Chapter 20 Follicular Thyroid Cancer
Introduction
Follicular thyroid cancer (FTC) is a subset of follicular cell–derived thyroid cancer. It falls within the broad category of differentiated thyroid cancer (DTC) and is the second most common histologic subtype behind papillary thyroid cancer (PTC). The frequency of FTC has been estimated between 9% and 40% depending on the population studied, iodine intake, and the use of the follicular variant of papillary thyroid cancer (FVPTC) as a subdiagnosis.1,2 In general, FTC represents approximately 10% to 15% of all thyroid cancers, with a majority of these being minimally invasive FTC. The incidence of DTC, including both PTC and FTC, has been rising over the last several decades. Though this rise was initially described as a surveillance bias detecting subcentimeter PTC,1 more recent data indicate there is likely a true rise in the incidence of DTC because of an increased incidence of all DTC including tumors > 4 cm.3,4
Epidemiology
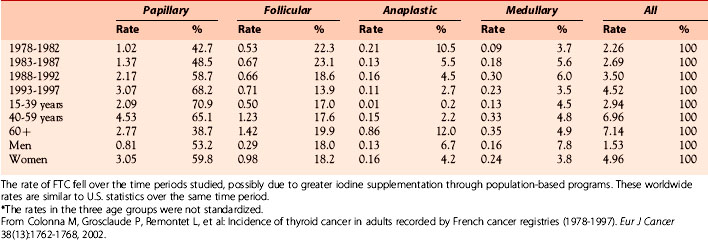
Caucasians account for the majority of new thyroid cancer diagnoses (> 90%) in the United States. However, the greatest annual percentage increase for both genders was among blacks (4.6% to 5.8%).3 The percentage of FTC relative to all thyroid cancer was estimated to be 14.2% between 1985 and 1990 and 11.4% from 1991 to 1995 in a U.S. cohort.5 The majority of patients with new FTC diagnoses during this time period, accounting for approximately 40% of all cases, were in their third to fifth decades of life. A more recent U.S. analysis shows that the incidence of FTC was stable between 1973 to 2002 at 1/100,000, whereas the rate of new PTC diagnoses rose from 3/100,000 to more than 7/100,000.1 A large Italian cohort study of 4187 patients with DTC showed a prevalence of 9% FTC in patients diagnosed after 1990, compared with a prevalence of 19.5% in those diagnosed between 1969 and 1990.6 One potential mechanism for the decline in FTC prevalence was the use of iodine prophylaxis in more recent years, as was shown in a world trend analysis7 (Table 20-1). An analysis of French thyroid cancer registries showed a similar small decline in the incidence of FTC cases from 1983 to 2000 with an absolute decrease of 2.2% and 0.5% annually in men and women, respectively.8 Most tumors in this analysis were 1 to 4 cm, and FTC tumors were larger than PTC tumors. The drop in incidence is not completely understood, though many think it could be partly related to a greater appreciation and diagnosis of the follicular variant of papillary thyroid carcinoma or changes in iodine supplementation programs.
Table 20-1 World Age Standardized Rates* (per 100,000 Person-Years) and Distributions of Cases per Histologic Type
There is a generally accepted correlation between endemic goiter in iodine-deficient regions and thyroid cancer, though the data are inconsistent. The relative risk of thyroid cancer in adjacent areas of Sicily with iodine deficiency compared to sufficiency was 1.4,9 though a similar study in Denmark found no difference.10 Other epidemiologic studies suggest that increased iodine supplementation (either as a public health measure or for individuals moving from an iodine-deficient region to a sufficient one) results in a transition from increased risk of follicular thyroid carcinoma to papillary thyroid carcinoma.11 Alternatively, the time of exposure to iodine deficiency (such as during childhood or puberty) may be a predisposing factor.12 One inherent challenge of all epidemiologic data correlating iodine status with FTC or DTC is the lack of a control group or analysis of other relevant variables. For example, selenium deficiency, which may play a role in cell differentiation, is often concurrent with iodine deficiency and is rarely accounted for in association studies of iodine and thyroid cancer.13
Etiology
The classic clinically evaluable risk factors for DTC include exposure to ionizing radiation (especially in youth) and family history of thyroid cancer. The nuclear reactor accident in Chernobyl, Russia, in 1986 illustrated the effects of radiation exposure on thyroid cancer risk (see Chapter 28, Chernobyl and Radiation-Induced Thyroid Cancer). Most cases of DTC associated with the Chernobyl accident were PTCs with RET/PTC rearrangements, and a few FTC were noted as well.14 Ionizing radiation appears to be a risk factor for PTC to a greater extent than FTC. There is larger literature of familial nonmedullary thyroid cancer (FNMTC) in relation to PTC relative to FTC, though no gene signature or gene cluster has been identified to predict risk (see Chapter 29, Familial Nonmedullary Thyroid Cancer). In a case control study of a Swedish population of DTC patients, there was no statistically significant increase in parental thyroid cancer for those subjects with FTC, though there was a greater than fourfold increased risk of thyroid cancer if the parent had PTC.15 As for other environmental and lifestyle factors, there is limited evidence for the role of diet and body mass index (BMI) as a risk factor, although consumption of iodized salt in a cohort study in Sweden did suggest protection against FTC.16–19
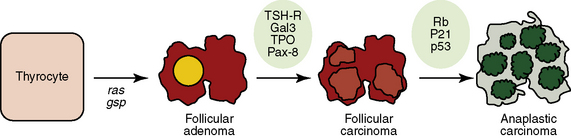
The recent era of molecular medicine has identified important mutations in FTC (Figure 20-1) (see Chapter 17, Molecular Pathogenesis of Thyroid Neoplasia).20 The underlying cause of oncogenesis, however, is poorly understood. Kroll et al. described the PAX8-PPARγ fusion oncogene that acts as a dominant negative transcription factor and is present in a subset of follicular carcinomas.21 In a study of 15 patients with histologically proved FTC, 8/15 (53%) had the PAX8-PPARγ rearrangement, and of the 3 patients with a history of radiation exposure, 100% had this mutation.22 A recent review of 17 studies shows that the Pax8-PPARγ rearrangement was identified in 36% (112/310) of FTC, 16% (13/83) of FVPTC, and 11% (27/247) of follicular adenomas (FTA), suggesting a significant role of this oncogene in the development of follicular tumors.23 Giordano and colleagues used global gene expression profiling to compare FTA and FTC with and without the Pax8-PPARγ rearrangement.24 They identified a 68-gene signature set that provided insight into potential molecular mechanisms governing Pax8-PPARγ dependent FTC. These included genes on chromosome 3p as well as genes involved in fatty acid and carbohydrate metabolism. Another common and distinct mutation associated with FTC is one that activates RAS (N-RAS, H-RAS, or K-RAS) through point mutations and thus constitutively activates the mitogen-activated protein kinase (MAPK) oncogenic signaling pathway. RAS mutations were identified in 49% of FTC. RAS mutations or the PAX-PPARγ rearrangement was present in 88% of this sample of FTC.25 Additionally, recent studies of FTC tissue shows a higher percentage of tumors with activated Akt (pAkt) that activated MAPK (pERK), suggesting that FTC is more dependent on the PI3K-Akt pathway than the MAPK pathway, which is important in PTC.26 Beyond providing evidence for the underlying genetic defects that may lead to FTC, these genetic signatures have proved to be useful for diagnostic purposes as will be discussed.
Diagnosis
Clinical Presentation
Similar to other forms of differentiated thyroid cancer, follicular thyroid cancer most commonly presents as an asymptomatic mass. The generally accepted risk of thyroid cancer in a nodule is 10% to 15%, including nodules that are discovered by the patient, by clinical neck exam, or incidentally on other imaging procedures (chest computed tomography [CT] scan, carotid ultrasound, etc.) (see Chapter 11, The Evaluation and Management of Thyroid Nodules).27 Concerning clinical features of thyroid nodules include rapid growth, firm or hard texture with fixation and occurrence in patients with either a family history of thyroid cancer or a personal history of external radiation to the head or neck, especially in childhood.28 Regardless of the method of initial detection, a complete thyroid and neck ultrasound (US) is recommended to define thyroid nodule characteristics, evaluate for additional nodules, and evaluate for characteristic lymph node metastases as described elsewhere in this book (see Chapter 13, Ultrasound of the Thyroid and Parathyroid Glands).29 A recent study compared US characteristics of FTC versus FTA. The sonographic features of follicular adenoma and follicular carcinoma were similar, but larger lesion size, lack of a sonographic halo, hypoechoic appearance, and absence of cystic change favored a follicular carcinoma diagnosis, although there was substantial overlap in these sonographic features between the two groups.30 Rarely, FTC will present with aggressive and widely metastatic disease as has been reported with lung, bone, and even parotid gland metastases during initial evaluation and staging.31–33
Cytopathology
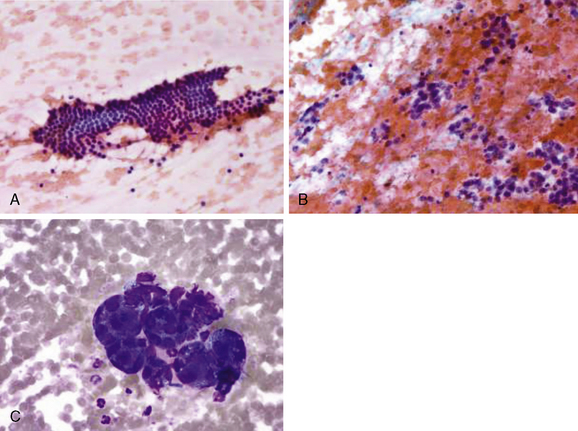
