FIGURE 33.1 Pharyngoesophageal deconstruction using the anterolateral thigh flap. A. The design of a two-skin island flap permits a trapezoidal design for the conduit and an elliptical design for the skin resurfacing. B. The proximal skin island based on the proximal perforator is tubed or used as a patch to form the conduit. C. This is sutured into the base of the tongue and posterior pharynx proximally and distally into the esophageal inlet. D. The external skin island is used for monitoring and resurfacing the defect in the neck.
Occasionally, circumferential defects of the cervical esophagus occur in patients in whom the anterolateral thigh donor site is unavailable. Reasons include previously harvested flaps, previous hemicorpectomies, obese thighs, critical atherosclerotic damage to the profunda femoris, or critical atherosclerotic disease of the superficial femoral artery, which is being effectively bypassed via the descending branch of the lateral femoral circumflex artery. In these cases, a jejunal free flap is the best option. Jejunal free flaps, which are often used in cases of cancer of the hypopharynx and cervical esophagus, are limited to short esophageal defects due to the segmental blood supply. The flap is based on a single branch of the superior mesenteric artery and vein. Generally speaking, one mesenteric branch supplies 15 to 20 cm of bowel. Therefore, in defects of 15 cm or less, for which the distal portion of the esophagus is accessible through the neck, the jejunal free flap is an excellent option.
For defects greater than 15 cm and for which the distal esophagus is inaccessible due to its intrathoracic location, a gastric conduit is the first choice for reconstruction due to its robust blood supply and requiring only one bowel anastomosis to reestablish continuity; however, in certain situations, the stomach is unusable. Colon interposition has been employed for several decades as an alternative to the gastric conduit; however, patients with atherosclerosis, inappropriate vascular anatomy, previous colon resection, or intrinsic colon disease, such as inflammatory bowel disease, cancer, or diverticulosis, are not candidates.
The pedicled supercharged jejunum is the preferable alternative because it closely approximates the size of the native esophagus, possesses peristaltic activity, and is typically free of intrinsic diseases. The addition of microsurgical techniques has allowed for the replacement of the entire esophagus using longer conduits through “supercharging.” Longmire described the first supercharged jejunal flap for reconstruction of the esophagus in 1947. Since then, the field of microsurgery has advanced significantly and the use of these techniques has become more widespread, making the supercharged jejunal flap the best option for total esophageal reconstruction when the stomach is unavailable.
CONTRAINDICATIONS
Total esophageal reconstruction using a free, segmental, or pedicled, supercharged jejunal flap is technically challenging and requires a multidisciplinary approach. Relative contraindications include a surgeon who is not working with a team to manage the patient and patients with prior extensive abdominal or specifically jejunal surgery.
PREOPERATIVE PLANNING
These procedures can be successfully performed as an alternative reconstruction when the stomach is unavailable with good long-term function and acceptable morbidity. Thorough surgical planning is critical to the success of this operation. When using the jejunum, preoperative planning requires that a careful operative history is taken to assess the potential for abdominal adhesions that may hinder the harvest of a jejunal segment.
SURGICAL TECHNIQUE
The initial approach to the abdomen is the same for both the free segmental and pedicled supercharged jejunum. Once resection is complete, the ligament of Treitz is identified and the small bowel with its mesentery is explored to ensure that no intrinsic abnormalities or iatrogenic injuries exist. The length of the esophageal defect is measured to estimate the length of the conduit needed (Fig. 33.2). The mesentery of the proximal and mid jejunum are closely examined using a fiberoptic light to transilluminate the tissues and elucidate the vascular anatomy (Fig. 33.3).
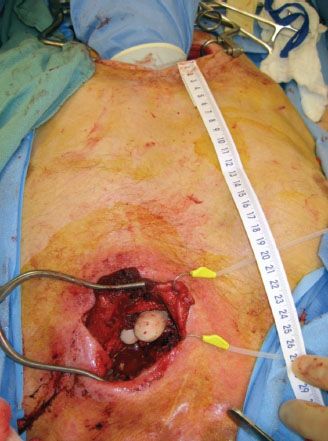
FIGURE 33.2 In esophageal reconstruction, the length of the conduit is measured by the distance from the proximal portion to the distal portion, often through the thoracic inlet and outlet.
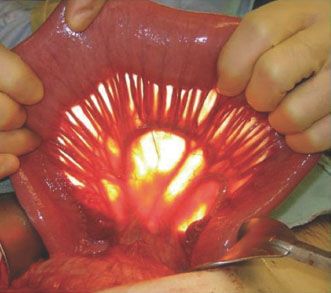
FIGURE 33.3 Transillumination of the small bowel is used to demonstrate the vascular anatomy of the inferior mesenteric artery and vein and identify the most appropriate vessels to divide and transfer.
Free Jejunal Segment
If a free jejunal segment is the reconstructive option of choice, a single mesenteric vessel is selected around 40 cm from the ligament of Treitz, which usually corresponds to the second or third mesenteric branch. The choice of these vessels yields the largest arc of mesentery and thus the longest vascular pedicle. Once the appropriate vessel is identified, it is dissected free from the mesentery. The secondary arcade emerging from this single mesenteric vessel must be kept intact to ensure vascularity of the entire segment. This is done by harvesting the wedge of mesentery between the pedicle and the jejunal segment (Fig. 33.4). Dissection through the mesentery can be assisted by transillumination. The mesenteric vessels are extremely friable, and great care should be taken to avoid rough dissection, which will result in a hematoma of the mesentery and obfuscate the dissection. This dissection is taken up to the serosal border distally, and the superior mesenteric artery and vein proximally, insuring maximum pedicle length and caliber.
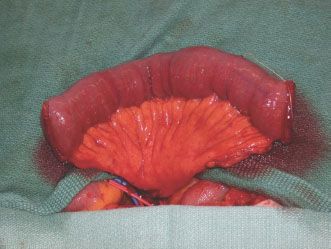
FIGURE 33.4 For a free jejunal segment, the mesenteric vessel that will yield the longest arc of mesentery is selected and dissected free from the mesentery. The secondary arcade emerging from this single mesenteric vessel must be kept intact to ensure vascularity of the entire segment. This is done by harvesting the wedge of mesentery between the pedicle and the jejunal segment.
Prior to dividing the bowel, the proximal or isoperistaltic portion of the jejunum is marked. This insures appropriate orientation in the neck. The bowel is divided with a standard endostapler. The pedicle is then ligated, and the flap is transferred to the head and neck. A stapled, side-to-side anastomosis of the remaining jejunum in the abdomen is performed, or a two-layer, hand-sewn end-to-end anastomosis, depending on surgeon preference, and the midline incision is closed in standard fashion.
Once in the neck, heparinized saline is run through the flap as is customary for all free flaps. The bowel is oriented appropriately, usually in an isoperistaltic fashion. Generally, I like to perform at least the distal esophageal anastomosis prior to the microvascular anastomosis since this will set the tension on the bowel. The proximal 3 to 5 cm of jejunum, based on one or two terminal arcade vessels, is exteriorized as a postoperative monitoring segment. The esophagojejunal anastomoses are then performed in the neck using a single layer of 3-0 Vicryl or polydioxanone suture (PDS) in an end-to-end fashion. In patients with a total laryngopharyngectomy, the proximal pharynx or the base of the tongue is larger than the jejunal diameter, so an end-to-side anastomosis is performed to accommodate the size discrepancy.
Recipient vessels are prepared prior to dividing the vascular pedicle and passing the jejunal conduit. The transverse cervical vessels (if the neck has been significantly damaged due to prior surgery and/or radiation), the external carotid system (the facial, lingual, or superior thyroid arteries), and jugular veins are common recipient vessels depending on the clinical scenario (Fig. 33.5). Arterial anastomoses are performed under the operating microscope using 9-0 Nylon sutures. Venous anastomoses are commonly completed using a venous coupling device (Synovis Surgical Innovations, St. Paul, MN).
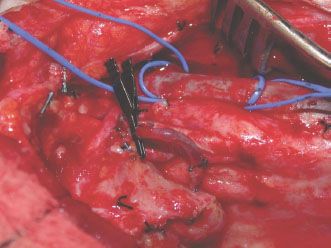
FIGURE 33.5 Recipient vessels are prepared prior to dividing the vascular pedicle and passing the jejunal conduit. The transverse cervical vessels (if the neck has been significantly damaged due to prior surgery and/or radiation), the external carotid system (the facial, lingual, or superior thyroid arteries), and jugular veins are common recipient vessels depending on the clinical scenario. In this case, the vessel loop is around the jugular vein, and the Ackland clamp is on the facial artery.
Pedicled Supercharged Jejunum
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


