Gene Defect (inheritance) |
Age of Onset |
Phenotype |
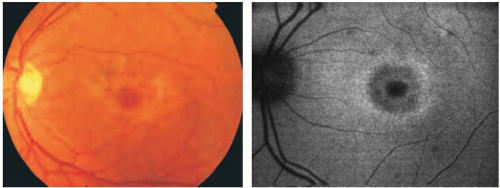
Fundus Changes |
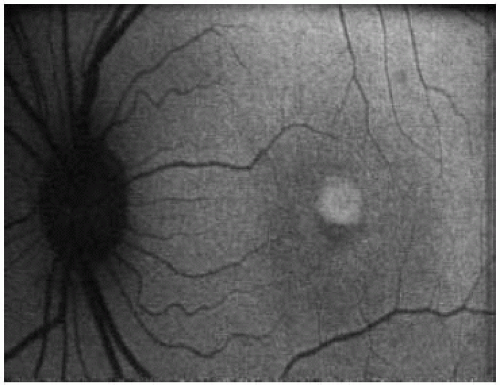
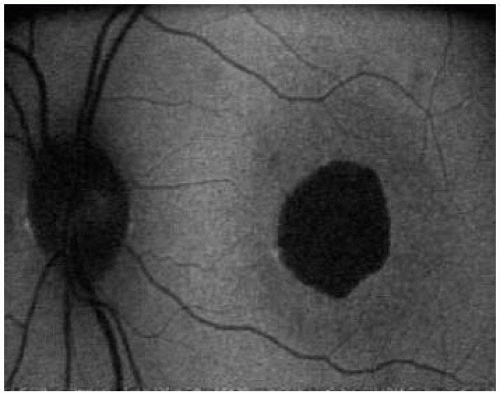
AF |
PERG |
ERG |
|---|
GUCA1A (AD) |
3rd-5th decade |
COD, CORD |
ranges from mild macular RPE disturbance to RPE atrophy, with normal peripheral retina |
focal increased AF at the macula; perifoveal rings of increased AF; reduced AF in areas of atrophy |
severely reduced or undetectable |
severely reduced amplitude of single flash and flicker with minimal or no 30Hz flicker implicit time shift; in CORD, additional reduction in rod responses |
GUCY2D (AD) |
1st, 2nd decade |
CORD |
macular and peripheral atrophy |
increased foveal in areas of atrophy AF; reduced AF in areas of atrophy |
severely reduced or undetectable |
reduced cone and rod responses;* “negative ERG” |
Peripherin (AD) |
2nd, 3rd decade |
CORD |
macular RPE mottling, macular atrophy, peripheral retinal atrophy and areas of peripheral RPE hyperpigmentation |
“speckled” AF |
reduced responses |
reduced cone and rod responses |
CRX (AD) |
1st decade |
CORD |
macular and later peripheral retinal degeneration |
reduced AF in areas of atrophy, “speckled” AF in the midperipheral retina |
reduced responses |
reduced cone and rod responses; “negative ERG” |
RIMS 1 (AD) |
2nd-5th decade |
CORD |
ranges from mild macular RPE disturbance to atrophy and pigmentation• |
reduced AF at the center of the macula surrounded by a ring of increased AF |
absent or severely reduced |
reduced cone and rod responses, often normal 30Hz flicker implicit time |
ABCA4 (AR) |
1st-3rd decade |
COD, CORD |
macular atrophy, bull’s eye appearance, peripheral pigmentary changes in advanced disease |
reduced AF at the center of the macula surrounded by a ring of increased AF; reduced AF only |
absent or severely reduced |
severely reduced cone responses; in CORD, additional reduction in rod responses |
KCNV2 (AR) |
1st, 2nd decade |
COD |
RPE disturbance at the macula |
most commonly reduced AF at the center of the macula surrounded by a ring of increased AF |
absent or severely reduced |
reduced rod b-wave amplitude with low intensity stimulus; often higher than normal with high flash energies, cone responses severely reduced |
RPGR (X-linked) |
5th decade |
COD |
macular atrophy |
perifoveal ring of increased AF |
absent or severely reduced |
reduced cone responses |
RPGR (X-linked) |
2nd-4th decade |
CORD |
range from mild macular RPE disturbance to extensive atrophy and hyperpigmentation |
reduced macular AF, reduced AF surrounded by a ring of increased AF |
absent or severely reduced |
reduced cone and rod responses |
AF, autofluorescence; ERG, full-field electroretinogram; PERG, pattern electroretinogram; RPE, retinal pigment epithelium;
* , ERG is usually nonrecordable by the fourth decade of life;
• , attenuation of retinal blood vessels and peripheral retinal atrophy can occasionally be observed. |
See text for detailed description of AF findings. In CORD, ERG reveals greater reductions in cone than in rod responses, AD, autosomal dominant, AR, autosomal recessive. |