Perhaps no other portion of the diagnostic workup for thyroid nodules causes as much consternation as the interpretation and differentiation of a benign follicular lesion versus a follicular carcinoma on cytology from a thyroid nodule fine-needle aspiration (FNA) biopsy (see Chapter 12, Fine-Needle Aspiration of the Thyroid Gland). Cytopathology expertise and communication between the clinician and cytopathologist are critical to appropriately categorize the risk of malignancy in follicular lesions. In 2007, the National Cancer Institute convened a state of the science meeting to attempt to consolidate and provide guidance regarding thyroid cytopathology terminology.34 This resulted in six distinct categories of thyroid FNA cytology readings, including benign, atypia of undetermined significance/follicular lesion of undetermined significance, follicular/Hurthle neoplasm, suspicious for malignancy, PTC, and nondiagnostic. Note that FTC and suspicious for FTC are not among these categories (Figure 20-2). The functional definition of FTC as opposed to a benign follicular adenoma is invasion of the tumor through the tumor capsule, which cannot be determined on an FNA cytology sample. As opposed to classic PTC, which has characteristic cellular and nuclear features, FTC can have bland-appearing follicular epithelium and only subtle changes suggestive of carcinoma. Additionally, in nodules larger than 4 cm, the false-negative rate of FNA can increase from the approximately 3% to 5% rate for smaller nodules to as high as 20%. In a recent study of nodules > 4 cm, 14% of “benign” biopsies turned out to be FTC on final histopathology.35 Similarly, in a study where cytologically indeterminate nodules were classified as follicular neoplasm (as opposed to follicular lesion), 37.5% of nodules > 4 cm in diameter were malignant as opposed to the 21% rate found in all nodules. The majority of the malignancies in the follicular neoplasm category were FTC at final histopathology.36 In the absence of a definitive diagnosis of FTC on FNA, a variety of cytologic, clinical, US, biochemical, and other features can help the practitioner estimate risk to aid in recommending surgery or conservative follow-up. In a recent study combining clinical and cytologic features of follicular lesions, the only significant clinical factor predicting malignancy was nodule size > 4 cm.37 Cytologic features predicting malignancy included transgressing vessels, anisokaryosis, nuclear pleomorphism, greater percentage of single cells, increased cellularity with crowding, nuclear grooves and atypia, and fewer macrofollicular formations. As an aid to risk stratify patients with an indeterminate FNA biopsy result, recent compelling data provide new evidence that higher serum thyroid-stimulating hormone (TSH) is directly associated with both risk of cancer in thyroid nodules as well as cancer aggressiveness.38 Finally, in the setting of an indeterminate thyroid nodule FNA, the American Thyroid Association guidelines suggest a thyroid scan (123I or 99Tc) can help stratify malignancy risk in a thyroid nodule (lower if there is relatively high uptake in the nodule compared to surrounding thyroid and higher if there is a cold defect on scan that correlates with the nodule).29,39 In a large subset of patients, definitive diagnosis cannot be made without surgical resection and surgical pathology assessment.
Histopathology
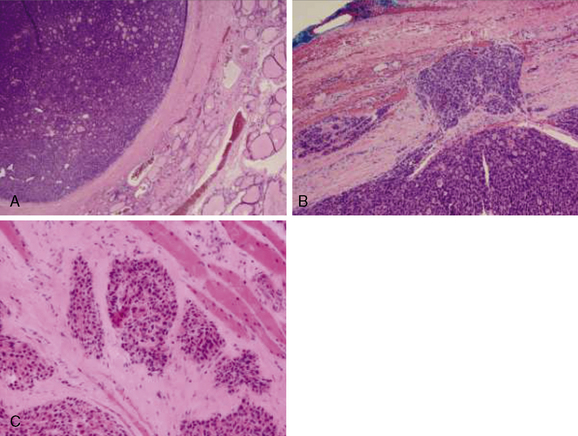
Follicular lesions of the thyroid can be separated into three distinct clinical entities: follicular adenomas, minimally invasive follicular carcinomas, and widely invasive follicular carcinomas (Figure 20-3). The proper diagnosis depends on a complete and thorough evaluation of the entire tumor capsule. Even with a complete appraisal of the tumor capsule, however, the absolute definition of a minimally invasive FTC is controversial.40 Minimally invasive FTC is basically defined as tumor penetration or disruption of the tumor capsule in at least one area. Even within this definition, there appears to be a relapse-free survival benefit when there are fewer than four foci of disruption/penetration in the tumor capsule or blood vessels.41,42 Minimally invasive FTC tends to behave like follicular adenomas and generally does not need remnant ablation or adjuvant therapy with radioiodine.43 FTC more commonly undergoes hematogenous spread as opposed to direct lymphatic spread as is seen in PTC. This is likely the reason for a higher risk of distant metastases with FTC as compared to PTC.32,44 However, the suggestion that FTC is never associated with regional lymph node metastases is incorrect and surgical planning should not be based on this misinformation.44,45 Patients with lesions suspicious for FTC by cytopathology undergoing surgical resection should still have a formal preoperative US to evaluate for lymph node metastases.29
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree