referred to by a variety of terms including “zygomycosis,” “mucormycosis” and “invasive aspergillosis,” which encompassed almost all of the pathogens associated with human-invasive fungal disease. Revisions of the phylogenetic classification within the kingdom Fungi have eliminated the phylum Zygomycota and therefore the pathologic term Zygomycosis has been made obsolete (7). Mucor, Rhizopus, and many of the organisms responsible for invasive disease of the Order Mucorales are instead assigned to the subphylum Mucoromycotina until further study better delineates the relationship between these organisms (8). Finally, it has become clear that the various forms of fungal rhinosinusitis are primarily determined by host factors; thus the current classification scheme does not include the names of the organism.
TABLE 37.1 CLASSIFICATION OF FUNGAL RHINOSINUSITIS | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||
TABLE 37.2 FEATURES OF INVASIVE FUNGAL RHINOSINUSITIS | |||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||
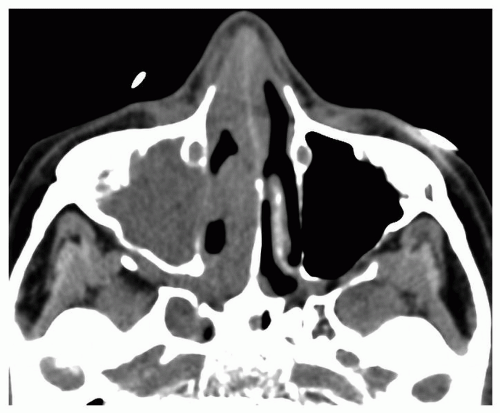
or chemotherapeutic agents, are particularly prone to development of AIFR (4,10). A high index of suspicion for invasive disease should be maintained in the immunocompromised patient with symptoms of rhinosinusitis, as early findings are often subtle.
studied by several authors, yet a clear consensus is lacking for the role of this type of histopathologic analysis in intraoperative management. Ghadiali et al. report the largest series to date for frozen section analysis in AIFR with an overall sensitivity of 84% for frozen section studies of biopsies to establish disease with no significant difference between the sensitivities of frozen section identification for Mucor when compared to that for Aspergillus (16). However, the use of histopathologic controls for surgical margins can be misleading and probably does not offer any additional assurance of disease eradication greater than the clinical appreciation of freely bleeding tissue margins (16). Tissue processing during preparation of the frozen section can be compromised by necrosis, hemorrhage, and distortion of the fungi such that even in cases where invasive fungi are identified, morphologic features that allow speciation cannot be appreciated (16,17). While fungal culture remains the gold standard for identifying the culprit fungi and guiding the choice of antifungal therapy, such cultures remain negative in 30% to 50% of cases (18,19), and prior or concomitant administration of antifungal agents also decrease yield on fungal culture. Newer methods such as in situ hybridization technology with oligonucleotide probes to identify fungus-specific ribosomal RNA sequences hold promise. This technique has advantages over traditional fungal culture of both greater speed and higher sensitivity in determining the involved fungal species, allowing more rapid institution of targeted antifungal therapy (20).
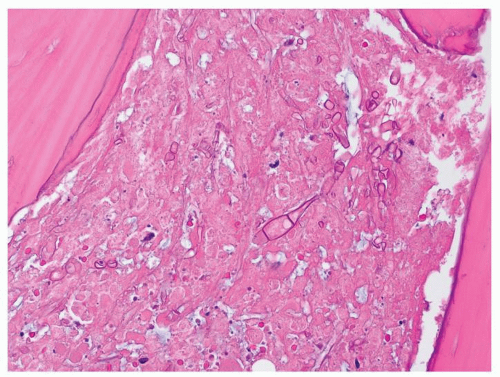
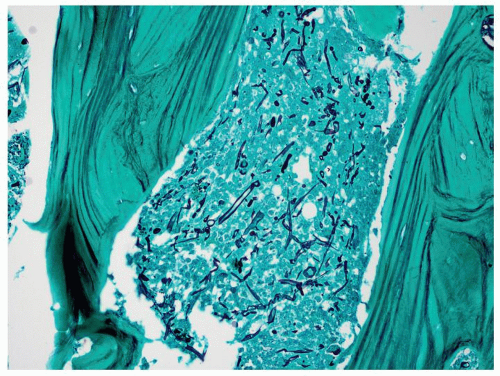
is defined by fungal forms invading into the submucosal tissue, often associated with angioinvasion resulting in vascular thrombosis, infarction, and necrosis. Areas of coagulative tissue necrosis are extensive and the total numbers of hyphae can be relatively sparse at times, with an associated neutrophilic infiltration (9). The acidotic environment of tissue ischemia and necrosis provide an ideal medium for fungal growth and further propagation of the fungal infestation (12).
well as prophylaxis of recurrent invasive fungal infections in immunocompromised populations. Voriconazole produces survival advantages with fewer side effects than amphotericin B in the treatment of systemic invasive aspergillosis (30), while posaconazole demonstrates both survival benefits and reduced drug costs over standard therapy with amphotericin B in the management of the same invasive fungal condition (31). Posaconazole is active against the mucoracea while voriconazole is not. Posaconazole has been shown to be a cost-effective medication in the prevention of invasive fungal disease in neutropenic patients (25). The efficacy of posaconazole and voriconazole in treating Aspergillus AIFR has been reported. Baumann reported the successful use of voriconazole following sphenoidotomy in four cases of AIFR resulting from Aspergillus, while Chirch reported a successful case of combination therapy with caspofungin and voriconazole for Aspergillus AIFR in the absence of surgery (32,33). The reduced side effects of these agents may prompt their use both in treatment and prophylaxis within the medically compromised patient population at risk for AIFR. At the present time, however, clinicians should institute voriconazole therapy without hesitancy when either Pseudoallescheria boydii or Aspergillosis is identified. P. boydii
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree