Fig. 1
Age at which tracheostomy was performed. Stratification by age group. Reprinted from Perez-Ruiz et al. [3]. Copyright 2012. with permission from the European Respiratory Society.
Table 1
Indications for Tracheostomy
Indication | Examples |
|---|---|
Structural anomalies | Anomalies of the Nasal Passages |
Congenital arhinia | |
Severe nasal deformity | |
Epignathus | |
Encephalocoele | |
Pharynx/Larynx | |
Pharyngeal cyst | |
Subglottic stenosis | |
Laryngeal atresia | |
Laryngeal cleft | |
Laryngotracheal hemangiomas or papillomas | |
Bilateral vocal cord paralysis | |
Trachea | |
Tracheomalacia | |
Tracheal stenosis | |
Tracheal atresia | |
Craniofacial anomalies | |
Head or neck mass obstructing the upper airway (e.g., giant cervical teratomas with airway obstruction) | |
Pierre-Robin sequence/retrognathia/micrognathia | |
Medical Problems | Laryngotracheal stenosis from prolonged intubation |
Bronchopulmonary dysplasia | |
Need for prolonged continuous positive airway pressure (CPAP) | |
Chronic aspiration syndrome | |
Seizures with airway complications | |
Spastic quadriplegic cerebral palsy | |
Osteogenesis imperfecta | |
Chiari II malformation | |
Diaphragmatic paralysis | |
Neuromuscular diseases, e.g., Spinal Muscular Atrophy type 1 | |
Medical support following other treatment | Post cardiac surgery |
Other congenital anomalies associated with difficulty with airway toileting, giant omphalocele with pulmonary hypoplasia, congenital diaphragmatic hernia, other congenital lung malformations |
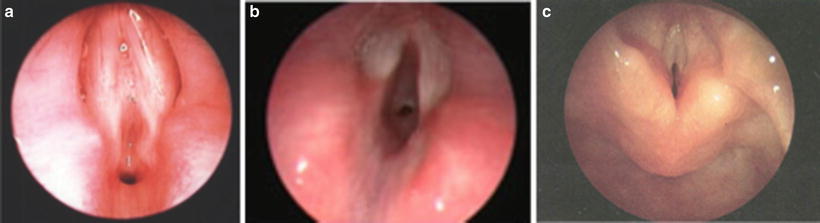
Fig. 2
Examples of airway anomalies necessitating tracheostomy. (a) near-complete glottic web. (b) laryngotracheal (subglottic) stenosis, (c) bilateral vocal cord paralysis. (Courtesy of Steve Sobol, The Children’s Hospital of Philadelphila (CHOP), Neonatal Airway Program)
Vocal cord paralysis may occur secondary to central nervous system abnormalities including Arnold-Chiari malformation, cerebral palsy, hydrocephalus, myelomeningocele, spina bifida, or hypoxia (Fig. 2c). In contrast to anatomic airway obstruction, laryngomalacia may not present until the second week of age or beyond (Fig. 3).
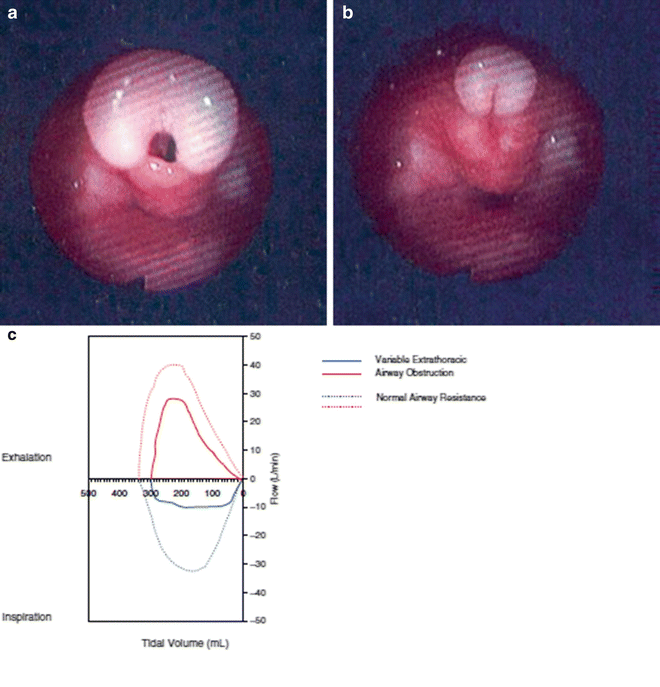

Fig. 3
Endoscopic appearance of the larynx in a patient with dynamic airway compression secondary to laryngomalacia. (a) Severe laryngomalacia during expiration. (b) Same patient during inspiration with total collapse of the airway. (c) Flow-volume loops further demonstrating evidence of worsening extra-thoracic airway obstruction during inspiration (Reprinted from Shanley et al. [66]. Copyright 2014 with permission from Springer)
Airway and lung diseases that arise as sequelae of postnatal therapies tend to account for most of the tracheostomies in older infants. The specific indications for tracheostomy placement in preterm infants have evolved as modalities for respiratory support for early preterm infants have changed over the years. Such new interventions and new approaches include less invasive respiratory support (continuous positive airway pressure (CPAP), high-flow nasal cannula), increased use of steroids (both systemic and inhaled) in the peri-extubation period and lung-protective ventilation strategies, such as permissive hypercarbia [9]. These newer therapies and changes in neonatal practice have resulted in decreased prevalence of iatrogenic laryngeal tracheal stenosis (LTS), which was long the most common indication for tracheostomy placement in the preterm population [10–13] (Fig. 2b). Today, the most common indication for tracheostomy in preterm infants is prolonged ventilation secondary to bronchopulmonary dysplasia, rather than pure airway disease [4, 14, 15] (Fig. 4). Murthy et al. report a study of over 5,500 infants born before 32 weeks gestation who were referred to regional NICUs [16]. Eight hundred and ninety eight of these infants were diagnosed with severe bronchopulmonary dysplasia (sBPD), defined as receiving more than 30 % oxygen with a flow rate more than 2 L per minute or positive-pressure ventilation at greater than 36 weeks post menstrual age. Of the infants with sBPD, 107 (12 %) infants received a tracheostomy to facilitate long-term ventilatory support. The median time from referral to tracheostomy was 52 days. Analysis of this large cohort of preterm infants has provided some insight into risk factors for requirement of tracheostomy (or for the competing outcome of death) in these infants. Surprisingly, infants with sBPD who were born at a later gestational age were more likely to die or require a tracheostomy. Thus, while infants born at a later gestational age are less likely to develop sBPD, those that do have a worse prognosis. Other risk factors include being born small for gestational age or having sBPD complicated by pulmonary hypertension and cor pulmonale. Pulmonary vascular disease leading to pulmonary hypertension is a life-threatening complication of sBPD that often contributes to the need for prolonged ventilation in preterm infants. Because of improvements in neonatal care leading to improved survival of very preterm infants, and advances in the surgical management of congenital and iatrogenic airway disorders, the need for prolonged ventilatory support for BPD is likely to remain an important indication for tracheostomy in the first year of life (Fig. 5a, b).
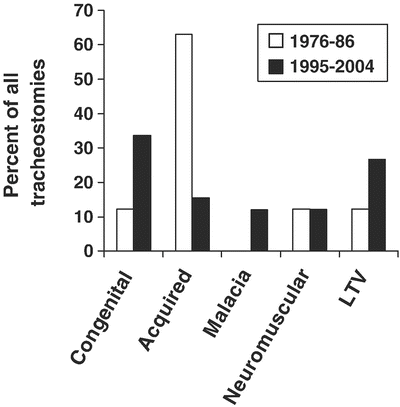
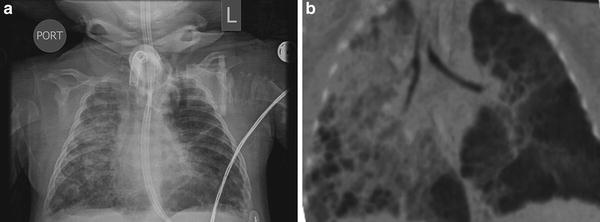

Fig. 4
Changes in indications for tracheostomy over time. From Corbett et al [65] Copyright 2007 with permission from Elsevier

Fig. 5
Infant with severe BPD. (a) Plain chest radiograph. (b) CT angiogram showing severe cystic changes (Courtesy of, (CHOP), Chronic Lung Disease program
Below are a variety of CT angiography (CTA) images showing the extensive different types of lung damage seen in premature infants who remain ventilator dependent. One can see both atelectasis and hyperinflation with cystic formation in the same lung. Also, evidence of pulmonary hypertension and airway disease can be seen simultaneously on the same image (Fig. 5c–m).
In addition to BPD, other chronic medical conditions, including congenital heart disease or neuromuscular disorders such as spinal muscular atrophy (SMA), congenital giant omphalocele, congenital diaphragmatic hernia and congenital cystic adenomatous malformation all can result in a need for long-term ventilation via tracheostomy (Table 1).
Complications of Tracheostomy
While tracheostomy-related deaths are uncommon, complications of tracheostomy are not infrequent. (Table 2). In studies where complication rates are reported, the rates of tracheostomy complications in infants less than 1 year of age ranged from 29 to 77 % [14, 17–21]. Most studies show an increased incidence of complications in younger, smaller patients. Complications can be classified by timing: intraoperative; early (usually defined as the first postoperative week); late; and post-decannulation. Intraoperative complications are rare, and range from desaturations to cardiopulmonary arrest. Complications in the first post-tracheostomy week include bleeding, cannula obstruction or mucous plugging, stomal erosion, and infection or cellulitis at the tracheostomy site. Accidental decannulation can occur as well, and has the potential to result in malposition of the cannula outside the trachea or creating of a tracheal false passage. Air leak, including pneumothorax, pneumomediastinum, and subcutaneous emphysema have been reported.
Table 2
Complications of tracheostomy
Timing | Complication |
|---|---|
Intraoperative | Cardiopulmonary arrest |
Posterior tracheal wall tear | |
Early | Hemorrhage |
Mucous plugging | |
Infection | |
Accidental decannulation | |
Pneumothorax | |
Subcutaneous emphysema | |
Tracheostomy malposition/false passage | |
Late | Granulation tissue at stoma |
Tracheal granulomas | |
Mucous plugging | |
Accidental decannulation | |
Stoma infection | |
Tracheitis | |
Hemorrhage | |
Suprastomal collapse | |
Subglottic stenosis | |
Tracheomalacia | |
Tracheoinnominate artery fistula | |
Post-decannulation | Tracheocutaneous fistula |
Hypertrophic scar | |
Recurrent stridor | |
Granulation tissue |
Overall, granulation tissue in the trachea or at the stoma site is the most common complication of tracheostomy. Tracheal wall granulomas occur in 4–80 % of pediatric tracheostomies [22]. Granulomas are frequently found at the superior margin of the tracheostomy tube due to irritation and inflammation of the tracheal mucosa by the cannula (Figs. 6 and 7). Granulomas may also be found at the distal tip of the tracheostomy tube, usually caused by malposition of the tube or by irritation from traumatic suctioning. Small to moderately sized asymptomatic granulomas can be managed expectantly, while large or symptomatic granulomas usually need to be excised. Pressure placed on the tracheal rings by the tracheostomy tube can cause chondritis, inflammation, and deterioration of the cartilage, resulting in suprastomal collapse. (Figs. 7c and 8). The collapse is usually moderate and does not need treatment prior to decannulation. When severe, treatment may be needed before successful decannulation can be achieved.
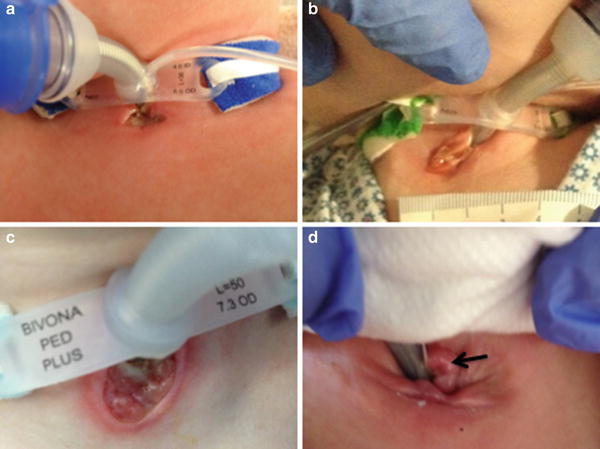
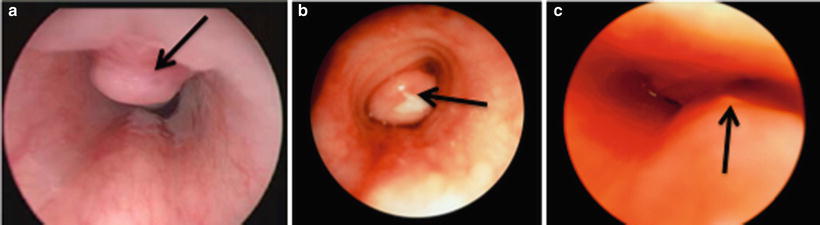
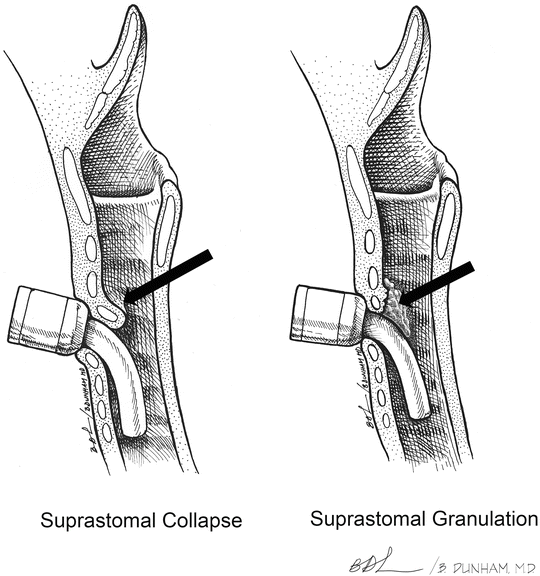

Fig. 6
Tracheostomy stomas. (a) Normally healing stoma. (b) Stomal breakdown. (c) Severe stomal breakdown due to excess secretions. (d) Stomal granuloma. (Courtesy of Steve Sobol, (CHOP), Neonatal Airway Program)

Fig. 7
Suprastomal collapse and granuloma (Courtesy of Michael Rutter, Department of Otolaryngology, Children’s Hospital Medical Center, Cincinnati, OH)

Fig. 8
Airway complications. (a) suprastomal granuloma (black arrow). (b) Subglottic granulomas (black arrow). (c) Suprastomal collapse (black arrow). (Reprinted from Zur et al. [63]. Pediatric Otolaryngology: The Requisites in Pediatrics, (Wetmore ed). Copyright 2007 with permission of Elsevier
As discussed above, Laryngotracheal stenosis (LTS) is a common indication for tracheostomy, particularly in preterm infants who have undergone prolonged endotracheal intubation. LTS can also be caused by irritation of the laryngotracheal tissues by the tracheostomy cannula. Whether it is an indication for or the result of the tracheostomy, the presence of LTS often necessitates laryngotracheal reconstruction (LTR) prior to decannulatio. Similarly, Tracheomalacia, the result of a deficiency of support by cartilage rings, often contributes to the need for tracheostomy in infants (Fig. 9). Long-term tracheostomy can also result in tracheomalacia. There is currently no definitive treatment for tracheomalacia in infants and young children. If severe enough to prevent successful decannulation, the presence of this complication may therefore result in the need for prolonged tracheostomy.
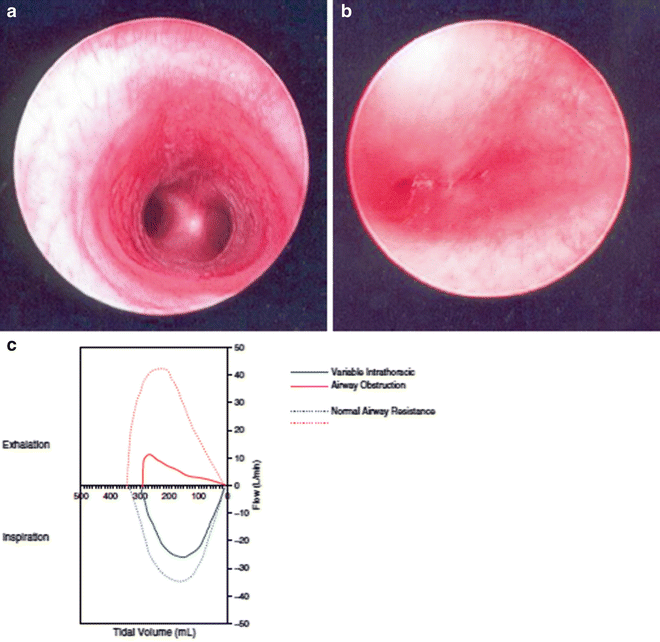

Fig. 9
Endoscopic appearance of the trachea in a patient with dynamic airway compression secondary to tracheomalacia. (a) Normal trachea. (b) Child with severe tracheomalacia demonstrating total collapse of the airway during inspiration. (c) Flow-volume loops further demonstrating evidence of worsening intrathoracic airway obstruction during expiration (Reprinted from Shanley et al. [66]. Copyright 2014 with permission from Springer)
Tracheostomy can be complicated by infection, either at the stomal site or in the trachea (Fig. 10). Stomal site infection represents a localized skin infection and is treated as cellulitis. A tracheostomy tube bypasses the normal host defenses against infection found in the nose, mouth, and upper airway, leaving children with tracheostomies more susceptible to infection of the trachea with a variety of bacterial, fungal, and viral pathogens. Tracheitis can often be difficult to distinguish from microbial colonization that is universally seen in children with long-standing artificial airways. The diagnosis depends on combining signs of clinical illness or respiratory deterioration with evidence of inflammation and identification of a pathogen.
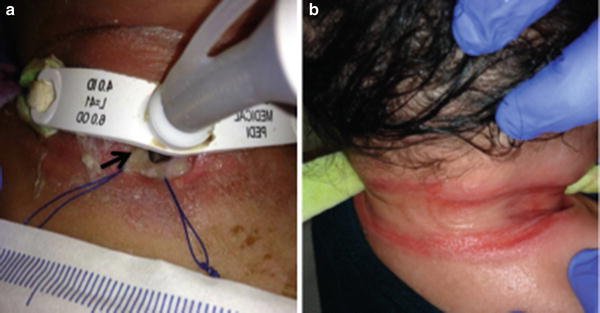

Fig. 10
Tracheostomy-associated infections. (a) stoma infection (black arrow). Surrounding erythema represents cellulitis. (b) Breakdown on the neck from tracheostomy ties. (Courtesy of Steve Sobol, CHOP Neonatal Airway Program)
Although tracheostomy-related deaths are less common than once thought, cannula occlusion by mucous plugs, accidental decannulation, malpositioning of cannulas into false passages, and less commonly, ventilator failures can result in morbidity and mortality, particularly after discharge. Prevention of these unexpected events depends on careful attention to discharge planning and caregiver education, assessment of the home environment and needed support structures, and proper equipment.
After decannulation, the tracheostomy site is routinely left to heal by secondary intent. This process can result in hypertrophic or depressed scars, at times necessitating tracheal scar revision. Perhaps the most common complication seen after decannulation is a persistent tracheocutaneous fistula (TCF), which is essentially a failure of the stoma to close (Fig. 11). Persistent TCF occurs in up to 50 % of patients after decannulation [14, 20, 23, 24]. The risk of TCF is inversely correlated with the age at cannulation and increases with the duration of cannulation. It is common practice to observe a TCF for 6–12 months before closing it surgically [25].
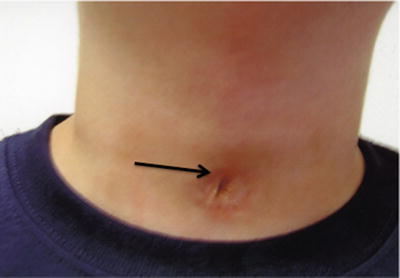

Fig. 11
Tracheocutaneous fistula (black arrow). (Courtesy of Steve Sobol, CHOP Neonatal Airway Program)
Timing of Tracheostomy
Very little research is available to guide decisions about the timing of tracheostomy placement in the neonate or infant. Existing data are universally retrospective and therefore reflect variations in surgical attitudes, rather than experimental approaches to determining best practice. There is a recent movement toward earlier tracheostomy in adult critical care. This is supported by data linking earlier tracheostomy (typically at 3–10 days) with improved outcomes including shorter durations of mechanical ventilation, intensive care, and hospitalization, and lower costs [26–28]. However, a 2012 Cochrane systematic review of randomized trials on this subject concludes that there is not sufficient evidence to support a decrease in mortality after early tracheostomy [29] Though the adult data are not conclusive, some authors are beginning to suggest that children or even infants might also benefit from earlier tracheostomy [5, 30, 31].
Two single-center studies have evaluated the relationship between timing of tracheostomy and outcomes. The first included 43 infants born before 36 weeks postmenstrual age (mean gestational age 27.9 weeks) [32]. The mean age at tracheostomy placement was 21.4 weeks, and the more preterm infants were more likely to be cannulated later in life. However, timing of cannulation did not correlate with successful weaning from mechanical ventilation, decannulation, or the timing of these events. The second included 127 infants with mean gestational age of 28 weeks [33]. Mean age at tracheostomy was similar at 45 weeks postmenstrual age. Twenty-one percent of the infants died and, after adjustment for gestational age and respiratory support, mortality was unrelated to the timing of tracheostomy. A large, multicenter study from the NICHD Neonatal Research Network reported the outcomes of 304 preterm infants (<30 weeks gestation) who underwent tracheostomy placement and were followed until 18–22 months of age. The study reported that the adjusted odds of death or developmental impairment at 18–22 months were significantly lower (adjusted odds ratio 0.5, 95 % CI 0.3–0.9) among infants who underwent tracheostomy before 120 days of life, as compared to those who underwent tracheostomy after 120 days of life. The authors were unable to report indication for tracheostomy, so it is possible that the patients who underwent later tracheostomy were a population with unmeasured risk factors for adverse outcome. Alternately, it is also possible that there is a developmental advantage to earlier tracheostomy in preterm infants [34].
Many factors other than postnatal age must be considered when determining the appropriate timing of tracheostomy. Sometimes the diagnosis or indication dictates the timing of tracheostomy. For instance, some anatomic anomalies (e.g., tracheal stenosis) cannot be managed without tracheostomy and the procedure must be performed urgently. However, in the majority of cases, the decision is less clear-cut. Some authors have suggested that infants, particularly preterm infants with chronic lung disease, undergo tracheostomy placement after a certain number of failed attempts at extubation. Sisk reported in 2006 that the median number of intubation events among a group (n = 18) of very low-birth weight infants who eventually underwent tracheostomy was 11.5 (range 7–25) [14]. Pereira et al. reported an average of 5.2 failed extubations among tracheostomized infants with birth weight <1 kg and 3.2 failed extubations among infants with birth weight >1 kg [4]. The same author concluded in another report that “infants with gestational ages of 30 weeks and below, those with low-birth weights and chronic lung disease are unlikely to benefit from more than three trials of extubation despite minimal ventilatory requirements at that time”[35]. On the other hand, among infants who underwent tracheostomy after repair or palliation of congenital heart disease, numbers of extubations prior to tracheostomy ranged from 0 attempts in those who could not wean from mechanical ventilation to more than three attempts [36]. Finally, while (as discussed above) complications of tracheostomy are more common among smaller and younger infants, it remains unclear whether complication rates differ significantly when tracheostomy is performed at different times within the first year of life. At a minimum, recent data suggest that for preterm infants, later tracheostomy (beyond 16 or 20 weeks of age) is not associated with increased need for tracheal reconstruction [32]. This finding suggests that acquired subglottic stenosis is not more common when the procedure is delayed for a few months.
Why might infants benefit from earlier tracheostomy? One hypothesis is that earlier tracheostomy allows faster weaning of sedating medications and muscle relaxants and earlier institution of developmentally appropriate activities [32, 34]. Early developmental interventions improve cognitive outcomes and may improve motor outcomes for preterm infants [37]. With the more stable airway provided by the tracheostomy, infants who have suffered prolonged intubation can often begin to sit in chairs and swings, lay prone, and participate more actively in interactions with parents and caregivers. However, more studies are needed to confirm that earlier tracheostomy is the safest option for infants before this strategy could be recommended to practitioners.
Survival and Decannulation After Neonatal Tracheostomy
Historically, there has been a perception that tracheostomy in infants resulted in increased risk of death. Indeed, in reports of tracheostomy in infants less than 1 year of age, published mortality rates range from 6 to 35 %. However, more recent data indicate that survival rates after neonatal tracheostomy may be improving over time. For example, in 1989, Singer reported 29 % mortality in a population of 130 infants who underwent tracheostomy before 13 months of age [38]. In 2013, Overman reported 89 % 5-year survival of 165 infants with tracheostomies [5]. However, this report excluded infants with congenital heart disease, who constitute an important segment of the population with particularly high mortality. More recently, DeMauro reported 92 % survival to 18–22 months among 304 preterm infants who survived to 36 weeks and then underwent tracheostomy placement [34]. More detailed evaluation of the changing survival rates over time is hindered by the inclusion of all children, rather than just neonates and infants, in many reports, or only subgroups of infants in other reports. All cause mortality in a population of 91 children who were discharged with tracheostomies has been described over a 20-year period [39] (Fig. 12).
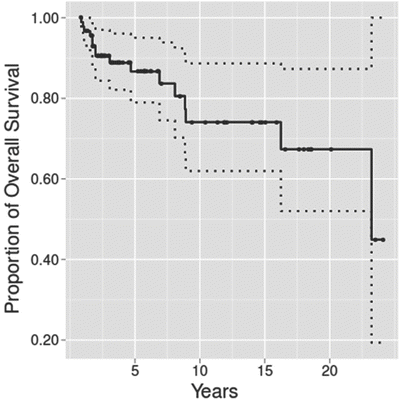

Fig. 12
Survival analysis of children treated with tracheostomy and positive-pressure ventilation at home: unadjusted Kaplan-Meier plot shows time of death with 95 % confidence band. (Reprinted from Com et al. [34]. Copyright 2014 with permission from Sage)
Survival rates after tracheostomy depend heavily on the initial indication for the tracheostomy, progression of underlying medical conditions and comorbidities. For instance, survival is higher for potentially reversible conditions such as airway obstruction and respiratory failure: survival for children with bronchopulmonary dysplasia (BPD) is about 80–90 %. On the other hand, mortality is higher among children with irreversible conditions such as progressive neurologic impairment, complex congenital heart disease, and genetic syndromes: survival for infants who undergo tracheostomy after surgery for complex congenital heart disease is 50–70 % [39–44]. One explanation for these widely disparate outcomes is that higher mortality rates in some subgroups are likely related to mortality from the underlying condition rather than differences in rates of “tracheostomy accidents” by subgroup [3, 39]. Certainly, both complication and mortality rates are also linked to the number of comorbid conditions present [45]. Unfortunately, cause of death after neonatal tracheostomy is rarely reported in the existing literature. When all infants and children with tracheostomies are considered, overall mortality is about 12–20 %; however, only about 10–25 % of these children die secondary to complications of the tracheostomy [3, 46].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


