Endoscopic sinus surgery (ESS) has been accepted as the treatment of choice for chronic sinusitis refractory to medical treatment. However, surgery of the frontal sinus is perhaps the most technically demanding of all ESS. The complex and varied anatomy, acute nasofrontal angle, and proximity to critical structures such as the olfactory fossa, skull base, and orbit contribute to the surgical difficulty. These narrow confines, and a penchant for postoperative scarring, make surgical treatment of chronic frontal sinusitis challenging.
The literature and evolution of endoscopic frontal sinus surgery supports a graduated stepwise approach to tackling disease in the frontal sinus (
1,
2,
3,
4). A wide spectrum of defined endonasal surgical procedures of the frontal sinus has been developed. These are based on the drainage or sinusotomy classification of Draf (
1) (
Table 46.1).
Our philosophy is an all-or-nothing approach to frontal sinus surgery once maximum medical treatment has failed. Limited surgery in the frontal recess places raw surfaces in close proximity to each other, which increases the likelihood of scarring and failure. Draf IIA sinusotomies are performed for primary surgery on the frontal sinus. Failed frontal sinus surgery is treated with a Draf III sinusotomy (otherwise known as endoscopic modified Lothrop procedure [EMLP] or frontal drillout procedure). At present, a primary EMLP is not performed for chronic rhinosinusitis.
SURGICAL ANATOMY
The key to successful frontal sinus surgery is a fundamental understanding of its anatomy.
Development of the frontal sinus begins in the fourth fetal month when the entire nasofrontal area is represented by the frontal recess. It is the last paranasal sinus to develop. At birth, the sinus has usually not formed. At around 2 years of age, the most anterior ethmoidal sinus invaginates into the frontal bone and continues its vertical growth trajectory at an average annual rate of 1.5 mm until the 15th year. Final growth is completed before 20.
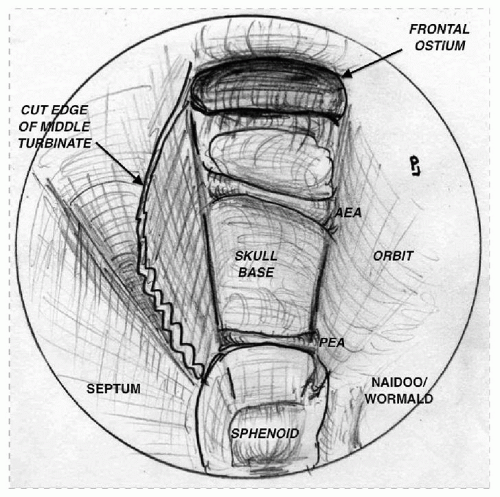
The sinus is compartmentalized by the intrasinus septa, which divides the sinus into halves. The frontal sinus has the most complex and variable drainage of any paranasal sinus. The frontal recess itself can be considered to be bordered by the frontal beak anteriorly, the anterior face of the bulla ethmoidalis posteriorly, the middle turbinate medially, and the lamina papyracea laterally (
Fig. 46.1).
These limits are altered by the variable pneumatization of anterior ethmoidal cells. Along the frontal beak, the agger nasi cell and other frontoethmoidal cells protrude into the frontal recess and sinus to alter the drainage pathway. Along the skull base, suprabullar cells may protrude anteriorly compressing the drainage pathway. When a suprabullar cell pneumatizes along the skull base into the frontal sinus, it is called a frontal bulla cell. Frontal bulla cells can considerably narrow the frontal sinus drainage pathway.
The types and variability of cells that can be found in the frontal recess and frontal sinus, which can markedly alter the frontal sinus drainage pathway, were first described by Kuhn (
5) in a landmark paper. He described four basic cells: the agger nasi cell, the frontoethmoidal cell(s), the intersinus septal cell(s), and the cell(s) originating from the bulla ethmoidalis/suprabullar region.
This classification was modified by Wormald (
6), who more clearly defined the frontoethmoidal cell and redefined type 3 and 4 cells (
Table 46.2).
The three-dimensional (3D) reconstruction of the frontal recess anatomy using the building block model, with each block representing a cell within the frontal recess, is an excellent aid to the surgeon in understanding the anatomy of the frontal sinus. It allows the surgeon to carefully
plan the surgical approach and to identify each cell to be dissected as well as to understand the drainage pathway of the frontal sinus allowing the dissecting instruments to be placed along the drainage pathway facilitating a safe dissection. This should allow a stepwise cell removal with complete exposure of the frontal ostium.
The first step in the reconstruction is to identify on the coronal computed tomography (CT) scan, the first cell anterior to and directly above the insertion of the middle turbinate. In most cases, this is the agger nasi cell (
Fig. 46.3A). This is confirmed on the parasagittal scan (
Fig. 46.3B). A building block is placed for the agger cell. The cell above the agger nasi cell is then identified and confirmed by cross reference with the parasagittal scan. This is a T1 cell and a block is placed to represent this. A cell is noted above this cell and on the parasagittal scan this cell is seen to extend above the frontal beak. This is a T3 cell and a block is placed to represent this cell.
Similarly, blocks are placed for the ethmoid bulla and suprabullar cell, which are seen on the coronal and parasagittal scans (
Fig. 46.3B).
The final aspect of the reconstruction is to identify how the frontal sinus drains in relation to these cells. This is done by following the frontal sinus from superiorly to inferiorly on sequential axial scans. In this way, the frontal sinus itself can be distinguished from cells that protrude into the sinus. An asterisk (*) marks the frontal sinus on the sequential scans (
Fig. 46.3C). This shows an anterior and medial drainage pathway. The completed building block model is shown in
Figure 46.3D.