Chapter 15 Function and Anatomy of the Mammalian Retina
![]() For additional online content visit http://www.expertconsult.com
For additional online content visit http://www.expertconsult.com
The vertebrate retina forms a thin sheet of neural tissue at the back of the eye that converts light to an electrical signal. The neural retina is approximately 100–200 µm thick, depending on the species, and represents a triumph of miniaturization.1 The retina is a spatial information processor built upon a mosaic of rod and cone photoreceptors, which are the light-responsive elements that initiate signaling using graded electrical signals.2 The spatial information content is preserved through the lateral geniculate nucleus, retinotopically mapped on to area V1 (area 17) of the primary visual cortex and from there on to a number of higher visual processing areas. There is considerable processing both within the retina as well as in higher brain structures and we interpret these electrical signals as vision. While vision is an analog system, a rough approximation to a digital system would result in a resolution in excess of 500 megapixels. Although initially conceived to be a simple model for the brain, more probably, the retina approaches limits imposed by metabolism, blood flow, and diffusion. This pressure to pack more function into a small volume of neural tissue leads to increased complexity.
Visual illusions and multiple channels
At first glance, Figure 15.1 looks like a random array of large pixels with three bright areas. Now, screw up your eyes, glance aside or look at it from the other side of the room, and suddenly you may recognize da Vinci’s Mona Lisa. The bright areas are the face, chest, and hands. But why does the image seem to be more recognizable when visual input is distorted? The high-spatial-frequency components have been removed by greatly reducing the pixel number. What detail does remain is a confusing array of boxes. However, this detailed view can be blurred by squinting, which removes the high-frequency components, leaving an easily recognized low-acuity version. So this is a direct demonstration that the visual system operates on at least two channels of information at different spatial scales simultaneously. In fact, there is empirical evidence that vision is composed of at least 15 parallel channels or streams of information that are transmitted simultaneously throughout the visual system. It is the purpose of this chapter to describe the functional anatomy of the retina that leads to the formation of some of these independent channels of visual information.
The retina is a piece of brain
Like the rest of the central nervous system (CNS), the retina is embryologically derived from the neural tube. It is formed from the same components as the rest of the CNS and like all other sensory systems has specialized structures, photoreceptors that transduce environmental energy into electrical potentials. As such, one can divide the retina in two parts3: (1) the sensory retina, concerned with phototransduction of light by rod and cone photoreceptors; and (2) the neural retina, consisting of more typical interneurons (bipolar, horizontal, and amacrine cells) and projection neurons (ganglion cells) that carry out the first steps in processing visual information.
The retina has been characterized by Dowling as an approachable part of the brain,4 because it is a ready-made brain slice with few barriers to the penetration of drugs or antibodies. In addition, its natural stimulus, light, is easily controlled and the same stimuli can be presented either to the intact animal or to the retina removed from the eye and placed in vitro. This chapter focuses on processing in the mammalian retina. However, many of the pioneering studies in retinal function were initially performed in fish and amphibian retina. In particular, the salamander retina has been a long-standing model because its large cells enhance the ease of electrophysiological recording.
Neuronal communication: chemical and electrical
Many neurons also are directly connected via electrical synapses known as gap junctions.5–9 Gap junctions are named after the narrow gap formed by docked hemichannels, or connexons aligned on either side of two cell membranes. Each hemichannel is built from six connexins that surround a central pore, forming an intercellular channel, which passes ions and small molecules (≤1 kDa). Gap junctions are not static pores; they are modulated by light and contribute to neural processing.
The retina is a layered structure
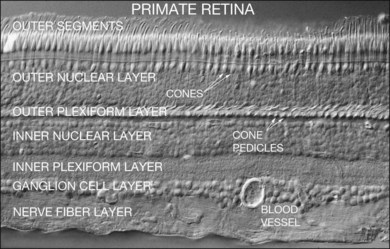
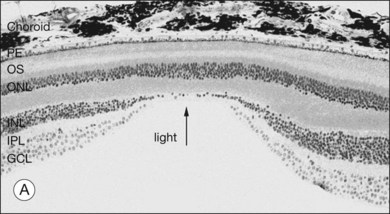
The retina is a beautifully layered structure in which three layers of neurons can be visualized without staining (Fig. 15.2). The outer nuclear layer (ONL) contains the cell bodies of photoreceptors, both rods and cones. The inner nuclear layer (INL) contains the cell bodies of horizontal, bipolar, amacrine and radial glial (or Müller) cells. The ganglion cell layer (GCL) contains displaced amacrine and ganglion cells. Ganglion cells are the projection neurons of the retina: their axons form the optic nerve and project to a variety of subcortical nuclei. The three nuclear layers are separated by two synaptic (plexiform) layers that contain the dendrites and synapses. The outer plexiform layer (OPL) lies between the ONL and the INL. This is where the photoreceptors, horizontal and bipolar cell dendrites interact. The inner plexiform layer (IPL) separates the INL and the GCL and this is where the bipolar cell axons, amacrine, and ganglion cells interact. When we speak of the retina, outer or distal refers to the scleral side of the retina and inner or proximal refers to the vitreal side of the retina.
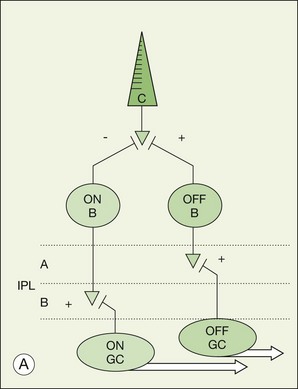
The functional stratification of the retina extends to the IPL, which is organized according to the polarity of bipolar cell inputs.10 Since the time of Cajal, the IPL has been divided into five layers: 1 and 2 for sublamina a and layers 3–5 for sublamina b. In mammalian species, there are 9–11 morphological types of bipolar cell, plus the rod bipolar cell, which terminate at different depths in the IPL.11–13 The polarity of a bipolar cell response is determined by the differential expression of postsynaptic glutamate receptors on their dendrites. The dendrites of OFF cone bipolar cells carry α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA)/kainate receptors and they ramify in sublamina a of the IPL.14 In contrast, ON cone bipolar cells and rod bipolar cells express mGluR6 receptors and their axons ramify in sublamina b.15–17 The separation of ON and OFF pathways appears to be a fundamental principle of retinal organization, which is reflected throughout the visual system.18,19
There is a wealth of evidence to support the functional division of the IPL into ON and OFF sublaminae. For example, cholinergic amacrine cells (ChACs), also known as starburst amacrine cells on account of their unique morphology, are present as mirror-image pairs.20 The conventionally placed ChACs have somas in the innermost layer of the INL. They have OFF responses to light stimulation and ramify in sublamina a. In contrast, the displaced ChACs reside in the GCL, produce ON responses, and ramify in sublamina b. Likewise, alpha ganglion cells are present as paramorphic pairs such that the dendritic trees of OFF alpha ganglion cells are stratified in sublamina a to receive input from OFF bipolar cells while ON alpha ganglion cells ramify in sublamina b to make contact with ON bipolar cells.21,22 In primate retina, both midget and parasol ganglion cells are present as paramorphic pairs, which conform to the stratification rules of the IPL. ON/OFF directionally selective (DS) ganglion cells produce both ON and OFF responses of short latency, indicating direct input, and they are bistratified with dendrites in sublaminae 2 and 4, coincident with the cholinergic bands.
The ON and OFF stratification of the IPL is a fundamental tenet of retinal organization which applies to many, if not most, cell types. However, several exceptions to this rule have now been identified. For example, the dopaminergic amacrine cells (DACs) stratify predominantly in sublamina a, the OFF layer, yet they apparently produce ON responses to light.23,24 Likewise, the intrinsically photosensitive retinal ganglion cells (ipRGCs), which stratify in sublamina a of the primate retina, were all ON cells.25 These cell types, among others, receive input from ON bipolar axons as they traverse sublamina a of the IPL. These axonal ribbons provide a set of inputs that break the stratification rules of the IPL. Thus, there is an additional accessory ON sublayer in the outer portion of the IPL26,27 (see sections on DACs and ipRGCs) (Table 15.1).
Table 15.1 Cellular and synaptic layers of the retina
| Layer | Contains: |
|---|---|
| Outer nuclear layer (ONL) | Photoreceptors, rods, and cones |
| Inner nuclear layer (INL) | Horizontal cells, bipolar cells, amacrine cells, Müller cells |
| Ganglion cell layer (GCL) | Ganglion cells, displaced amacrine cells |
| Outer plexiform layer (OPL) | Photoreceptors talk to horizontal cells and bipolar cells |
| Inner plexiform layer (IPL) | Bipolar cells talk to amacrine cells and ganglion cells |
Gross retinal morphology
The fovea
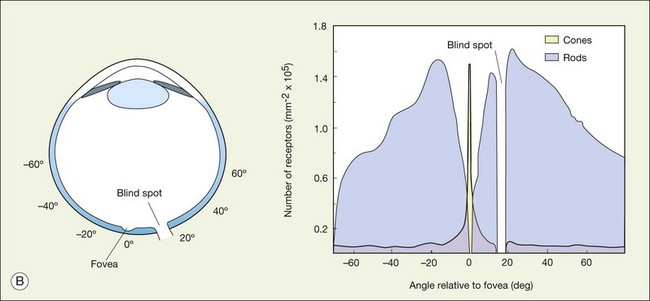
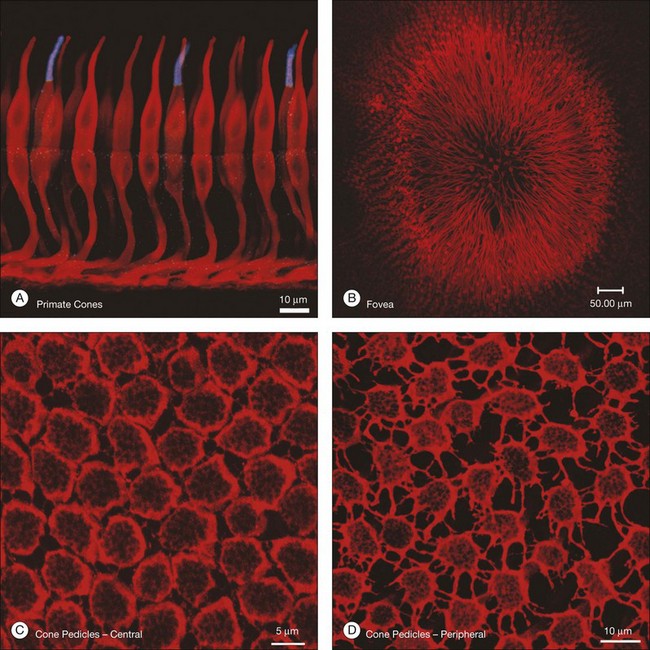
When a peripheral visual stimulus gets our attention, we automatically focus the retinal image in the center of our gaze. In the primate retina this central region of highest visual acuity is known as the fovea. Here there is a depression in the retina, known as the foveal pit, where cone photoreceptor density is highest and there are no rods. The pit results from the lack of overlying neurons (Fig. 15.3). Instead, cone axons, Henle fibers, run obliquely to cone pedicles away from the fovea where the bipolar and ganglion cells connected to the central cones are “piled up” in an annular zone with the GCL 6–8 cells thick. The foveal structure is thought to maximize sensitivity because light cannot be scattered by passing through other retinal layers. It also optimizes acuity by packing the maximum number of cones and reducing their size. In human retina, the peak cone density approaches 200 000/mm2 and the ONL is slightly thicker to accommodate these extra cells.28 There are no blood vessels in the fovea and in the central fovea there are no blue cones. The low density of blue cones lowers their acuity,29 to match the blurring caused by chromatic aberration in the lens.30 Other mammals have an area centralis (cat) or a visual streak (rabbit, swine) with similar high cell density but these structures lack the central depression. A consequence of the exclusion of rods from the fovea is that in dark-adapted conditions, say looking for a dim star, it is necessary to look slightly off the visual axis to focus the image in the region of high rod density.
The blind spot and how to find it
There are no photoreceptors where the optic nerve exits the eye and so any image that falls on this region cannot be processed by the retina. Curiously, we do not perceive a hole in the visual scene because the visual system fills in. To demonstrate the blind spot, hold the page level at normal reading distance. Look at the O in Figure 15.4, and then close your right eye. The X on the left should disappear because the image of the cross falls on the optic nerve head. You can reverse this demonstration: look at the X, then close the left eye to make the O disappear. Remember, the blind spot is nasal to the fovea. The light rays cross over at the lens so the blind spot is lateral to the point of focus.
Painting the retina – techniques to label and visualize retinal neurons
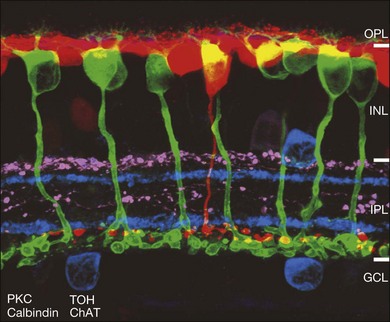
The major landmarks of the retina, two synaptic layers sandwiched between three nuclear layers, are obvious even in the unstained section of macaque retina shown in Figure 15.2. In fact, there is an immense amount of detail present in this image. For example, individual cones can be seen with their descending axons terminating in a row of cone pedicles in the OPL. However, the somas of all the other retinal neurons are indistinguishable and the details of their structures cannot be visualized. One goal of vision scientists is to have a complete map of all the interconnections between the retinal neurons so that the functional outputs can be fully understood. To uncover the hidden pathways and circuits through the retina, we need to stain specific cell types selectively (Fig. 15.5), so that we can differentiate between rod and cone pathways, for example. Furthermore, it is very useful to visualize several cell types at once so that the number and position of one cell type relative to another can be determined. This can be achieved by confocal microscopy, which permits the simultaneous acquisition of three or more different labels in addition to providing improved resolution and three-dimensional visualization (Fig. 15.5). With care, confocal microscopy can be used to visualize both chemical and electrical synapses in the retina.31 Two-photon confocal microscopy permits imaging using an infrared laser, which does not stimulate the visual pigments. It has been used to measure changes in intracellular calcium concentration of single neurons or dendrites in response to stimulation with visible light.32,33 One disadvantage of these methods is that individual synapses cannot easily be visualized because of the inherent resolution limits of confocal microscopy. However, high-throughput electron microscopy methods have been developed that allow three-dimensional reconstruction of blocks of retina, albeit in fixed tissue. By careful analysis and multiple staining approaches it is possible to identify all the synapses and cells to which an individual neuron makes connections.34
The mainstay of structure, function, and morphology studies is immunocytochemical methods, which can be used to stain structural components, enzymes, neurotransmitters, synaptic proteins, and postsynaptic receptors (Fig. 15.5). Specific primary antibodies are currently available for many of these components or they can be generated. Secondary antibodies conjugated to an ever-increasing variety of fluorochromes are readily available as standard reagents and allow for multiple staining. Single neurons can also be filled with fluorescent dyes via glass microelectrodes. Diffusible tracers such as Neurobiotin can be used to label a network of coupled cells.5 The optic nerve can be back-filled with fluorescent dyes, which in one useful variant are concentrated in vacuoles that explode when excited to release dye, which diffuses throughout the dendritic structure of individual ganglion cells.35 Certain ganglion cell types can be selectively labeled by stereotactically injecting a central target with retrograde tracers.36 Another method to label single cells involves labeling the whole retina with dihydrorhodamine, which can then be photoconverted to a fluorescent product in selected cells by illumination.37,38 Individual neurons of all types can be randomly labeled with ballistic particles coated with fluorescent dyes or DNA to synthesize a specific marker.39 Finally, there is increasing use of transgenic animals such as mice engineered to express green fluorescent protein (GFP) variants under the control of a specific promoter.40
Six major neuronal cell classes
The retina contains six major neuronal classes. Photoreceptors are located in the ONL and can be subdivided into rods and cones. Bipolar cells take the signals from photoreceptors and transmit them to the inner retina. Horizontal cells and amacrine cells are laterally extensive interneurons in the outer and inner retina, respectively. Ganglion cells receive input from bipolar and amacrine cells and form the output from the retina. In addition, interplexiform cells share many properties with amacrine cells but project back to the outer retina. Finally, the radially oriented Müller cells are the predominant glial cells (Table 15.2).
Table 15.2 Major cell types in the retina
| Neuronal types | Role | Types |
|---|---|---|
| Photoreceptors | Rod and cones | 2 |
| Horizontal cells | Lateral interneurons, OPL | 2 |
| Bipolar cells | Vertical connection | 10–12 |
| Amacrine cells | Lateral interneurons, IPL | ~30 |
| Ganglion cells | Output neurons | ~20 |
| Interplexiform cells | Feedback, IPL to OPL | ? |
| Total | ~65 |
OPL, outer plexiform layer; IPL, inner plexiform layer.
Classification of retinal cells
Although the retina contains six major cell classes, these may be further divided into many distinct subtypes for a total of about 65 different neurons. Thus, the retina is certainly complex, but it is not chaotic. Cells of a given type form nonrandom mosaics across the retina.41 A single type shares essential morphological features and uses the same neurotransmitter(s). Furthermore, they ramify at a characteristic depth in the retina and make stereotyped synaptic connections, including the same type and number of postsynaptic cells, and even the same number of synapses.42,43 As we learn more about neuronal circuits and the functional anatomy we see repeated patterns across the retina, and we are reminded that it is a two-dimensional array of recurring circuits.
The classification of retinal neurons, which dates back to the work of Cajal more than a century ago, is not a simple matter.44 However, it is important that we are at least aware of all the major cell types in the retina. Even a basic understanding of a circuit cannot be achieved if critical parts are missing. Classification is complicated by the large number of different cell types, the low probability of obtaining rare types, variation in size with eccentricity, and the normal morphological variation within a cell type. While the staining and sampling issues have largely been resolved by modern methods, the question of what constitutes a separate cell type, as opposed to subtle differences between examples from the same type, is still a problem. The resolution lies partly in understanding the significance of some morphological features and partly in considering more variables, i.e., independent criteria to classify a specific cell type. Finally, as a kind of parity check, it is necessary to consider the properties of a whole population for a given cell type to see if it is consistent.
Neurons may be classified into unique cell types on the basis of several properties:
1. their morphology, meaning the size, shape, and structure of a neuron, especially its dendritic field. This may include measures of dendritic density and branching patterns
2. the depth within the retina, particularly the level in the IPL where its dendrites or axons stratify. We have come to realize that depth in the IPL functions as a simple addressing system to determine which target neurons are in synaptic contact
3. their electrophysiology, particularly their excitatory and inhibitory inputs
4. their biochemistry, particularly with regard to the different neurotransmitters used to communicate between neurons and structural proteins or receptors
5. the pattern of connections with other neurons, which should be consistent for a unique cell type
6. finally, the properties of the whole population for a specific cell type.
A group of cells that are all the same type have certain special properties. The retina is a two-dimensional array and most cell types tile the retina in a consistent manner so that an even coverage is obtained. Cells of the same type are usually not adjacent. The position of the cells relative to others of the same class is also an indicator of cell type because unique populations form nonrandom mosaics.41 Consider a handful of marbles dropped on the floor. Some will stop close by but others may roll into a corner. If a measure is taken between the nearest neighbors, it will be found to have a high variation. In contrast, for a nonrandom mosaic of neurons, the spacing is controlled such that the distance to the nearest cell of the same type is relatively constant and the variance of this measure is low. The ratio of mean nearest neighbor distance to the variance gives an index of regularity for a cell population. We would expect the ratio to be low for the randomly scattered marbles, but indices of three or higher are common for the typical nonrandom, orderly mosaics of retinal neurons. The most satisfying results for classification come from the convergence of these methods to indicate the function of a retinal cell in the process of vision.
Photoreceptors
Cones
The mammalian retina contains two types of photoreceptor, rods and cones.45 Rods account for 95% of all photoreceptors. They are numerous, with slender outer segments, densely packed and specialized for high sensitivity under dark or starlight conditions. Cones are larger, with tapering outer segments, and they are found in the top row of the ONL (see Fig. 15.3). Cones make up only ~5% of photoreceptors28,45 but they provide high-acuity color vision in daylight conditions when photons are abundant. This versatile combination of rods and cones and their associated circuits covers an intensity range of around 10 log units from the darkest night to bright sunlight.46 While the average visual scene has a range of intensities covering 2–3 log units, the continual adaptation of retinal sensitivity slides this operating range through the entire range of light intensities. This is a critical function of the retina because outside the normal operating range we are functionally blind. Common examples include the inability to see momentarily when entering a dark cinema or driving into the setting sun. Much of the adaptation takes place in photoreceptors but, as we shall see below, this is accompanied by major changes in the neural pathways through the retina. This is arguably the most important function of the retina, after light detection itself.
There are approximately 5 million cones in the human retina and 190 000 in the mouse retina.47,48 Cones make up approximately 5% of the total photoreceptors in humans, compared to 2.8% in the mouse,49 so we are all rod-dominated in terms of absolute numbers. One exception is the ground squirrel, which is truly cone-dominated. Importantly, cones are not evenly distributed. In human retina, there is a massive peak at the fovea (Fig. 15.3) where the density reaches around 200 000/mm2, approximately 100 times the density in the periphery.28,47 This is the region of maximum acuity, although the peak density slightly exceeds experimentally measured visual acuity due to blurring by the optics of the eye.50–52 Where the ganglion cell axons gather to form the optic nerve there are no photoreceptors and their absence from this location is the cause of the blind spot (Fig. 15.3).
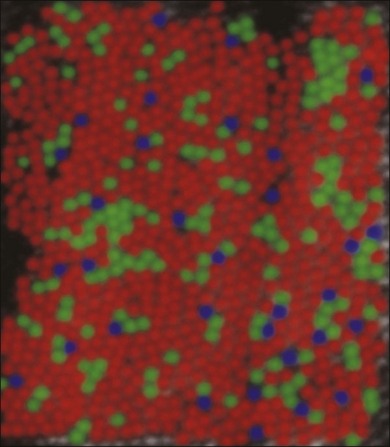
Cones support color vision and, in old-world primates and humans, there are three classes: blue, green and red. They are maximally sensitive to 430, 530, and 561 nm light, respectively.53–55 Other mammals, including cats, rabbits, and rodents, have an evolutionary ancient form of color vision based on green and blue cones only. The presence of red and green cone opsins in a tandem array on the X chromosome is thought to be due to a recent gene duplication and underlies the preponderance of color blindness among males.56 Using adaptive optics to correct for blurring in the lens and cornea, the distribution of red, green, and blue cones can be mapped in the living human eye.57 Surprisingly, the distribution of cones was random and clumpy (Fig. 15.6). In addition, there appears to be enormous variation in the red/green cone ratio among individuals with normal color vision.
Blue cones are present as a minority: they make up approximately 10% of the cone population (Fig. 15.6). This is not enough to support high acuity but calculations show that the blue cone density is sufficient to support the reduced resolution caused by visual aberration at the blue end of the spectrum.30 At the very center of the fovea, blue cones are absent.29,58 The fact that this is not readily apparent is due to the relatively poor spatial acuity of color vision compared to the luminance-driven pathways.
Red and green cones cannot be differentiated morphologically and the red and green cone opsins are so closely related that they are also indistinguishable. However, blue cones can be mapped with a selective antibody against blue cone opsin29,59 or by their selective accumulation of certain dyes.58
Rods
The ONL contains photoreceptor cell bodies, both rods and cones.45 Even in the cone-dominated human retina, rods far outnumber cones, by a factor of 20 : 1, so they account for most of the ONL except at the fovea. The human retina contains approximately 100 million rods, and they pool signals to provide high sensitivity for dark-adapted vision, say starlight, which appears monochromatic. A lack of color vision is the hallmark of rod-mediated vision. Rods are absent within 350 µm of the fovea but reach a peak density in an annular region at about 20° eccentricity (Fig. 15.3). This does not match the area of maximum scotopic acuity, which occurs around 5°,60 so it has been suggested that another component of the rod pathway, such as the AII amacrine cells, present at a much lower density, forms a bottleneck to limit acuity61,62 (see below).
Cone pedicles and rod spherules
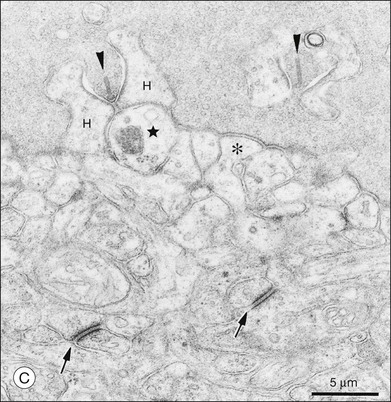
Cones and rods make contact with bipolar and horizontal cells in the OPL. Cone axons descend through the massed ranks of rod somas in the ONL to terminate in a two-dimensional array of cone pedicles at the OPL (Fig. 15.7). Near the fovea in primate retinas, the cone axons are splayed out radially so that the pedicles form an annular array around the center (Fig. 15.7B). Cones form two specialized contacts, ribbon synapses and flat contacts, with postsynaptic neurons. Ribbon synapses are so named because of electron-dense structures at invaginations where they contact depolarizing (or ON) bipolar and horizontal cells (Fig. 15.8). Cone pedicles also make flat contacts along the base of the pedicle with hyperpolarizing (or OFF) bipolar cells. Rod axons also descend to form synapses with a single type of depolarizing bipolar cell as well as horizontal cells. The terminals of rods are called spherules, and, similar to cones, they use a ribbon synapse. In contrast to cones, there is only a single invagination in each, containing two rod bipolar and two horizontal cell dendrites.
Each cone pedicle is 6–8 µm in diameter and, near the fovea, contains 20 synaptic ribbons and, in the periphery, around 40.63 Cones release glutamate constantly in the dark and the synaptic ribbons are thought to support this high rate of release. As we will describe further below, the multiple ribbon and flat synapses on each cone pedicle form connections with many different types of bipolar cell.
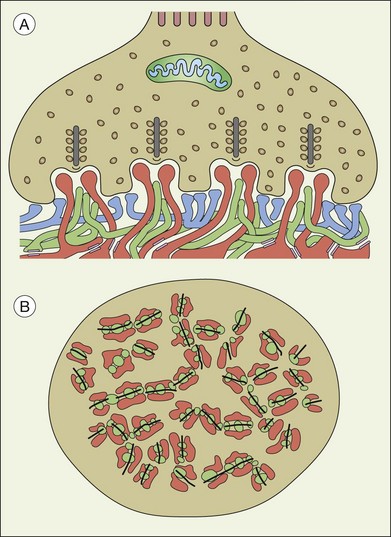
A sense of the complexity of these synapses can be obtained by labeling some of the individual components. Antibodies reactive against a vesicle protein such as synaptophysin provide a way to label photoreceptor terminals in the OPL, because photoreceptor axon terminals are filled with vesicles. To visualize the synaptic ribbons, antibodies to kinesin II can be used. Finally, antibodies to the mGluR6 receptor, which is located on the dendritic tip of the depolarizing bipolar cells, mark one of the postsynaptic processes. When visualizing just the cone pedicle, there are a number of indentations or invaginations (Fig. 15.8). Within each invagination, horizontal cell dendrites are lateral and high, approaching the synaptic ribbons. ON cone bipolar cell processes are central but slightly lower at the synaptic ribbon. In contrast, OFF bipolar dendrites form basal synapses with the cone pedicle at a distinctly lower level. Staining a piece of retina for synaptophysin, kinesin II, and mGluR6, begins to show the complexity of the structures (Fig. 15.9). Rod spherules contain a single large horseshoe-shaped ribbon with two bipolar cell dendrites nestled within. The cones have numerous and slightly smaller ribbons with each also nestled against an mGluR6-labeled dendrite. What is not visible are the horizontal cell dendrites that also invaginate, but are slightly lower than the bipolar cells.
Around 10–12 cone bipolar cells plus multiple horizontal cell dendrites contact each cone, so under the pedicle there may be as many as 200 postsynaptic processes.64 In fact, the cone pedicle may be the most complex synaptic structure in the brain. The distribution of postsynaptic processes under the cone pedicle is laminated in a stereotyped fashion (Fig. 15.8).63,64
The fundamental problem for rods is to detect a small brief hyperpolarization due to a single photon against a noisy background of thermal noise and the stochastic nature of transmitter release.65,66 One strategy is to minimize background variation by maintaining a high sustained-release rate in the dark. The synaptic terminal of a rod, or rod spherule, is about 2 µm in diameter and contains a very large synaptic ribbon (Fig. 15.9), a specialization thought to be associated with a high rate of transmitter release. The spherule is packed with synaptic vesicles, containing the neurotransmitter glutamate, to support sustained transmitter release. There is a single invagination (imagine a closed fist and insert a finger between thumb and forefinger to make a pocket) containing a tetrad of processes, two from horizontal cells and two rod bipolar dendrites.67 This structure brings the postsynaptic processes close to the release site and may prevent spillover to adjacent rods. These anatomical specializations appear to reduce variation in rod signaling so that small single-photon responses can be detected with high reliability.
Photoreceptor coupling
While the synapses between photoreceptors and second-order neurons are extremely complex, there are still additional connections between photoreceptors. These take the form of electrical coupling, mediated by gap junctions. Close to the fovea, the cone pedicles are densely packed, almost touching, and connected by very fine processes known as telodendria (Fig. 15.7C).45 Rod terminals are either absent or displaced slightly higher, towards the outer segments, in this region. More peripherally, the cone pedicles are widely spaced and the telodendria are much more prominent (Fig. 15.7D). The contact points between telodendria of neighboring cones are the sites of connexin (Cx)36 gap junctions, which mediate electrical coupling between cones.68,69 Recordings between neighboring cones also show that they are coupled.68,70 This may seem puzzling at first because it should lead to a loss of acuity and blurring. However, the cone array actually oversamples the signal so this leads to little or no loss. Instead, the coupling is thought to reduce noise, which is random, while the light-driven signals, which are correlated between close neighbors, will be reinforcing. It has been calculated that this may improve the signal-to-noise ratio by 77% – a large gain for little loss.71 Coupling between red/green cones is indiscriminate, which may reflect the close absorption curves of red and green cones.70 Morphologically, blue cone pedicles are slightly smaller than those of red/green cones and they have only a few withered dendrites, which rarely touch neighboring cones.72 Hence, blue cones are not coupled into the red/green network.68,70 This may serve to preserve spectral information in the color pathways.
There are also gap junctions between cone pedicles and rods.73 These allow rod signals to enter cones, and rod-mediated signals can be detected in second-order neurons that are thought to be connected exclusively to cones.74 Responses with the signature of rod origin have also been recorded in primate cones.75 Because rods far outnumber cones, the influence of many rods on a single cone may be substantial. It is now thought that rod–cone coupling forms an alternative rod-driven pathway that may be important at intermediate light intensities, in the mesopic range.76,77 This influence may relate to the enhancement of cone electroretinogram (ERG) amplitudes in humans and mice by light adaptation, where the cone ERG gradually increases in amplitude following the onset of a steady adapting field.78–81 Blocking gap junctions inhibits this effect in the mouse retina. In the human, however, the adaptation dependence of these changes has a photopic (cone) signature. Rod–cone coupling also is very dynamic and influenced by circadian rhythms, increasing at night and decreasing during the day.82
Photoreceptors release glutamate in the dark
The first intracellular recordings from cones were surprising because they showed that photoreceptors hyperpolarize in response to light.4 This means they are relatively depolarized and release their neurotransmitter, glutamate, in the dark. From the viewpoint of signal information, the sign of the photoreceptor response makes no logical difference; photoreceptors produce graded responses modulated around the mean light level. When a photon is absorbed, the visual pigment is activated and then a cascade of other biochemical events is triggered.83 This is known as phototransduction and it leads to the closure of a cGMP-gated cation channel in the cell membrane to produce hyperpolarization and a reduction in the release of glutamate.84 The postsynaptic responses to light are in fact due to a decrease of glutamate release. Thus, glutamate uptake also plays a key role in retinal neurotransmission because the clearance of glutamate must be rapid to provide a fast postsynaptic response to light.85 The hyperpolarizing light response of photoreceptors is now well established and it is supported by studies of vesicle turnover, which is much higher in the dark.86 Furthermore, synaptic blocking studies produce the appropriate responses in the different second-order neurons and there is a stereotyped array of distinct glutamate receptors associated with specific cell types. Glutamate, with its library of postsynaptic receptors, seems particularly suitable to orchestrate the large variety of postsynaptic responses at the cone pedicle.
Rods operate in a manner essentially similar to cones: they hyperpolarize in response to light increases, albeit there are many molecular differences. However, everything about the rod pathway in the retina is designed for maximum sensitivity. This is reflected in the anatomical details and synaptic connections of rods.66 Rods can respond to single photons, obviously the design limit, with a binary (all or nothing) signal, but visual threshold requires a signal in 5–10 rods. Depending on the species, about 20–100 rods converge on to a single rod bipolar cell87,88 and this high convergence also contributes to the high sensitivity of the rod pathway. If 100 rods converge to a single rod bipolar cell and 100 rod bipolar cells converge to a ganglion cell, then the absolute threshold for vision is determined by the product, approximately 1 photoisomerization per 10 000 rods.
Second-order neurons: horizontal and bipolar cells
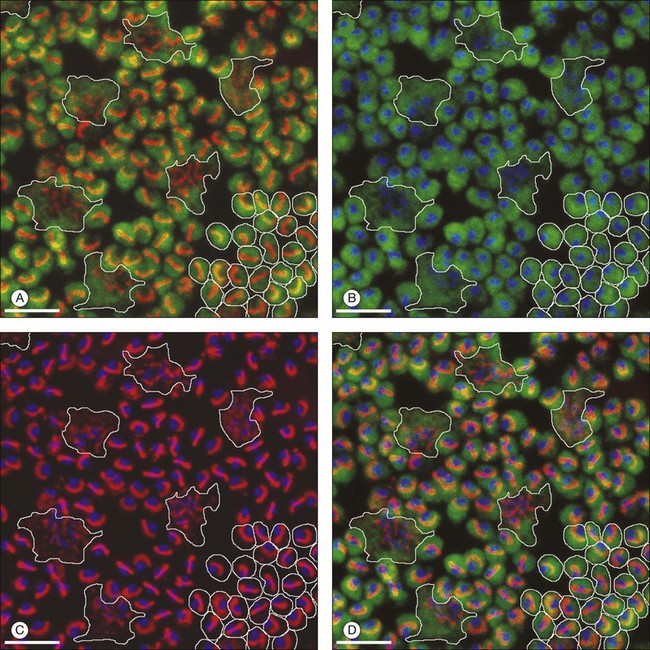
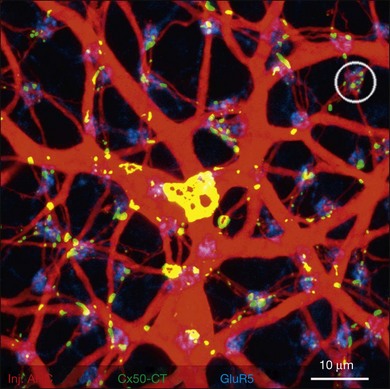
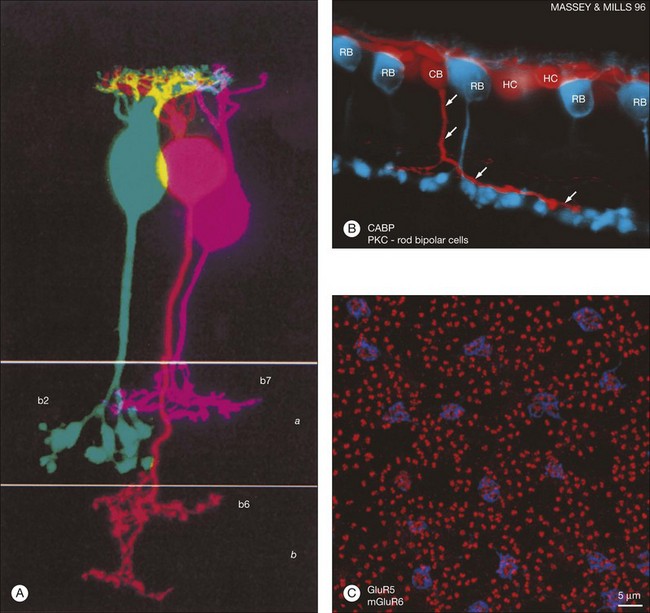
Rods and cones make synaptic connections with bipolar and horizontal cells. Horizontal cells are laterally extensive interneurons located in the outer row of the INL, adjacent to the OPL. They respond to diffuse light with a large hyperpolarization. This is the same as cones so we say that the input is sign-conserving. It appears to be mediated by excitatory glutamate receptors of the AMPA subtype, which have been located on horizontal cell dendrites.89,90 In most mammals there are two morphological types of horizontal cell, but only one in rodents.91–94 In all species, horizontal cells are extensively coupled, which dramatically increases the size of the receptive field. In Figure 15.10 the entire A-type horizontal cell network in the rabbit retina has been labeled by injecting several cells with a diffusible tracer, Neurobiotin, which readily passes through gap junctions.95 A-type horizontal cells are axonless and have a large irregular shape, giving rise to many fine terminals which contact every cone pedicle within the dendritic field. A high-resolution image shows how fine horizontal cell dendrites converge at cone terminals while ignoring the numerous rod spherules (Fig. 15.10). The cone terminals were marked by the labeling of two different glutamate receptors, GluR5, which marks the basal contacts of certain OFF bipolar cells,96 and mGluR6, which is expressed by ON cone bipolar cells17,97 (see below). It should be noted that the three labels are nonoverlapping at the cone terminal, consistent with the presence of three separate neurons, horizontal cells, ON cone bipolar cells, and OFF cone bipolar cells, which all converge independently at the cone pedicles (Fig. 15.10).64
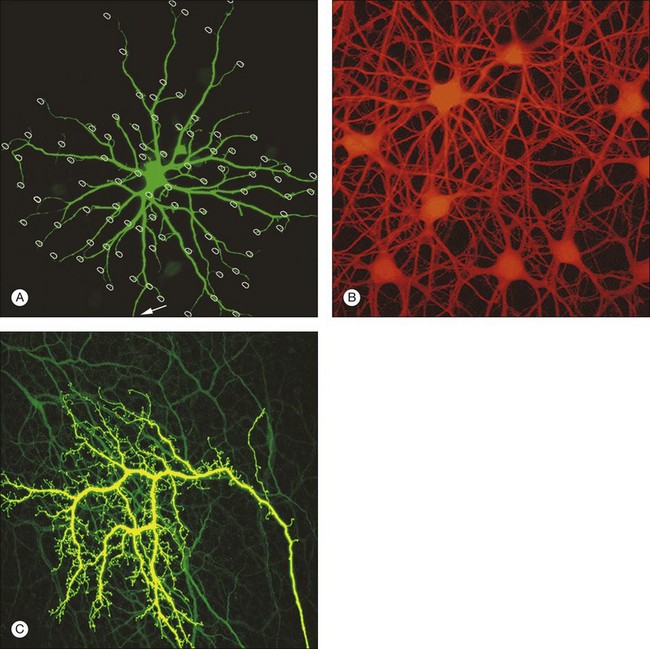
B-type horizontal cells in the rabbit retina are smaller and radially symmetrical (Fig. 15.11). While the somatic dendrites of the B-type horizontal cell also contact cones, each cell gives rise to a long axon that meanders randomly before branching into an elaborate terminal structure (Fig. 15.11C).95 The electrotonic length of the axon apparently isolates the somatic region from the axon terminal whose branches contact rods instead of cones.98 B-type horizontal cells are also coupled by gap junctions and so are the axon terminals (Fig. 15.11B).95,99–101 For both types of horizontal cell, as the cell density falls with eccentricity, the dendritic field size increases so that an even coverage of 6–8 is maintained across the retina. B-type horizontal cells are always smaller and more numerous – two to three times the A-type density.95
In primates, there are also two kinds of horizontal cell, both axon-bearing, but the H2 only contacts cones and the axon is not well developed.102 H1 is a large, well-coupled cell that contacts all red/green cones but not blue cones.103 H2 has a smaller soma and finer dendrites that make sparse contacts with red/green cones but densely innervate blue cone pedicles.103 Recording from both horizontal cell types shows that they receive sign-conserving inputs from both red and green cones (plus blue for H2).103 The wiring of the H2 horizontal cell suggests it plays a role in blue/yellow (red +green) processing.
In central retina, there are four times as many H1s as H2s, decreasing to twice as many in peripheral retina.104 Increasing size with eccentricity compensates for decreasing density so coverage for both types is 3–5 evenly across most of the retina. The peak density for H1 horizontal cells reaches about 18 000/mm2 near the fovea. This is an order of magnitude higher than rabbit retina. Packer and Dacey105 have recorded from many primate H1 cells and they make the interesting observation that central cells are not only smaller but less coupled, perhaps because they overlap less. Thus, the H1 receptive field in the fovea may be small enough, 20–30 µm, to match the receptive field surround of midget ganglion cells.
Horizontal cells are extensively coupled by gap junctions and their molecular identity has been determined for many species. In the rabbit retina, the gap junctions between A-type horizontal cells are labeled with an antibody against Cx50 at many contact points in the matrix (Fig. 15.12).106 Some of the gap junction plaques are very large and the unitary conductance of Cx50 channels is also high. This explains why A-type horizontal cells form an electrical syncytium. B-type horizontal cells are not labeled for Cx50. These gap junctions have different properties so they are likely to be assembled from different connexins. In rodents, there is only one type of axon-bearing horizontal cell.94 In a Cx57 knockout mouse, these cells are no longer coupled.107 This suggests that multiple neuronal connexins are present in the retina and that Cx57 gap junctions may be responsible for coupling in axon-bearing horizontal cells. The situation in the primate retina is still unknown but future progress should make it possible to test theories concerning horizontal cell coupling.
Horizontal cell function
As pointed out by Sterling, reading high-contrast images in bright artificial light is one thing,108 but step into the outside world of the naturalist and visual objects often have very low contrast from the background. What we need is a way to subtract the common background so we can focus on the small, low-contrast details in the image. At least in part, this role is accomplished by horizontal cells in the outer retina. The horizontal cell network holds a big slow picture of the visual scene that is subtracted by feedback to cones. Because horizontal cells have a large receptive field, due to their size and coupling, they subtract the mean illumination, allowing the photoreceptors to respond to local changes in visual illumination. Because they are slow, horizontal cells always lag behind and damp the visual signal without blocking the rapidly changing peaks that carry the most important visual information. In one in silico model of the retina, the bipolar cells amplify the difference between the cone signal and the horizontal cell network.109 Horizontal cells also may have other specific roles in vision. Recent studies have led to the hypothesis that they are responsible for red-green opponency in the primate midget ganglion cells.110
There is general consensus that horizontal cells provide a negative-feedback signal to cones and rods. When a light flash hyperpolarizes the photoreceptors and the horizontal cells, the horizontal cell input results in sign-inverted voltage change in rods.111 This negative feedback results in an antagonistic center surround receptive field in the photoreceptors. The mechanism is thought to consist of a shift in the voltage sensitivity of the calcium channels in the photoreceptors, which results in a leftward shift of the calcium channels I/V relationship and increased currents. Increased currents in photoreceptors inhibit the light-mediated hyperpolarization and alter glutamate release.
The mechanism that mediates feedback remains under considerable debate. Three models have been proposed as the basis of negative feedback at this synapse. The first proposed that horizontal cells release GABA to hyperpolarize the cone photoreceptor.112 There are data to support this idea. GABA has been found in some mammalian horizontal cells,113,114 as has VGAT, the vesicular GABA transporter,115 and there is mounting evidence that many components required for GABA release also are present.116–118 In addition, GABA receptors are located on bipolar cell dendrites119 or perhaps cone pedicles themselves,120 providing another mechanism for feedback. However, GABA antagonists do not block surround antagonism either in photoreceptors121 or at the level of the ganglion cell receptive field.122 An ephaptic mechanism is proposed in which a change in the transmembrane potential of the cone photoreceptor occurs via hemichannels and glutamate receptors on the horizontal cell dendrites and creates a voltage drop in the synapse; a subsequent increase in photoreceptor calcium channel activity modifies the gain in the cone synapse.123,124 Finally, a model is proposed in which horizontal cell depolarization produces an efflux of protons, which acidifies the extracellular space and inhibits cone voltage-gated calcium channels.125,126 While these discrepancies in the mechanism of negative feedback remain to be resolved, results of a recent publication suggest that a positive-feedback mechanism may also contribute to signaling at this synapse.127
Studies of this important mechanism have been undertaken primarily in species other than mouse, which has meant the analyses have required pharmacological manipulations, rather than genetic approaches. However, recently a zebrafish model, in which a specific connexin was thought to make up the horizontal cell hemichannels, was identified. In these fish feedback was reduced, providing support for the ephaptic hypothesis.128 Studies in mice also have shown that feedback is present. This model organism provides the possibility that the underlying mechanism can be dissected genetically. No doubt in the coming years the details of this feedback mechanism, which is critical to vision, will be fully characterized.
Bipolar cell function
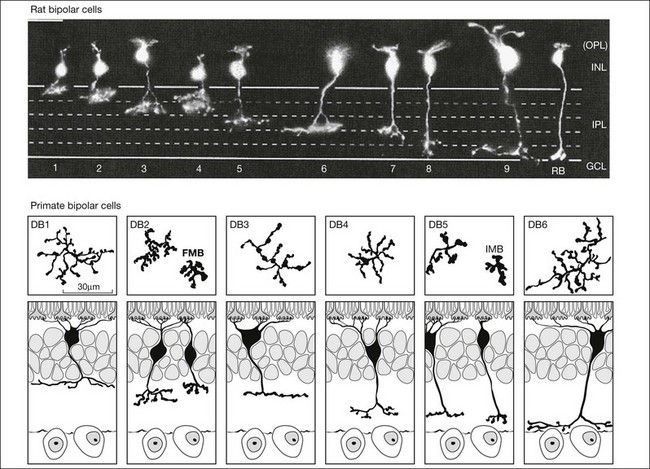
Bipolar cells receive input from photoreceptors and then conduct the visual signal to the inner retina. They are excitatory interneurons, using glutamate as a neurotransmitter, specialized for sustained transmitter release, with terminals containing synaptic ribbons, similar to those of photoreceptors, only smaller. There are approximately 9–12 kinds of cone bipolar cell in the mammalian species that have been studied thoroughly, i.e., primates, rabbits, cats, rats, and mice (Fig. 15.13).12,129–132 In contrast to the cone bipolar cells, there is only one type of rod bipolar cell, which is morphologically and physiologically distinct from the cone bipolar cells. Rod bipolar cells are numerous – three times the density of any diffuse cone bipolar type. However, there are around 10 types of cone bipolar cell so, in total, they outnumber the rod bipolar cells by a factor of three to four.
Cone bipolar cells are difficult to identify in a retinal slice preparation. Once filled with a tracer they can be distinguished by the locations of their somas in the INL, by the morphology of their dendritic and axon terminals, and by the depth of termination in the IPL. An increasing number of selective antibodies are now available to aid in the identification of specific cone bipolar cells.133–136 In addition, several transgenic reporter mouse lines that mark specific types of bipolar cells have been created.137,138
There is a major functional division in the cone pathways that arises at the first synapse and is preserved in all higher visual centers. The retina is divided into ON and OFF channels served by ON and OFF cone bipolar cells (Fig. 15.13).15 The ON pathway is optimized to detect increases in intensity and the OFF pathway decreases in intensity. Reflecting the importance of depth in the retina, these two types of bipolar cells terminate at different levels in the IPL. OFF bipolar cells ramify in the top half, sublamina a, where they synapse with OFF ganglion cells. ON bipolar cells descend further in the IPL to sublamina b, where they synapse with ON ganglion cells. Now, ON and OFF bipolar cells produce opposite signals in response to changes in light intensity. But how is this achieved if both bipolar types contact the same cones? The short answer is via different postsynaptic receptors and, indeed, glutamate produces opposing responses in ON and OFF bipolar cells15,139,140 This simple trick, dividing the cone signal into ON and OFF components, is thought to double the dynamic range of the retina. Half the bipolar cells carry signals greater than the local mean and the other half dimmer than the average.
OFF cone bipolar cells
The dendrites of OFF bipolar cells branch in the OPL and contact every cone within the dendritic field at basal synapses (Fig. 15.14). OFF bipolar cells, like cones themselves, are hyperpolarized by light. So these are sign-conserving excitatory synapses, shown by a + in Figure 15.14, served by ionotropic glutamate receptors, i.e., conventional glutamate receptors of the AMPA/kainate type. Thus, the dark release of glutamate from cones holds OFF bipolar cells at a relatively depolarized potential and the reduction of glutamate release in response to light produces a hyperpolarization in OFF bipolar cells. In this regard, the photoreceptor/OFF bipolar cell synapse is similar to the photoreceptor/horizontal cell synapse.
Recordings from cone and OFF bipolar cell pairs in ground squirrel, which has very large cones particularly suitable for recordings, has shown that three different OFF bipolar cells use three different glutamate receptors – one AMPA receptor and two kainate receptors (Fig. 15.15).141 This dovetails very well with the differential distribution of glutamate receptors at the cone pedicle complex.90,96 Furthermore, the characteristic rate at which each receptor recovers from desensitization effectively divides the cone signal into different bandwidths. The fast AMPA receptors are well suited to convey transient signals, while the slower kainate receptors may transmit sustained responses.141 All three OFF bipolar types terminate in sublamina a, as expected, but each one branches at a different depth within the OFF sublamina (Fig. 15.15). Thus, the division of the visual signal into different channels, for delivery to different addresses in the IPL, begins at the first synapse in the retina.
ON cone bipolar cells
The dendrites of ON bipolar cells invaginate into the cone pedicle and approach the synaptic ribbons in a central position. ON bipolar cells are depolarized by light. This is opposite to the cone signal so we refer to this as a sign-inverting synapse, hence the minus sign at the cone/ON bipolar cell synapse in Figure 15.14.15 The dark release of glutamate from the photoreceptors holds ON bipolar cells relatively hyperpolarized. Light turns off the cone transmitter, and the decrease of glutamate release produces a depolarization in ON bipolar cells. Bipolar cells obey the division of the IPL and so ON bipolar cells are stratified in sublamina b (Fig. 15.13).
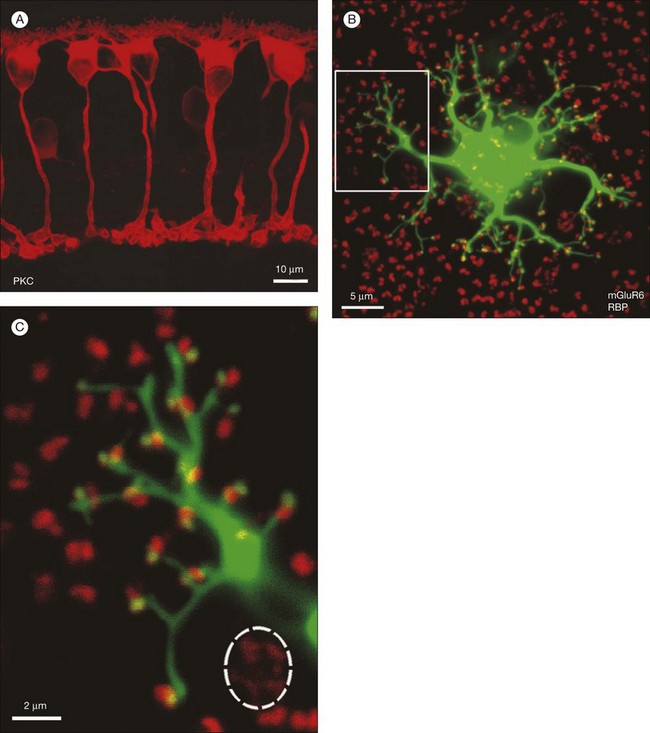
A hyperpolarizing response to glutamate is unusual and this is an unusual receptor, which is only expressed in the retina. It has now been identified as mGluR6, one in a series of eight metabotropic glutamate receptors, so called because activation of these receptors turns on an intracellular signaling cascade.142,143 The mGluR6 receptor is selectively activated by glutamate or the glutamate analog 2-amino-4-phosphonobutyrate (APB).15 Activation of the mGluR6 receptor leads to the closure of a cation channel, producing a hyperpolarization in ON bipolar cells. Light decreases glutamate release from photoreceptors and produces the opposite response. The cation channel is now known to be the transient receptor melastatin 1 (TRPM1) channel.144–146 The exact mechanism by which mGluR6 activation closes TRPM1 is not known, but it does not seem to use a cyclic nucleotide-mediated mechanism.147,148 While the details of the mechanisms remain uncertain, the signal inversion that occurs at the ON bipolar mGluR6 receptor underlies the separation between ON and OFF channels throughout the visual system.
Labeling the retina with an antibody against the mGluR6 receptor stains a narrow band at the level of bipolar dendrites in the OPL.17,97 In whole mount, the distribution of mGluR6 labeling shows a distinct pattern with two types of terminals (Fig. 15.15C). There are lightly labeled mGluR6-positive clusters of fine ON cone bipolar terminals at each cone pedicle and bright mGluR6-positive terminals, which insert into each rod spherule. These are the terminal dendrites of rod bipolar cells. This mGluR6 pattern also provides a simple way to map the location of rod and cone terminals in the outer retina. All ON bipolar cells, which include ON cone bipolar cells, blue cone bipolar cells, and rod bipolar cells, express mGluR6 at the tips of their dendrites.17 This is quite different from the multiple glutamate receptors used by OFF cone bipolar cells. However, a variety of responses may still be produced by the different types of ON cone bipolar cell due to modulation of the mGluR6 cascade or to the action of calcium channels or amacrine cells at the bipolar cell terminal.149
Midget bipolar cells
The primate retina is unusual in that the central retina is dominated by midget bipolar cells. Within the central 10 mm, there is one OFF midget bipolar cell and one ON midget bipolar cell for each cone. In this area, they account for more than 80% of all cone bipolar cells.130,150 Midget bipolar cells have very small dendritic fields and receive input from single cones and make output to single midget ganglion cells. This is the so-called private line, one cone to one midget bipolar cell to one midget ganglion cell. The ON midget bipolar cells invaginate the cone pedicle, ramify in sublamina b of the IPL, and contact ON midget ganglion cells. OFF midget bipolar cells make flat or basal contacts with cones and terminate in sublamina a where they contact OFF midget ganglion cells. Most investigators think this specialization of the primate retina was designed to achieve maximum acuity at high cone density. It may also, by virtue of the single cone connections, which are automatically color-coded, serve red/green color vision.
Blue cone bipolar cells
In general, diffuse cone bipolar cells contact every cone within the dendritic field and this gives them a characteristic appearance. However, in the primate retina, one bipolar cell type is distinctly different in that it has long dendrites that bypass many cones to seek out only blue cones.151,152 The dendrites are labeled for mGluR6 and the axon descends deep into sublamina b so the blue cone bipolar cells are ON cells.17,153
Rod bipolar cells
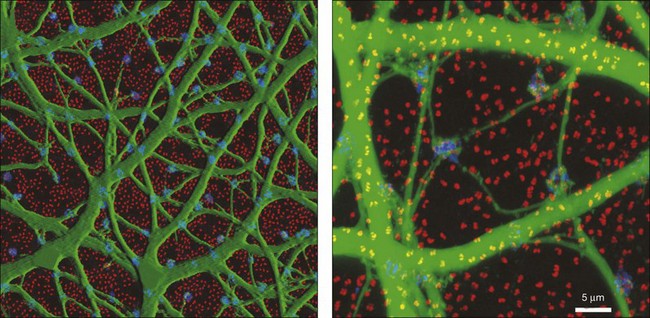
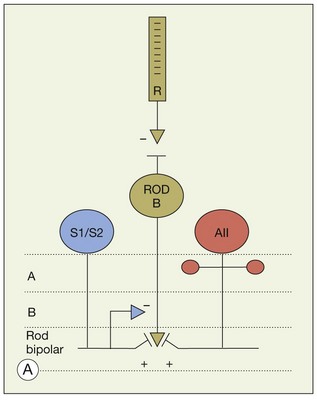
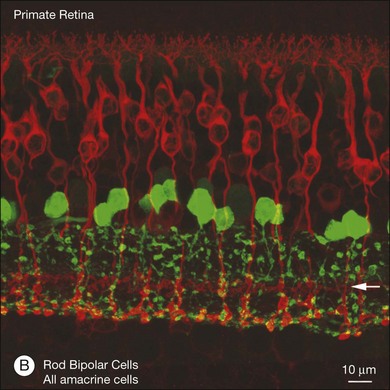
In contrast to the cone bipolar cells, there is only one morphological type of rod bipolar cell. They are numerous, with a mop-head of fine dendrites that may receive input from as many as 80–120 rods in the rabbit retina (Fig. 15.16A).88 This very high convergence contributes to the sensitivity of the primary rod pathway. A single dye-injected rod bipolar cell is shown in Figure 15.16B. The dendrites branch profusely but avoid the cone pedicles within the dendritic field. Instead, all the terminal dendrites invaginate a rod spherule and they are double-labeled for mGluR6. Only one terminal at each rod spherule comes from this rod bipolar cell. The other half of each mGluR6 doublet is claimed by another unlabeled rod bipolar dendrite. Thus, each rod contacts two different rod bipolar cells.97 Each rod bipolar cell gives rise to a long slender axon that descends to sublamina 5 of the IPL (Fig. 15.16). Physiologically, rod bipolar cells give ON responses to light stimulation and this is consistent with the labeling for mGluR6 and the depth of stratification.13,154 Rod bipolar cells do not usually contact ganglion cells directly. Instead, the primary output of rod bipolar cells is to AII amacrine cells, which pass on the rod signal, or to S1 and S2 amacrine cells42,154 that provide a powerful negative-feedback signal to rod bipolar terminals (Fig. 15.17; see below). More than 90% of the rod bipolar output is on to these amacrine cells.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree