themes discussed and to take primary responsibility for assuring a controlled surgical environment according to the existing evidence in the medical literature, official surgical practice standards, surgical experience, and general attention to detail. The author has published in-depth descriptions of the preferred techniques of middle ear and mastoid surgery outlined in this chapter.1
Temporal bone surgery is complex and challenging; therefore, it is imperative that a structured approach to each stage of surgery is adopted to optimize patient outcomes. Complications and difficulties that arise in temporal bone surgery can often relate to inadequate exposure or suboptimal operating conditions. Surgeons must recognize and minimize these conditions given that they could increase the risk of complications in advance. Many of these factors are outlined below. Correct patient positioning, local anesthesia infiltration for hemostasis, and correct surgical angles affording wide exposure, with identification and skeletonization of landmarks, are among some of the critical principles for performing effective and efficient microsurgery of the ear and skull base.
Factors that may contribute to a poor otologic surgical environment include the following:
Malfunctioning operating room equipment
Defective surgical instruments
Disruptive noise or commotion
Operating room staff and anesthesia colleagues unfamiliar with otologic surgery
Excessively bloody surgical field
Insufficient hypotensive anesthesia
Poor preoperative local vasoconstrictor medication infiltration
Coagulopathy
Severe acute tympanomastoid inflammation
Distorted tissue planes
Trauma
Prior surgery
Tumor
Infection
Unfavorable patient anatomy
Large body habitus with prominent shoulder/chest
Poor neck flexibility
Sclerotic temporal bone
Congenital abnormalities
Inadequate surgical exposure
Surgeon avoidance of identifying critical landmarks
Lack of canalplasty
Inadequate soft tissue (postauricular) exposure
Narrow mastoidectomy without saucerization
18.2 Operating Room Layout
The operating room is laid out as shown in ▶ Fig. 18.1. This is the positioning for a left ear. The surgeon sits to the patient’s left, with the scrub nurse directly opposite. The microscope is at the cranial end of the table and is connected to a monitor. This must be at a comfortable viewing angle for the scrub nurse, whose role is as vital as that of the surgeon given that it will facilitate ease of instrument transfer and allow the nurse to anticipate what will be needed within the operative field. The anesthetist, anesthetic equipment, facial nerve monitoring equipment, and other space-occupying items (e.g., drip stands) are at the caudal end of the table. This minimizes the number of leads and wires around the surgeon’s chair. The surgeon controls the drill foot pedal, and the scrub nurse or assistant controls the foot pedal for the bipolar diathermy.
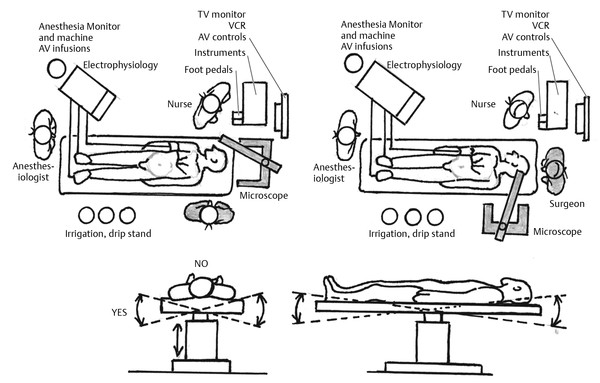
Fig. 18.1 Layout of operating room and personnel positioning for ear surgery. (Top Left) Standard layout for tympanomastoid surgery. (Top Right) Modified layout for middle cranial fossa approach. (Bottom left) The bed can be “airplaned” toward or away from the surgeon during tympanomastoid surgery to change the angle of view into the ear. (Bottom right) The incline of the bed is raised with “reverse Trendelenburg” positioning to increase the height of the head relative to the feet. The opposite “Trendelenburg” positioning may be undertaken during middle cranial fossa surgery to lower the head relative to the feet.
18.3 Patient Positioning
The patient should be prone and the operating table tilted head up (reverse Trendelenburg). The head is flat on the table and turned to the opposite side, supported by a padded sandbag. The angle between the head and shoulder should be obtuse to allow enough space for the surgeon’s hands to work comfortably. The patient is positioned so the shoulder is in line with the edge of the table, and the position is secured with three straps across the shoulders, abdomen, and feet, respectively. This facilitates changing the angle of view by moving the whole table up or down or by rotating the table to or away from the surgeon (▶ Fig. 18.2).
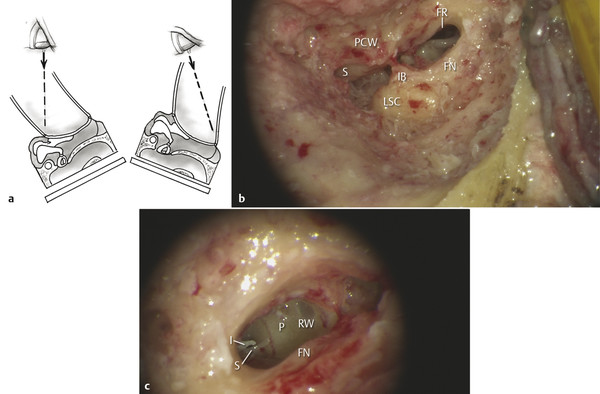
Fig. 18.2 (a) View down microscope with accompanying diagram of table position. The angle of rotation impacts the view into the (b) facial recess (S, short process of incus; PCW, posterior canal wall; IB, incus buttress; LSC, lateral semicircular canal; FN, facial nerve; FR, facial recess) and (c) round window niche (RW, round window; P, promontory; FN, facial nerve (in Fallopian canal); S, stapes; I, Incus (long crus)).
Certain patient factors can make optimal positioning difficult. Frequently encountered conditions include an immobile neck with a fixed flexion deformity, which can affect surgical lines of vision, or obesity, which can diminish the angle between the shoulder and neck and affect soft tissue and the parotid gland. To prevent patient injury and optimize operating conditions, a patient with an immobile neck may need a padded block or head ring under the head. The angle between neck and shoulder in an overweight patient can be increased by gently retracting the shoulder caudally and securing it with surgical adhesive tape.
18.4 Surgeon Position
It is important to consider briefly the ergonomics of the surgeon’s sitting position and his or her ability to carry out fine movements of the hand under the microscope without difficulty. The surgeon should be seated comfortably. Correct positioning of the operating table, chair, and microscope can facilitate this by enabling the surgeon to sit in the correct position with feet flat on the floor, back straight and supported, and hips at a 90-degree angle. The chair should have wheels and the armrests should be draped to decrease tension on the arms during prolonged periods operating through the microscope. The focal length (typically 240–260 cm) and microscope body size will determine the surgeon’s distance from the operative field, and, therefore, will maintain the arms in the optimal position.
18.5 Preoperative Surgical Safety Protocol
In 2009 the World Health Organization (WHO) published guidelines for the provision of safe surgery (“Safe Surgery Saves Lives”). The first objective described in this guideline is the recommendation that all surgical operations are preceded by use of a universal protocol designed to ensure that the correct operation is carried out at the correct site on the correct patient. This protocol contains three steps:
Step 1. Verification. Identification of the correct procedure, site, and patient is checked and documented at every stage of the patient’s preoperative assessment, including when the procedure is initially scheduled, at the time of the patient’s entry to the operating room, at any stage when the patient’s care is being transferred to another person, and before the patient leaves the operating room.
Step 2. Marking. The patient must be marked at the operative site with a clear unambiguous mark using a marking agent that will allow the mark to remain visible during the patient’s preparation for surgery. The mark should be made by the surgeon performing the operation or a delegate who will be present during surgery.
Step 3. Time Out. A short pause, often referred to as a surgical pause or team time out, should take place immediately before the operation commences. All team members should be involved to allow any concerns or inconsistencies to be raised.
In our unit we follow a five-step process, similar to that described above, recommended by The Australian Commission on Safety and Quality in Healthcare2 that also requires confirmation that appropriate and available diagnostic imaging is available in the operating theatre. This protocol has been shown to reduce the incidence of wrong-site surgery in Western Australia.3
18.6 Preparation and Draping of Operative Site
A 10% povidone-iodine solution is used to prepare the surgical site. Judicious hair clipping is undertaken to prevent hair from interfering with the operation and is limited to the incision site. The remaining hair is brushed out of the surgical field. Hair removal preoperatively does not seem to reduce the rate of surgical site infection.4 Specifically, a randomized controlled trial comparing the rate of surgical site infections in ear surgery between groups, with and without hair removal, has demonstrated that hair removal prior to surgery via a postauricular incision had no effect on the rate of surgical site infection.5
The surgical site is draped in layers, and measures are taken to ensure the drapes are effectively adherent to the skin to prevent irrigation fluid leaking into the non-sterile area. Four sterile absorbable cotton towels create an initial square drape. An otologic ENT split drape with adhesive edges forms the second layer. An irrigation pouch is secured to the split drape. The pinna is reflected forward and held in position with an adhesive antimicrobial incision drape. Once the initial skin flaps have been raised, they are retracted out of the surgical field with a posterior blanket suture through the drapes and skin (▶ Fig. 18.3).
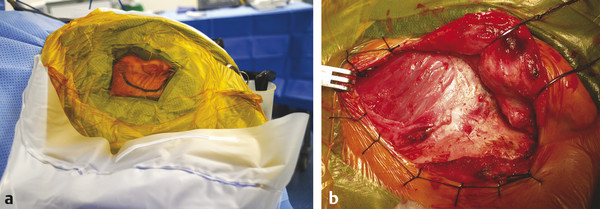
Fig. 18.3 Prepped and draped operative site. (a) As a final layer, an adhesive antimicrobial drape is placed to hold the pinna of a left ear forward and to expose the postauricular skin prior to incision. The adhesive drape passes into the irrigation pouch, thereby providing a watertight sealed junction so that irrigation fluid is properly contained. (b) A large piece of temporalis fascia has been harvested from this right ear prior to planned canal wall-down mastoid surgery. The soft tissues are held out of the surgical field with a running locking silk blanket suture and the pinna is reflected forward with the assistance of blunt hooked retractors.
18.7 Anesthesia
The type of anesthesia used depends on both patient factors and the type of surgery planned. The authors use general anesthesia for most ear surgery and consider it preferable for operations that include significant bone work. Local anesthesia can be used successfully in several areas of otology, particularly for stapes and middle ear surgery6,7 and, more recently, for cochlear implantation.8
18.7.1 General Anesthesia
Traditionally, induction of general anesthesia involves use of both inhalational and intravenous agents; however, some inhalational drugs, particularly nitrous oxide, have been shown to alter middle ear pressure.9 This may increase the risk of surgical complications during or after ear surgery by inflating the middle ear space and causing graft detachment. Avoidance of inhalation anesthetic agents is therefore preferable for otologic procedures, with the lone exception of temporary usage during induction specifically in cases with tympanic membrane atelectasis as a means of promoting outward eversion of the retracted drumhead.
At our institution, the combination of remifentanil and propofol is preferred for general anesthesia. Anesthesia is induced with propofol and remifentanil and maintained with 40% oxygen and 60% air, together with propofol and remifentanil. Total intravenous anesthesia (TIVA) not only avoids inducing pressure changes in the middle ear, but also facilitates maintenance of hypotensive anesthesia. The combined use of both a centrally-acting sedative (frequently a potent opiate, such as remifentanil, or a benzodiazepine, such as midazolam) with an intravenous anesthetic agent (such as propofol) allows for effective anesthesia to be achieved. Agents such as those highlighted above have desirable pharmacokinetic properties, with rapid onset and offset of effect, and a favorable adverse event risk, particularly in relation to optimizing the condition of the surgical field and minimizing nausea in the postoperative period.10
Hypotensive anesthesia, defined as the use of anesthetic agents to achieve a systolic blood pressure during surgery of 80–90 mm Hg or a mean arterial pressure of 50–65 mmHg, is often used in ear surgery with the aim of reducing bleeding during surgery to maintain a bloodless field.11 Hypotensive anesthesia can be achieved with several different inhalational and intravenous agents. Cardiovascular active medications, such as sodium nitroprusside, clonidine, or nifedipine, or beta-adrenergic receptor antagonists, such as esmolol, can be used concurrently with anesthetic agents to achieve appropriate blood pressure and heart rate control. However, continuous monitoring of cardiovascular status and urine output intraoperatively and during the postoperative period is required with the use of these agents. This may involve use of central venous pressure catheters and intra-arterial catheters in some patient groups, including those undergoing surgical procedures with longer duration of anesthesia or those with additional comorbidities.
18.7.2 Local Anesthesia
Local anesthesia infiltration is undertaken for all otologic procedures. Local anesthetic agents produce a reversible block in the transmission of impulses along nerve fibers and, when used in combination with a vasoconstrictor, reduce the risk of systemic toxicity and intraoperative bleeding and prolong duration of action. Local anesthetic agents vary in terms of their potency and toxicity. Both lidocaine and bupivacaine are vasodilating amides available in injectable forms. Lidocaine can be administered to a maximum dose of 3 mg/kg or 7 mg/kg with adrenaline. The maximum dose of bupivacaine is 2 mg/kg or 3 mg/kg with adrenaline.
Complications are rare but may be potentially serious and are related either to toxicity or hypersensitivity. Manifestations of toxicity typically start as the drug is being administered or directly afterward and include cardiovascular complications, such as hypotension, bradycardia, or cardiac arrest, or neurological complications, such as seizures or coma. Recognition of impending toxicity requires immediate cessation of drug administration and may require monitoring in a High Dependency Unit in cases of severe, non-self-limiting reactions. For procedures carried out under general anesthetic, we use a solution of 0.5% lidocaine with 1:100,000 adrenaline. A higher concentration of 2% lidocaine is used when the procedure is carried out under local anesthesia.
Knowledge of the sensory innervation of the ear is necessary to achieve thorough local anesthesia. Four sensory nerves predominantly supply the external ear and external auditory canal: the greater auricular nerve, the auriculotemporal nerve, the lesser occipital nerve, and the auricular branch of the vagus nerve.
The greater auricular nerve (C2–3 branches of the cervical plexus) supplies the lower two-thirds of the anterior and posterior external ear. The auriculotemporal nerve (branch of V3) supplies the anterior superior one-third of the ear (tragus, crus helix, superior helix) and provides sensory information from the anterior wall and roof of the external auditory canal. The lesser occipital nerve (C2 branch of the cervical plexus) supplies sensory information from the posterior surface of the superior one-third of the external ear. The auricular branch of the vagus nerve (Arnold nerve) innervates the concha and posterior wall and floor of the external auditory canal. Small branches from the facial nerve contribute to the sensory innervation of the concha and the roof of the external auditory canal (▶ Fig. 18.4).
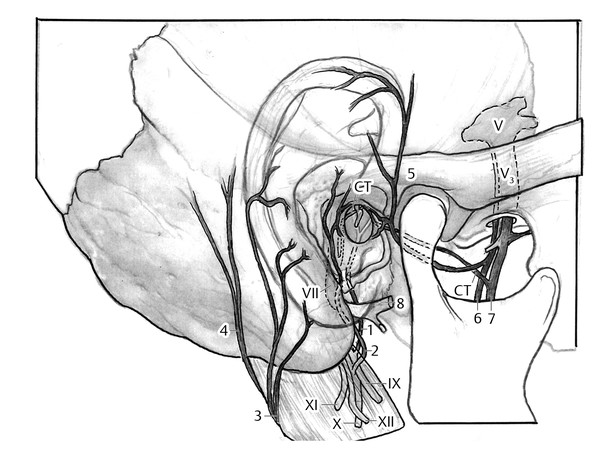
Fig. 18.4 Illustration of innervation of the ear. 1, auricular branch of CN IX; 2, auricular branch of CN X; 3, greater auricular n.; 4, lesser occipital n.; 5, auriculotemporal branch of CN V; 6, inferior alveolar n.; 7, lingual n. and chorda tympani n.; 8, CN VII (facial nerve).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


